Abstract
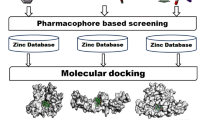
Toll-like receptor 7 (TLR7) is highly expressed in dendritic cells (DCs) and B cells, and its aberrant activation can promote disease progression in systemic lupus erythematosus (SLE). We utilized structure-based virtual screening and experimental validation to screen natural products from TargetMol for potential TLR7 antagonists. Our results of molecular docking and molecular dynamics simulation showed that Mogroside V (MV) strongly interacted with TLR7, with stable open-TLR7-MV and close-TLR7-MV complexes. Furthermore, in vitro experiments demonstrated that MV significantly inhibited B cell differentiation in a concentration-dependent manner. In addition to TLR7, we also revealed a strong interaction of MV with all TLRs, including TLR4. The above results suggested that MV might be a potential TLR7 antagonist deserving of further study.








Similar content being viewed by others
Data availability
The all datasets are available from the corresponding author.
Abbreviations
- TLR7:
-
Toll-like receptor 7
- SLE:
-
Systemic lupus erythematosus
- HCQ:
-
Hydroxychloroquine
- MD:
-
Molecular dynamic simulation
- PCA:
-
Principal component analysis
- RMSD:
-
The root-mean-square deviations
- RMSF:
-
The root-mean-square fluctuation
- SASA:
-
The solvent-accessible surface area
- FELs:
-
Free energy landscapes
- PBMCs:
-
Peripheral blood mononuclear cells
References
Brown GJ, Cañete PF, Wang H et al (2022) TLR7 gain-of-function genetic variation causes human lupus. Nature 605:349–356. https://doi.org/10.1038/s41586-022-04642-z
Santiago-Raber M-L, Dunand-Sauthier I, Wu T et al (2010) Critical role of TLR7 in the acceleration of systemic lupus erythematosus in TLR9-deficient mice. J Autoimmun 34:339–348. https://doi.org/10.1016/j.jaut.2009.11.001
Christensen SR, Shupe J, Nickerson K et al (2006) Toll-like receptor 7 and TLR9 dictate autoantibody specificity and have opposing inflammatory and regulatory roles in a murine model of lupus. Immunity 25:417–428. https://doi.org/10.1016/j.immuni.2006.07.013
Pisitkun P, Deane JA, Difilippantonio MJ et al (2006) Autoreactive B cell responses to RNA-related antigens due to TLR7 gene duplication. Science 312:1669–1672. https://doi.org/10.1126/science.1124978
Subramanian S, Tus K, Li Q-Z et al (2006) A Tlr7 translocation accelerates systemic autoimmunity in murine lupus. Proc Natl Acad Sci USA 103:9970–9975. https://doi.org/10.1073/pnas.0603912103
Goel RR, Wang X, O’Neil LJ et al (2020) Interferon lambda promotes immune dysregulation and tissue inflammation in TLR7-induced lupus. Proc Natl Acad Sci USA 117:5409–5419. https://doi.org/10.1073/pnas.1916897117
Tojo S, Zhang Z, Matsui H et al (2020) Structural analysis reveals TLR7 dynamics underlying antagonism. Nat Commun 11:5204. https://doi.org/10.1038/s41467-020-19025-z
Murakami Y, Fukui R, Tanaka R et al (2021) Anti-TLR7 antibody protects against lupus nephritis in NZBWF1 mice by targeting B cells and patrolling monocytes. Front Immunol 12:777197. https://doi.org/10.3389/fimmu.2021.777197
Santiago-Raber M-L, Baudino L, Izui S (2009) Emerging roles of TLR7 and TLR9 in murine SLE. J Autoimmun 33:231–238. https://doi.org/10.1016/j.jaut.2009.10.001
Desnues B, Macedo AB, Roussel-Queval A et al (2014) TLR8 on dendritic cells and TLR9 on B cells restrain TLR7-mediated spontaneous autoimmunity in C57BL/6 mice. Proc Natl Acad Sci USA 111:1497–1502. https://doi.org/10.1073/pnas.1314121111
Jenks SA, Cashman KS, Zumaquero E et al (2018) Distinct effector B cells induced by unregulated toll-like receptor 7 contribute to pathogenic responses in systemic lupus erythematosus. Immunity 49:725-739.e6. https://doi.org/10.1016/j.immuni.2018.08.015
Fillatreau S, Manfroi B, Dörner T (2021) Toll-like receptor signalling in B cells during systemic lupus erythematosus. Nat Rev Rheumatol 17:98–108. https://doi.org/10.1038/s41584-020-00544-4
Wallace DJ, Gudsoorkar VS, Weisman MH, Venuturupalli SR (2012) New insights into mechanisms of therapeutic effects of antimalarial agents in SLE. Nat Rev Rheumatol 8:522–533. https://doi.org/10.1038/nrrheum.2012.106
Flares after hydroxychloroquine reduction or discontinuation: results from the Systemic Lupus International Collaborating Clinics (SLICC) inception cohort - PMC. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8862090/#R4. Accessed 3 Feb 2023
Schrezenmeier E, Dörner T (2020) Mechanisms of action of hydroxychloroquine and chloroquine: implications for rheumatology. Nat Rev Rheumatol 16:155–166. https://doi.org/10.1038/s41584-020-0372-x
Dai P, Ruan P, Mao Y et al (2022) The antiviral efficacies of small-molecule inhibitors against respiratory syncytial virus based on the F protein. J Antimicrob Chemother. https://doi.org/10.1093/jac/dkac370
Vlach J, Bender AT, Przetak M et al (2021) Discovery of M5049: a novel selective toll-like receptor 7/8 inhibitor for treatment of autoimmunity. J Pharmacol Exp Ther 376:397–409. https://doi.org/10.1124/jpet.120.000275
Hansson T, Oostenbrink C, van Gunsteren W (2002) Molecular dynamics simulations. Curr Opin Struct Biol 12:190–196. https://doi.org/10.1016/s0959-440x(02)00308-1
Vanommeslaeghe K, Hatcher E, Acharya C et al (2010) CHARMM general force field: a force field for drug-like molecules compatible with the CHARMM all-atom additive biological force fields. J Comput Chem 31:671–690. https://doi.org/10.1002/jcc.21367
Pires DEV, Blundell TL, Ascher DB (2015) pkCSM: predicting small-molecule pharmacokinetic and toxicity properties using graph-based signatures. J Med Chem 58:4066–4072. https://doi.org/10.1021/acs.jmedchem.5b00104
Wang N, Gao J-G, Wu M-W (2023) Molecular docking and molecular simulation studies for N-degron selectivity of chloroplastic ClpS from Chlamydomonas reinhardtii. Comput Biol Chem 103:7825. https://doi.org/10.1016/j.compbiolchem.2023.107825
Yang M, Long D, Hu L et al (2021) AIM2 deficiency in B cells ameliorates systemic lupus erythematosus by regulating Blimp-1–Bcl-6 axis-mediated B-cell differentiation. Signal Transduct Target Ther 6:341. https://doi.org/10.1038/s41392-021-00725-x
Fu X, Du Y, Zou L et al (2022) Acidified glycerol as a one-step efficient green extraction and preservation strategy for anthocyanin from blueberry pomace: New insights into extraction and stability protection mechanism with molecular dynamic simulation. Food Chem 390:133226. https://doi.org/10.1016/j.foodchem.2022.133226
Souyris M, Cenac C, Azar P et al (2018) TLR7 escapes X chromosome inactivation in immune cells. Sci Immunol 3:eaap8855. https://doi.org/10.1126/sciimmunol.aap8855
Huang C, Yi P, Zhu M et al (2022) Safety and efficacy of fecal microbiota transplantation for treatment of systemic lupus erythematosus: An EXPLORER trial. J Autoimmun 130:102844. https://doi.org/10.1016/j.jaut.2022.102844
Mohamed A, Chen Y, Wu H et al (2019) Therapeutic advances in the treatment of SLE. Int Immunopharmacol 72:218–223. https://doi.org/10.1016/j.intimp.2019.03.010
Balak DMW, van Doorn MBA, Arbeit RD et al (2017) IMO-8400, a toll-like receptor 7, 8, and 9 antagonist, demonstrates clinical activity in a phase 2a, randomized, placebo-controlled trial in patients with moderate-to-severe plaque psoriasis. Clin Immunol 174:63–72. https://doi.org/10.1016/j.clim.2016.09.015
Bou Karroum N, Moarbess G, Guichou J-F et al (2019) Novel and Selective TLR7 Antagonists among the Imidazo[1,2-a]pyrazines, Imidazo[1,5-a]quinoxalines, and Pyrazolo[1,5-a]quinoxalines Series. J Med Chem 62:7015–7031. https://doi.org/10.1021/acs.jmedchem.9b00411
Lamphier M, Zheng W, Latz E et al (2014) Novel small molecule inhibitors of TLR7 and TLR9: mechanism of action and efficacy in vivo. Mol Pharmacol 85:429–440. https://doi.org/10.1124/mol.113.089821
Acknowledgements
This work was supported by the China Postdoctoral Science Foundation (2022M723563) and Natural Science Foundation of Hunan Province (2022JJ40722).
Author information
Authors and Affiliations
Contributions
PL R wrote the manuscript; YJ C, M Y and SS W did the editing; M Y designed and revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ruan, P., Wang, S., Yi, P. et al. Identification of the potential TLR7 antagonists by virtual screening and experimental validation. Mol Divers (2023). https://doi.org/10.1007/s11030-023-10660-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11030-023-10660-4