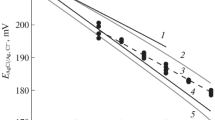
The technique of manufacturing silver–silver chloride electrodes and the method of their subsequent selection for the State Primary Standard of pH activity of hydrogen ions in aqueous solutions are described.



Similar content being viewed by others
References
Yu. Yu. Lurie, Analytical Chemistry of Industrial Wastewater, Khimiya, Moscow (1984).
G. S. Fomin, Water. Control of Chemical, Bacterial, and Radiation Safety by International Standards, Protektor, Moscow (2010).
D. A. Skoog, D. M. West, F. J. Holler, and S. R. Crouch, Fundamentals of Analytical Chemistry, Brooks/Cole-Thompson Learning, Belmont, US (2004), 8th ed.
P. Quevauviller and K. C. Thompson, Analytical Methods for Drinking Water: Advances in Sampling and Analysis, Wiley & Sons Ltd., The Atrium, Southern Gate, Chichester, West Sussex PO19 8SQ, England (2006).
GOST 8.120–2014, GSI. State Verification Schedule for Means of pH Measurement.
R. P. Buck, S. Rondinini, A. K. Covington, et al., “Measurement of pH. Definition, standards, and procedures,” Pure Appl. Chem., 74, No. 11, 2169–2200 (2002).
R. Bates, Theory and Practice of pH Measurement [Russian translation], Khimiya, Leningrad (1972).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Izmeritel’naya Tekhnika, No. 9, pp. 68–70, September, 2016.
Rights and permissions
About this article
Cite this article
Dobrovol’skii, V.I., Prokunin, S.V., Morozov, I.V. et al. Investigation of Metrological Characteristics of Silver–Silver Chloride Electrodes of GET 54-2011, the State Primary Standard for the pH Activity of Hydrogen Ions in Aqueous Solutions. Meas Tech 59, 1013–1016 (2016). https://doi.org/10.1007/s11018-016-1085-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11018-016-1085-z