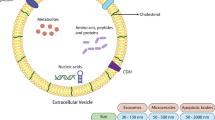
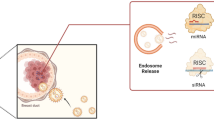
Abstract
Exosomes have come to the fore in drug delivery systems due to their biological-based and immune-suppressing properties. In this study, we investigated the effect of doxorubicin loading of exosomes isolated from human platelets on breast cancer.
Exosomes released from ADP (1 µM)-activated platelets were isolated by the ultracentrifugation method, and their size and charge were measured with a TEM and zeta sizer. Then doxorubicin (Dox) loading into exosomes (PLT-Exo-Dox) was done by electroporation and incubated with MDA-MB-231 cells. In exosome characterization, CD62 positivity was higher in platelet pellets, while CD9 positivity was higher in released exosomes. The size of PLT-Exo and PLT-Exo-Dox was 82.02 ± 5.21 nm and 116 ± 3.73 nm, with a polydispersity index of 0.26 ± 0.04 and 0.39 ± 0.06, and the Zeta potential was − 16.45 mV and 24.07 mV, respectively. The encapsulation efficiency of the preparation was 86.02 ± 6.16%, with a drug loading capacity of 4.75 ± 0.16 µg/µg of the exosome. In MDA-MB-231 cells, PLT-Exo increased cell viability, while PLT-Exo-Dox decreased in 24 h. The Annexin-V binding level and Bax gene expression were increased in PLT-Exo-Dox and Bcl-2 gene expression was decreased. This study will shed light on the development of release systems that can be effective with chemotherapeutic agents by using exosomes released by cells in the development of personalized treatments.
Graphical abstract






Similar content being viewed by others
Data Availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Hakim NS, Canelo R (2007) Haemostasis in surgery. Imperial College Press, London
Machlus KR, Italiano JE Jr (2013) The incredible journey: from megakaryocyte development to platelet formation. J Cell Biol 201:785–796. https://doi.org/10.1083/jcb.201304054
Holinstat M (2017) Normal platelet function. Cancer Metastasis Rev 36:195–198. https://doi.org/10.1007/s10555-017-9677-x
Gresele P, Malvestiti M, Momi S (2018) Anti-platelet treatments in cancer: basic and clinical research. Thromb Res 164:S106–S111
Nooshabadi VT, Khanmohammadi M, Shafei S, Banafshe HR, Malekshahi ZV, Ebrahimi-Barough S et al (2020) Impact of atorvastatin loaded exosome as an anti-glioblastoma carrier to induce apoptosis of U87 cancer cells in 3D culture model. Biochem Biophys Reports 23:100792
Cevik O, Li D, Baljinnyam E, Manvar D, Pimenta EM, Waris G et al (2017) Interferon regulatory factor 5 (IRF5) suppresses hepatitis C virus (HCV) replication and HCV-associated hepatocellular carcinoma. J Biol Chem 292:21676–21689
Cenik M, Abas B, Kocabıyık B, Demirbolat G, Cevik O. Development of a new drug delivery system from HELA-derived exosomes and the effect of docetaxel-loaded exosomes on mitochondrial apoptosis. J Pharm Innov. 2021;
Siegel RL, Miller KD, Jemal A (2020) Cancer statistics, 2020. CA Cancer J Clin 70:7–30. https://doi.org/10.3322/caac.21590
Prados J, Melguizo C, Ortiz R, Vélez C, Alvarez PJ, Arias JL et al (2012) Doxorubicin-loaded nanoparticles : new advances in breast cancer therapy. Anticancer Agents Med Chem 12(9):1058–1070
Coelho SC, Pereira MC, Juzeniene A, Juzenas P, Coelho MAN (2015) Supramolecular nanoscale assemblies for cancer diagnosis and therapy. J Control Release 213:152–167
Curigliano G, Criscitiello C (2014) Successes and limitations of targeted cancer therapy in breast cancer. Prog Tumor Res 41:15–35
Hernandez-Aya LF, Gonzalez-Angulo AM (2013) Adjuvant systemic therapies in breast cancer. Surg Clin N Am 93:473–491
Petros RA, DeSimone JM (2010) Strategies in the design of nanoparticles for therapeutic applications. Nat Rev Drug Discov 9:615–627. https://doi.org/10.1038/nrd2591
Di Francesco M, Celia C, Cristiano MC, d’Avanzo N, Ruozi B, Mircioiu C et al (2021) Doxorubicin hydrochloride-loaded nonionic surfactant vesicles to treat metastatic and non-metastatic breast cancer. ACS Omega 6:2973–2989
Sun T, Zhang YS, Pang B, Hyun DC, Yang M, Xia Y (2014) Engineered nanoparticles for drug delivery in cancer therapy. Angew Chem Int Ed 53:12320–12364. https://doi.org/10.1002/anie.201403036
Luan X, Sansanaphongpricha K, Myers I, Chen H, Yuan H, Sun D (2017) Engineering exosomes as refined biological nanoplatforms for drug delivery. Acta Pharmacol Sin 38:754–763. https://doi.org/10.1038/aps.2017.12
Kim MS, Haney MJ, Zhao Y, Mahajan V, Deygen I, Klyachko NL et al (2016) Development of exosome-encapsulated paclitaxel to overcome MDR in cancer cells. Nanomed Nanotechnol 12:655–664
Dai J, Su Y, Zhong S, Cong L, Liu B, Yang J et al (2020) Exosomes: key players in cancer and potential therapeutic strategy. Signal Transduct Target Ther 5:145
Kim H, Lee S, Shin E, Seong KM, Jin YW, Youn H et al (2020) The emerging roles of exosomes as EMT regulators in cancer. Cells 9:861
Li M, Xia B, Wang Y, You MJ, Zhang Y (2019) Potential therapeutic roles of exosomes in multiple myeloma: a systematic review. J Cancer 10:6154–6160
Jurasz P, Alonso-Escolano D, Radomski MW (2004) Platelet–cancer interactions: mechanisms and pharmacology of tumour cell-induced platelet aggregation. Br J Pharmacol 143:819–826
Bambace NM, Holmes CE (2011) The platelet contribution to cancer progression. J Thromb Haemost 9:237–249. https://doi.org/10.1111/j.1538-7836.2010.04131.x
Liu X, Wang L, Ma C, Wang G, Zhang Y, Sun S (2019) Exosomes derived from platelet-rich plasma present a novel potential in alleviating knee osteoarthritis by promoting proliferation and inhibiting apoptosis of chondrocyte via Wnt/β-catenin signaling pathway. J Orthop Surg Res 14:470
Taus F, Meneguzzi A, Castelli M, Minuz P (2019) Platelet-derived extracellular vesicles as target of antiplatelet agents. What is the evidence? Front Pharmacol. https://doi.org/10.3389/fphar.2019.01256
Rui S, Yuan Y, Du C, Song P, Chen Y, Wang H et al (2021) Comparison and investigation of exosomes derived from platelet-rich plasma activated by different agonists. Cell Transplant 30:1–13
Goetzl EJ, Goetzl L, Karliner JS, Tang N, Pulliam L (2016) Human plasma platelet-derived exosomes: effects of aspirin. FASEB J 30:2058–2063. https://doi.org/10.1096/fj.201500150R
Stranska R, Gysbrechts L, Wouters J, Vermeersch P, Bloch K, Dierickx D et al (2018) Comparison of membrane affinity-based method with size-exclusion chromatography for isolation of exosome-like vesicles from human plasma. J Transl Med 16:1. https://doi.org/10.1186/s12967-017-1374-6
Pordzik J, Pisarz K, De Rosa S, Jones AD, Eyileten C, Indolfi C et al (2018) The potential role of platelet-related microRNAs in the development of cardiovascular events in high-risk populations, including diabetic patients: a review. Front Endocrinol (Lausanne) 9:74
Janowska-Wieczorek A, Wysoczynski M, Kijowski J, Marquez-Curtis L, Machalinski B, Ratajczak J et al (2005) Microvesicles derived from activated platelets induce metastasis and angiogenesis in lung cancer. Int J Cancer 113:752–760. https://doi.org/10.1002/ijc.20657
Torreggiani E, Perut F, Roncuzzi L, Zini N, Baglìo SR, Baldini N (2014) Exosomes: novel effectors of human platelet lysate activity. Eur Cells Mater 28:137–151
Valadi H, Ekström K, Bossios A, Sjöstrand M, Lee JJ, Lötvall JO (2007) Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol 9:654–659. https://doi.org/10.1038/ncb1596
Iyer SR, Scheiber AL, Yarowsky P, Henn RF, Otsuru S, Lovering RM (2020) Exosomes isolated from platelet-rich plasma and mesenchymal stem cells promote recovery of function after muscle injury. Am J Sports Med 48:2277–2286
Kailashiya J, Gupta V, Dash D (2019) Engineered human platelet-derived microparticles as natural vectors for targeted drug delivery. Oncotarget 10:5835–5846
Hadla M, Palazzolo S, Corona G, Caligiuri I, Canzonieri V, Toffoli G et al (2016) Exosomes increase the therapeutic index of doxorubicin in breast and ovarian cancer mouse models. Nanomedicine 11:2431–2441. https://doi.org/10.2217/nnm-2016-0154
Zhang W, Jiang H, Kong Y (2020) Exosomes derived from platelet-rich plasma activate YAP and promote the fibrogenic activity of Müller cells via the PI3K/Akt pathway. Exp Eye Res 193:107973
Kim MS, Haney MJ, Zhao Y, Yuan D, Deygen I, Klyachko NL et al (2018) Engineering macrophage-derived exosomes for targeted paclitaxel delivery to pulmonary metastases: in vitro and in vivo evaluations. Nanomed Nanotechnol 14:195–204
Liu Y, Zhao Y, Wang Y, Zhu P, Wei Z, Wang S et al (2017) Suppressive role of diallyl trisulfide in the activated platelet-mediated hematogenous metastasis of MDA-MB-231 human breast cancer cells. Int J Mol Med 39:1516–1524. https://doi.org/10.3892/ijmm.2017.2953
Gockel LM, Ponert JM, Schwarz S, Schlesinger M, Bendas G (2018) The low molecular weight heparin tinzaparin attenuates platelet activation in terms of metastatic niche formation by coagulation-dependent and independent pathways. Molecules 23:2753
Schindler C, Collinson A, Matthews C, Pointon A, Jenkinson L, Minter RR et al (2019) Exosomal delivery of doxorubicin enables rapid cell entry and enhanced in vitro potency. PLoS ONE 14:4545. https://doi.org/10.1371/journal.pone.0214545
Toffoli G, Hadla M, Corona G, Caligiuri I, Palazzolo S, Semeraro S et al (2015) Exosomal doxorubicin reduces the cardiac toxicity of doxorubicin. Nanomedicine 10:2963–2971. https://doi.org/10.2217/nnm.15.118
Wiklander OPB, Nordin JZ, O’Loughlin A, Gustafsson Y, Corso G, Mäger I et al (2015) Extracellular vesicle in vivo biodistribution is determined by cell source, route of administration and targeting. J Extracell Vesicles 4:26316. https://doi.org/10.3402/jev.v4.26316
Tian Y, Li S, Song J, Ji T, Zhu M, Anderson GJ et al (2014) A doxorubicin delivery platform using engineered natural membrane vesicle exosomes for targeted tumor therapy. Biomaterials 35:2383–2390
Ohno S, Takanashi M, Sudo K, Ueda S, Ishikawa A, Matsuyama N et al (2013) Systemically injected exosomes targeted to EGFR deliver antitumor microRNA to breast cancer cells. Mol Ther 21:185–191. https://doi.org/10.1038/mt.2012.180
Acknowledgements
This work was supported by Aydin Adnan Menderes University Research Grant (ADU-TPF-20042).
Funding
This work was supported by Aydin Adnan Menderes University Research Grant (ADU-TPF-20042).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by DU, BIA, GMD. The first draft of the manuscript was written by OC and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The author reports no conflict of interest relevant to this article.
Ethical approval
This study was approved from Adnan Menderes University Ethics Committee (53043469-050.04.04/12). All methods were performed in accordance with the relevant guidelines and regulations and were approved by our ethics committee.
Consent to PARTICIPATE
Written informed consent was obtained from all volunteers.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Uslu, D., Abas, B.I., Demirbolat, G.M. et al. Effect of platelet exosomes loaded with doxorubicin as a targeted therapy on triple-negative breast cancer cells. Mol Divers 28, 449–460 (2024). https://doi.org/10.1007/s11030-022-10591-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-022-10591-6