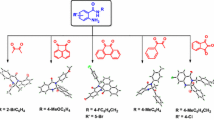
Abstract
An efficient and simple protocol for the synthesis of trifluoromethylated quinazolines has been described by I2-/KI-promoted oxidative C(sp3)–C(sp2) bond under the optimal oxidative cyclization reaction conditions. The required 2,2,2-trifluoro-N-benzyl-N′-arylacetimidamides are readily prepared from the corresponding acetimidoyl chlorides and benzylamines under a nucleophilic substitution reaction in the form of in situ. The merits of this protocol are the use of inexpensive molecular iodine, metal-free oxidative coupling and good to excellent yields.







Similar content being viewed by others
References
Ugale VG, Bari SB (2014) Quinazolines: new horizons in anticonvulsant therapy. Eur J Med Chem 80:447–501. https://doi.org/10.1016/j.ejmech.2014.04.072
Alafeefy AM, Kadi AA, Al-Deeb OA, El-Tahir KE, Aljaber NA (2010) Synthesis, analgesic and anti-inflammatory evaluation of some novel quinazoline derivatives. Eur J Med Chem 45:4947–4952. https://doi.org/10.1016/j.ejmech.2010.07.067
Marzaro G, Guiotto A, Chilin A (2012) Quinazoline derivatives as potential anticancer agents: a patent review (2007–2010). Expert Opin Ther Pat 22:223–252. https://doi.org/10.1517/13543776.2012.665876
Mahdavi M, Lotfi V, Saeedi M, Kianmehr E, Shafiee A (2016) Synthesis of novel fused quinazolinone derivatives. Mol Divers 20:677–685. https://doi.org/10.1007/s11030-016-9675-x
Khan I, Ibrar A, Abbas N, Saeed A (2014) Recent advances in the structural library of functionalized quinazoline and quinazolinone scaffolds: synthetic approaches and multifarious applications. Eur J Med Chem 76:193–244. https://doi.org/10.1016/j.ejmech.2014.02.005
Ge WL, Zhu X, Wei YY (2013) Iodine-catalyzed oxidative system for cyclization of primary alcohols with o-aminobenzamides to quinazolinones using DMSO as the oxidant in dimethyl carbonate. RSC Adv 3:10817–10822. https://doi.org/10.1039/C3RA40872H
Machara A, Lux V, Kozisek M, Grantz-Saskova K, Stepanek O, Kotora M, Parkan K, Pavova M, Glass B, Sehr PJ (2016) Specific inhibitors of HIV capsid assembly binding to the c-terminal domain of the capsid protein: evaluation of 2-arylquinazolines as potential antiviral compounds. Med Chem 59:545–558. https://doi.org/10.1021/acs.jmedchem.5b01089
Liu LT, Yuan TT, Liu HH (2007) Synthesis and biological evaluation of substituted 6-alkynyl-4-anilinoquinazoline derivatives as potent EGFR inhibitors. Bioorg Med Chem Lett 17:6373. https://doi.org/10.1016/j.bmcl.2007.08.061
Michael JP (2008) Quinoline, quinazoline and acridone alkaloids Nat. Prod Rep 25:166. https://doi.org/10.1039/B612168N
Luth A, Lowe W (2008) Syntheses of 4-(indole-3-yl)quinazolines-A new class of epidermal growth factor receptor tyrosine kinase inhibitors. Eur J Med Chem 43:1478. https://doi.org/10.1016/j.ejmech.2007.09.018
Fang J, Zhou JG, Fang ZJ (2013) Synthesis of 2-substituted quinazolines via iridium catalysis. RSC Adv 3:334. https://doi.org/10.1039/C2RA22278G
Zhang J, Yu C, Wang S, Wan C, Wang Z (2010) A novel and efficient methodology for the construction of quinazolines based on supported copper oxide nanoparticles. Chem Commun 46:5244–5246. https://doi.org/10.1039/C002454F
Panja SK, Saha S (2013) Recyclable, magnetic ionic liquid bmim[FeCl4]-catalyzed, multicomponent, solvent-free, green synthesis of quinazolines. RSC Adv 3:14495. https://doi.org/10.1039/C3RA42039F
Portela-Cubillo F, Scott JS, Walton JC (2009) Microwave-promoted syntheses of quinazolines and dihydroquinazolines from 2-aminoarylalkanone O-phenyl oximes. J Org Chem 74:4934–4942. https://doi.org/10.1021/jo900629g
Ju J, Hua R, Su J (2012) Copper-catalyzed three-component one-pot synthesis of quinazolines. Tetrahedron 68:9364–9370. https://doi.org/10.1016/j.tet.2012.09.035
Truong VL, Morrow M (2010) Mild and efficient ligand-free copper-catalyzed condensation for the synthesis of quinazolines. Tetrahedron Lett 51:758–760. https://doi.org/10.1016/j.tetlet.2009.11.133
Malakar CC, Baskakova A, Conrad J, Beifuss U (2012) Copper-catalyzed synthesis of quinazolines in water starting from o-Bromobenzylbromides and Benzamidines. Chem Eur J 18:8882–8885. https://doi.org/10.1002/chem.201200583
Omar MA, Conrad J, Beifuss U (2014) Assembly of 4H-chromenes, imidazobenzothiazines and quinazolines via copper-catalyzed domino reactions using 2-halobenzyl tosylates as substrates. Tetrahedron 70:5682–5695. https://doi.org/10.1016/j.tet.2014.06.071
Omar MA, Conrad J, Beifuss U (2014) Copper-catalyzed domino reaction between 1-(2-halophenyl) methanamines and amidines or imidates for the synthesis of 2-substituted quinazolines. Tetrahedron 70:3061–3072. https://doi.org/10.1016/j.tet.2014.02.066
Lin JP, Zhang FH, Long YQ (2014) Solvent/oxidant-switchable synthesis of multisubstituted quinazolines and benzimidazoles via metal-free selective oxidative annulation of arylamidines. Org Lett 16:2822. https://doi.org/10.1021/ol500864r
Wang CS, Li F, Liu HX, Jiang YY, Fu H (2010) Copper-catalyzed synthesis of quinazoline derivatives via Ullmann-type coupling and aerobic oxidation. J Org Chem 75:7936. https://doi.org/10.1021/jo101685d
Zhang W, Guo F, Wang F, Zhao N, Liu L, Li J, Wang ZH (2014) Synthesis of quinazolines via CuO nanoparticles catalyzed aerobic oxidative coupling of aromatic alcohols and amidines. Org Biomol Chem 12:5752. https://doi.org/10.1039/C4OB00569D
Zhao D, Shen Q, Li JX (2015) Potassium iodide-catalyzed three-component synthesis of 2-arylquinazolines via amination of benzylic C–H bonds of methylarenes. Adv Synth Catal 357:339. https://doi.org/10.1002/adsc.201400827
Lv Z, Wang B, Hu Z, Zhou Y, Yu W, Chang J (2016) Synthesis of quinazolines from N,N′-disubstituted amidines via I2/KI-mediated oxidative C–C bond formation. J Org Chem 81:9924–9930. https://doi.org/10.1021/acs.joc.6b02100
Rahmani F, Darehkordi A (2017) Synthesis of trifluoromethylated pyrroles via a one-pot three-component reaction. Synlett 28:1224–1226. https://doi.org/10.1055/s-0036-1588732
Darehkordi A, Rahmani F, Hashemi V (2013) Synthesis of new trifluoromethylated indole derivatives. Tetrahedron Lett 54:4689–4692. https://doi.org/10.1016/j.tetlet.2013.06.093
Darehkordi A, Rahmani F (2016) Synthesis of new α-trifluoromethyl substituted formamidines framework by using N-nucleophiles and N, S bidentate nucleophiles. J Fluorine Chem 190:41–47. https://doi.org/10.1016/j.jfluchem.2016.08.010
Pang X, Chen C, Su X, Li M, Wen L (2014) Diverse tandem cyclization reactions of o-cyanoanilines and diaryliodonium salts with copper catalyst for the construction of quinazolinimine and acridine scaffolds. Org Lett 16:6228. https://doi.org/10.1021/ol503156g
Liu Q, Zhao YF, Fu H, Cheng CM (2013) Copper-catalyzed sequential n-arylation and aerobic oxidation: synthesis of quinazoline derivatives. Synlett 24:2089. https://doi.org/10.1055/s-0033-1339800
Liu Q, Zhang Q (2013) Copper-catalyzed annulation of amidines for quinazoline synthesis. Chem Commun 49:6439. https://doi.org/10.1039/C3CC43129K
Finkbeiner P, Nachtsheim B (2013) Iodine in modern oxidation catalysis. J. Synth 45:979. https://doi.org/10.1055/s-0032-1318330
Guo X, Pan S, Liu J, Li ZJ (2009) One-pot synthesis of symmetric and unsymmetric 1,1-bis-indolylmethanes via tandem iron-catalyzed C–H bond oxidation and C–O bond cleavage. Org Chem 74:8848. https://doi.org/10.1021/jo902093p
Li H, He Z, Guo X, Li W, Zhao X, Li Z (2009) Iron-catalyzed selective oxidation of n-methyl amines: highly efficient synthesis of methylene-bridged bis-1,3-dicarbonyl compounds. Org Lett 11:4176. https://doi.org/10.1021/ol901751c
Li Z, Cao L, Li C (2007) FeCl2-catalyzed selective C–C bond formation by oxidative activation of a benzylic C–H bond. J Angew Chem Int Ed 46:6505
Parvatkar PT, Parameswaran PS, Tilve SG (2012) FeCL2-catalyzed selective C-C bond formation by oxidative activation of a benzylic C–H bond. Chem Eur J 18:5460–5489. https://doi.org/10.1002/anie.200701782
Cheng X, Wang H, Xiao F, Deng GJ (2016) Lewis acid-catalyzed 2-arylquinazoline formation from N′-arylbenzimidamides and paraformaldehyde. Green Chem 18:5773–5776. https://doi.org/10.1039/C6GC02319C
Acknowledgements
We gratefully acknowledge the Vail-e-Asr University of Rafsanjan Faculty Research Grant for financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Darehkordi, A., Kazemi, E. Iodine/potassium iodide catalyst for the synthesis of trifluoromethylated quinazolines via intramolecular cyclization of 2,2,2-trifluoro-N-benzyl-N′-arylacetimidamides. Mol Divers 24, 131–139 (2020). https://doi.org/10.1007/s11030-019-09933-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-019-09933-8