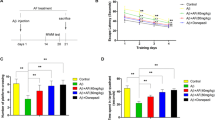
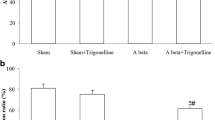
Abstract
Diosgenin is a neurosteroid derived from the plants and has been previously reported for its numerous health beneficial properties, such as anti-arrhythmic, hypolipidemic, and antiproliferative effects. Although several studies conducted earlier suggested cognition enhancement actions of diosgenin against neurodegenerative disorders, but the molecular mechanisms underlying are not clearly understood. In the present study, we investigated the neuroprotective effect of diosgenin in the Wistar rats that received an intracerebroventricular injection of Amyloid-β (1–42) peptides, representing a rodent model of Alzheimer’s disease (AD). Animals were treated with 100 and 200 mg/kg/p.o of diosgenin for 28 days, followed by Amyloid-β (1–42) peptides infusion. Animals were assessed for the spatial learning and memory by using radial arm maze and passive avoidance task. Subsequently, animals were euthanized and brains were collected for biochemical estimations and histopathological studies. Our results revealed that, diosgenin administration dose dependently improved the spatial learning and memory and protected the animals from Amyloid-β (1–42) peptides induced disrupted cognitive functions. Further, biochemical analysis showed that diosgenin successfully attenuated Amyloid-β (1–42) mediated plaque load, oxidative stress, neuroinflammation and elevated acetylcholinesterase activity. In addition, histopathological evaluation also supported neuroprotective effects of diosgenin in hippocampus of rat brain when assessed using hematoxylin–eosin and Cresyl Violet staining. Thus, the aforementioned effects suggested protective action of diosgenin against Aβ (1–42) induced neuronal damage and thereby can serve as a potential therapeutic candidate for AD.





Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- 1,25D3–MARRS:
-
1,25D3 -membrane-associated, rapid response steroid-binding protein
- ACh:
-
Acetylcholine
- AChE:
-
Acetylcholinesterase
- aCSF:
-
Artificial cerebrospinal fluid
- AD:
-
Alzheimer’s disease
- Akt:
-
Protein kinase B
- ANOVA:
-
Analysis of variance
- AP:
-
Anterior–posterior
- Aβ:
-
Amyloid beta
- BFCN:
-
Basal forebrain cholinergic neurons
- CAT:
-
Catalase
- CMC:
-
Carboxymethyl cellulose
- CV:
-
Cresyl violet
- DTNB:
-
5,5-dithio-bis-(2-nitrobenzoic acid)
- DV:
-
Dorsal-ventral
- EDTA:
-
Ethylenediamine tetraacetic acid
- ELISA:
-
Enzyme-linked Immune Sorbent Assay
- ERK:
-
Extracellular signal-regulated kinase
- GSR:
-
Glutathione reductase
- H&E:
-
Hematoxylin and eosin
- H2O2 :
-
Hydrogen peroxide
- i.c.v:
-
Intracerebroventricular
- IAEC:
-
Institutional Animal Ethics Committee
- IL-6:
-
Interleukin 6
- JNK:
-
c-Jun N-terminal kinase
- MAPK:
-
Phosphorylated-p38 mitogen activated protein kinase
- ML:
-
Medial-lateral
- NADH:
-
Nicotinamide adenine dinucleotide
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate
- NF-κB:
-
Nuclear factor kappa B
- NGF:
-
Nerve growth factor
- p75NTR:
-
p75 pan neurotrophin receptor
- PI3K:
-
Phosphatidylinositol 3-kinase
- RAM:
-
Radial arm maze
- ROS:
-
Reactive oxygen species
- SD:
-
Standard deviation
- SOD:
-
Superoxide dismutase
- TLR:
-
Toll-like receptor
- TNF-α:
-
Tumour necrosis factor
- TrkA:
-
Tyrosine kinase A receptor
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. https://doi.org/10.1016/s0076-6879(84)05016-3
Alzoubi KH, Khabour OF, Salah HA, Hasan Z (2013) Vitamin E prevents high-fat high-carbohydrates diet-induced memory impairment: the role of oxidative stress. Physiol Behav 119:72–78. https://doi.org/10.1016/j.physbeh.2013.06.011
Andreeva TV, Lukiw WJ, Rogaev EI (2017) Biological Basis for Amyloidogenesis in Alzheimer’s Disease. Biochemistry (Mosc) 82:122–139. https://doi.org/10.1134/S0006297917020043
Baker-Nigh A, Vahedi S, Davis EG, Weintraub S, Bigio EH, Klein WL, Geula C (2015) Neuronal amyloid-β accumulation within cholinergic basal forebrain in ageing and Alzheimer's disease. Brain 138(Pt 6): 1722–1737. https://doi.org/10.1093/brain/awv024
Barkur RR, Bairy LK (2015) Evaluation of passive avoidance learning and spatial memory in rats exposed to low levels of lead during specific periods of early brain development. Int J Occup Med Environ Health 28:533–544. https://doi.org/10.13075/ijomeh.1896.00283
Baxter MG, Chiba AA (1999) Cognitive functions of the basal forebrain. Curr Opin Neurobiol 9:178–183. https://doi.org/10.1016/s0959-4388(99)80024-5
Bayrakdar TE, Uyanikgil Y, Kanit L, Koylu E, Yalcin A (2014) Nicotinamide treatment reduces the levels of oxidative stress, apoptosis, and PARP-1 activity in Aβ (1–42)-induced rat model of Alzheimer’s disease. Free Radic Res 48:146–158. https://doi.org/10.3109/10715762.2013.857018
Binesh A, Devaraj SN, Halagowder D (2018) Atherogenic diet induced lipid accumulation induced NFκB level in heart, liver and brain of Wistar rat and diosgenin as an anti-inflammatory agent. Life Sci 196:28–37. https://doi.org/10.1016/j.lfs.2018.01.012
Boyd-Kimball D, Sultana R, Mohmmad-Abdul H, Butterfield DA (2004) Rodent A beta (1–42) exhibits oxidative stress properties similar to those of human A beta(1–42): Implications for proposed mechanisms of toxicity. J Alzheimers Dis 6:515–525. https://doi.org/10.3233/jad-2004-6509
Burg VK, Grimm HS, Rothhaar TL, Grösgen S, Hundsdörfer B, Haupenthal VJ, Zimmer VC, Mett J, Weingärtner O, Laufs U, Broersen LM, Tanila H, Vanmierlo T, Lütjohann D, Hartmann T, Grimm MO (2013) Plant sterols the better cholesterol in Alzheimer’s disease? A mechanistical study. J Neurosci 33:16072–16087. https://doi.org/10.1523/JNEUROSCI.1506-13.2013
Butterfield DA, Swomley AM, Sultana R (2013) Amyloid β-peptide (1–42)-induced oxidative stress in Alzheimer disease: importance in disease pathogenesis and progression. Antioxid Redox Signal 19:823–835. https://doi.org/10.1089/ars.2012.5027
Cai B, Seong KJ, Bae SW, Kook MS, Chun C, Lee JH, Choi WS, Jung JY, Kim WJ (2019) Water-soluble Arginyl-Diosgenin analog attenuates hippocampal neurogenesis impairment through blocking microglial activation underlying NF-κB and JNK MAPK signaling in adult mice challenged by LPS. Mol Neurobiol 56:6218–6238. https://doi.org/10.1007/s12035-019-1496-3
Calsolaro V, Edison P (2016) Neuroinflammation in Alzheimer’s disease: Current evidence and future directions. Alzheimers Dement 12:719–732. https://doi.org/10.1016/j.jalz.2016.02.010
Carlberg I, Mannervik EB (1975) Glutathione level in rat brain. J Biol Chem 250:4475–4480
Chen Y, Tang YM, Yu SL, Han YW, Kou JP, Liu BL, Yu BY (2015) Advances in the pharmacological activities and mechanisms of diosgenin. Chin J Nat Med 13:578–587. https://doi.org/10.1016/S1875-5364(15)30053-4
Chiu CS, Chiu YJ, Wu LY, Lu TC, Huang TH, Hsieh MT, Lu CY, Peng WH (2011) Diosgenin ameliorates cognition deficit and attenuates oxidative damage in senescent mice induced by D-galactose. Am J Chin Med 39:551–563. https://doi.org/10.1142/S0192415X11009020
Choi KW, Park H, Hye DH, Kim TW, Park YM, Kim BO, Sohn EH, Moon EY, Um SH, Rhee DK, Pyo S (2010) Inhibition of TNF- α -induced adhesion molecule expression by diosgenin in mouse vascular smooth muscle cells via downregulation of the MAPK, Akt and NF- κ B signaling pathways. Vascul Pharmacol 53:273–280. https://doi.org/10.1016/j.vph.2010.09.007
Ciocchi S, Passecker J, Malagon-Vina H, Mikus N, Klausberger T (2015) Brain computation. Selective information routing by ventral hippocampal CA1 projection neurons. Science 348:560–563. https://doi.org/10.1126/science.aaa3245
Counts SE, Mufson EJ (2005) The role of nerve growth factor receptors in cholinergic basal forebrain degeneration in prodromal Alzheimer disease. J Neuropathol Exp Neurol 64:263–272. https://doi.org/10.1093/jnen/64.4.263
Ellman GL, Courtney KD, Andres V Jr, Feather-Stone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95. https://doi.org/10.1080/10715760802277388
Essawy AE, Abdou HM, Ibrahim HM, Bouthahab NM (2019) Soybean isoflavone ameliorates cognitive impairment, neuroinflammation, and amyloid β accumulation in a rat model of Alzheimer’s disease. Environ Sci Pollut Res Int 26:26060–26070. https://doi.org/10.1007/s11356-019-05862-z
Fahnestock M, Shekari A (2019) ProNGF and Neurodegeneration in Alzheimer’s Disease. Front Neurosci 13:129. https://doi.org/10.3389/fnins.2019.00129
Feng Y, Wang X (2012) Antioxidant therapies for Alzheimer’s disease. Oxid Med Cell Longev 3:472932. https://doi.org/10.1155/2012/472932
Fu AL, Dong ZH, Sun MJ (2006) Protective effect of N-acetyl-L-cysteine on amyloid beta-peptide-induced learning and memory deficits in mice. Brain Res 1109:201–206. https://doi.org/10.1016/j.brainres.2006.06.042
Hartley T, Lever C, Burgess N, O’Keefe J (2013) Space in the brain: how the hippocampal formation supports spatial cognition. Philos Trans R Soc Lond B Biol Sci 369:20120510. https://doi.org/10.1098/rstb.2012.0510
Hirai S, Uemura T, Mizoguchi N, Lee JY, Taketani K, Nakano Y, Hoshino S, Tsuge N, Narukami T, Yu R, Takahashi N, Kawada T (2010) Diosgenin attenuates inflammatory changes in the interaction between adipocytes and macrophages. Mol Nutr Food Res 54:797–804. https://doi.org/10.1002/mnfr.200900208
Hu W, Gray NW, Brimijoin S (2003) Amyloid-beta increases acetylcholinesterase expression in neuroblastoma cells by reducing enzyme degradation. J Neurochem 86:470–478. https://doi.org/10.1046/j.1471-4159.2003.01855.x
Hua S, Li Y, Su L, Liu X (2016) Diosgenin ameliorates gestational diabetes through inhibition of sterol regulatory element-binding protein-1. Biomed Pharmacother 84:1460–1465. https://doi.org/10.1016/j.biopha.2016.10.049
Iulita MF, Cuello AC (2014) Nerve growth factor metabolic dysfunction in Alzheimer’s disease and Down syndrome. Trends Pharmacol Sci 35:338–348. https://doi.org/10.1016/j.tips.2014.04.010
Izquierdo I, Furini CR, Myskiw JC (2016) Fear Memory. Physiol Rev 96:695–750. https://doi.org/10.1152/physrev.00018.2015
Jagadeesan J, Nandakumar N, Rengarajan T, Balasubramanian MP (2012) Diosgenin, a steroidal saponin, exhibits anticancer activity by attenuating lipid peroxidation via enhancing antioxidant defense system during NMU-induced breast carcinoma. J Environ Pathol Toxicol Oncol 31:121–129. https://doi.org/10.1615/jenvironpatholtoxicoloncol.v31.i2.40
Kakkar P, Das B, Viswanathan PN (1984) A modified spectrophotometric assay of superoxide dismutase. Indian J Biochem Biophys 21:130–132
Kalailingam P, Kannaian B, Tamilmani E, Kaliaperumal R (2014) Efficacy of natural diosgenin on cardiovascular risk, insulin secretion, and beta cells in streptozotocin (STZ)-induced diabetic rats. Phytomedicine 21:1154–1161. https://doi.org/10.1016/j.phymed.2014.04.005
Kaplan DR, Miller FD (2000) Neurotrophin signal transduction in the nervous system. Curr Opin Neurobiol 10:381–391. https://doi.org/10.1016/s0959-4388(00)00092-1
Khalil KA, Abass KS (2017) The measurements of the cholinesterase activity of brain and plasma in rabbits by using modified michel and Ellman assays. Insights Enzyme Res 1:2
Kim JE, Go J, Koh EK, Song SH, Sung JE, Lee HA, Kim DS, Son HJ, Lee HS, Lee CY, Hong JT, Hwang DY (2016) Diosgenin effectively suppresses skin inflammation induced by phthalic anhydride in IL-4/Luc/CNS-1 transgenic mice. Biosci Biotechnol Biochem 80:891–901. https://doi.org/10.1080/09168451.2015.1135040
Koh EK, Yun WB, Kim JE, Song SH, Sung JE, Lee HA, Seo EJ, Jee SW, Bae CJ, Hwang DY (2016) Beneficial effect of diosgenin as a stimulator of NGF on the brain with neuronal damage induced by Aβ-42 accumulation and neurotoxicant injection. Lab Anim Res 32:105–115. https://doi.org/10.5625/lar.2016.32.2.105
Lad SP, Neet KE, Mufson EJ (2003) Nerve growth factor: structure, function and therapeutic implications for Alzheimer’s disease. Curr Drug Targets CNS Neurol Disord 2:315–334. https://doi.org/10.2174/1568007033482724
Lecanu L, Rammouz G, McCourty A, Sidahmed EK, Greeson J, Papadopoulos V (2010) Caprospinol reduces amyloid deposits and improves cognitive function in a rat model of Alzheimer’s disease. Neuroscience 165:427–435. https://doi.org/10.1016/j.neuroscience.2009.10.033
Lecanu L, Tillement L, Rammouz G, Tillement JP, Greeson J, Papadopoulos V (2009) Caprospinol: moving from a neuroactive steroid to a neurotropic drug. Expert Opin Investig Drugs 18:265–276. https://doi.org/10.1517/13543780902762827
Lecanu L, Yao W, Teper GL, Yao ZX, Greeson J, Papadopoulos V (2004) Identification of naturally occurring spirostenols preventing beta-amyloid-induced neurotoxicity. Steroids 69:1–16. https://doi.org/10.1016/j.steroids.2003.09.007
Ledezma C, Coria-Lucero C, Delsouc MB, Casais M, Della Vedova C, Ramirez D, Devia CM, Delgado SM, Navigatore-Fonzo L, Anzulovich AC (2021) Effect of an intracerebroventricular injection of aggregated Beta-amyloid (1–42) on daily rhythms of oxidative stress parameters in the prefrontal cortex. Neuroscience 458:99–107. https://doi.org/10.1016/j.neuroscience.2020.08.016
Lee BK, Kim CJ, Shin MS, Cho YS (2018) Diosgenin improves functional recovery from sciatic crushed nerve injury in rats. J Exerc Rehabil 14:566–572. https://doi.org/10.12965/jer.1836340.170
Lennon SV, Martin SJ, Cotter TG (1991) Dose-dependent induction of apoptosis in human tumour cell lines by widely diverging stimuli. Cell Prolif 24:203–214. https://doi.org/10.1111/j.1365-2184.1991.tb01150.x
Li B, Xu P, Wu S, Jiang Z, Huang Z, Li Q, Chen D (2018) Diosgenin attenuates Lipopolysaccharide-induced Parkinson’s Disease by inhibiting the TLR/NF-κB pathway. J Alzheimers Dis 64:943–955. https://doi.org/10.3233/JAD-180330
Lian W, Fang J, Xu L, Zhou W, Kang D, Xiong W, Jia H, Liu AL, Du GH (2017) DL0410 Ameliorates Memory and Cognitive Impairments Induced by Scopolamine via Increasing Cholinergic Neurotransmission in Mice. Molecules 22:410. https://doi.org/10.3390/molecules22030410
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the folin phenol reagent. J Bio Chem 193:265–275
Mahdy K, Shaker O, Wafay H, Nassar Y, Hassan H, Hussein A (2012) Effect of some medicinal plant extracts on the oxidative stress status in Alzheimer’ s disease induced in rats. Eur Rev Med Pharmacol Sci 16:31–42
Masilamoni JG, Jesudason EP, Dhandayuthapani S, Ashok BS, Vignesh S, Jebaraj WC, Paul SF, Jayakumar R (2008) The neuroprotective role of melatonin against amyloid beta peptide injected mice. Free Radic Res 42:661–673. https://doi.org/10.1080/10715760802277388
Mitra S, Behbahani H, Eriksdotter M (2019) Innovative therapy for Alzheimer’s disease-with focus on biodelivery of NGF. Front Neurosci 13:38. https://doi.org/10.3389/fnins.2019.00038
Mufson EJ, Conner JM, Kordower JH (1995) Nerve growth factor in Alzheimer’s disease: defective retrograde transport to nucleus basalis. Neuroreport 6:1063–1066. https://doi.org/10.1097/00001756-199505090-00028
Nakamura S, Murayama N, Noshita T, Annoura H, Ohno T (2001) Progressive brain dysfunction following intracerebroventricular infusion of beta (1–42)-amyloid peptide. Brain Res 912:128–136. https://doi.org/10.1016/s0006-8993(01)02704-4
National Research Council (US), Guide for the Care and Use of Laboratory Animals, 8th ed, National Academies Press, Washington (DC), 2011.
Niewiadomska G, Mietelska-Porowska A, Mazurkiewicz M (2011) The cholinergic system, nerve growth factor and the cytoskeleton. Behav Brain Res 221:515–526. https://doi.org/10.1016/j.bbr.2010.02.024
Okuyama T, Kitamura T, Roy DS, Itohara S, Tonegawa S (2016) Ventral CA1 neurons store social memory. Science 353:1536–1541. https://doi.org/10.1126/science.aaf7003
Papadopoulos V, Lecanu L (2012) Caprospinol: discovery of a steroid drug candidate to treat Alzheimer’s disease based on 22R-hydroxycholesterol structure and properties. J Neuroendocrinol 24:93–101. https://doi.org/10.1111/j.1365-2826.2011.02167.x
Pi R, Mao X, Chao X, Cheng Z, Liu M, Duan X, Ye M, Chen X, Mei Z, Liu P, Li W, Han Y (2012) Tacrine-6-Ferulic Acid, a novel multifunctional dimer, inhibits amyloid- β-mediated Alzheimer ’ s disease- associated pathogenesis in vitro and in vivo. PLoS ONE 7:e31921. https://doi.org/10.1371/journal.pone.0031921
Postu PA, Noumedem JAK, Cioanca O, Hancianu M, Mihasan M, Ciorpac M, Gorgan DL, Petre BA, Hritcu L (2018) Lactuca capensis reverses memory deficits in Aβ1-42-induced an animal model of Alzheimer’s disease. J Cell Mol Med. 22:111–122. https://doi.org/10.1111/jcmm.13299
Rahman SO, Panda BP, Parvez S, Kaundal M, Hussain S, Akhtar M, Najmi AK (2019) Neuroprotective role of astaxanthin in hippocampal insulin resistance induced by Aβ peptides in animal model of Alzheimer’ s disease. Biomed Pharmacother 110:47–58. https://doi.org/10.1016/j.biopha.2018.11.043
Salinas M, Diaz R, Abraham NG, Ruiz de Galarreta CM, Cuadrado A (2003) Nerve growth factor protects against 6-hydroxydopamine-induced oxidative stress by increasing expression of heme oxygenase-1 in a phosphatidylinositol 3-kinase-dependent manner. J Biol Chem 278:13898–13904. https://doi.org/10.1074/jbc.M209164200
Sehgal N, Gupta A, Valli RK, Joshi SD, Mills JT, Hamel E, Khanna P, Jain SC, Thakur SS, Ravindranath V (2012) Withania somnifera reverses Alzheimer ’ s disease pathology by enhancing low-density lipoprotein receptor-related protein in liver. Proc Natl Acad Sci U S A 109:3510–3515. https://doi.org/10.1073/pnas.1112209109
Seiler M, Schwab ME (1984) Specific retrograde transport of nerve growth factor (NGF) from neocortex to nucleus basalis in the rat. Brain Res 300:33–39. https://doi.org/10.1016/0006-8993(84)91338-6
Serrano F, Klann E (2004) Reactive oxygen species and synaptic plasticity in the aging hippocampus. Ageing Res Rev 3:431–443. https://doi.org/10.1016/j.arr.2004.05.002
Sethi G, Shanmugam MK, Warrier S, Merarchi M, Arfuso F, Kumar AP, Bishayee A (2018) Pro-Apoptotic and Anti-Cancer Properties of Diosgenin: A Comprehensive and Critical Review. Nutrients 10:645. https://doi.org/10.3390/nu10050645
Shal B, Ding W, Ali H, Kim YS, Khan S (2018) Anti-neuroinflammatory Potential of Natural Products in Attenuation of Alzheimer’s Disease. Front Pharmacol 9:548. https://doi.org/10.3389/fphar.2018.00548
Son IS, Kim JH, Sohn HY, Son KH, Kim JS, Kwon CS (2007) Antioxidative and hypolipidemic effects of Diosgenin, a steroidal saponin of Yam (Dioscorea spp.), on high-cholesterol fed rats. Biosci Biotechnol Biochem 71:3063–3071. https://doi.org/10.1271/bbb.70472
Souza LC, Jesse CR, Antunes MS, Ruff JR, de Oliveira Espinosa D, Gomes NS, Donato F, Giacomeli R, Boeira SP (2016) Indoleamine-2,3-dioxygenase mediates neurobehavioral alterations induced by an intracerebroventricular injection of amyloid-β1-42 peptide in mice. Brain Behav Immun 56:363–377. https://doi.org/10.1016/j.bbi.2016.03.002
Spires TL, Orne JD, SantaCruz K, Pitstick R, Carlson GA, Ashe KH, Hyman BT (2006) Region-specific dissociation of neuronal loss and neurofibrillary pathology in a mouse model of tauopathy. Am J Pathol 168:1598–607. https://doi.org/10.2353/ajpath.2006.050840
Tohda C, Urano T, Umezaki M, Nemere I, Kuboyama T (2012) Diosgenin is an exogenous activator of 1,25D 3-MARRS/Pdia3/ERp57 and improves Alzheimer’s disease pathologies in 5XFAD mice. Sci Rep 2:535. https://doi.org/10.1038/srep00535
Tönnies E, Trushina E (2017) Oxidative stress, synaptic dysfunction, and Alzheimer’s disease. J Alzheimers Dis 57:1105–1121. https://doi.org/10.3233/JAD-161088
Turnbull MT, Boskovic Z, Coulson EJ (2018) Acute down-regulation of BDNF signaling does not replicate exacerbated amyloid-β levels and cognitive impairment induced by cholinergic basal forebrain lesion. Front Mol Neurosci 11:51. https://doi.org/10.3389/fnmol.2018.00051
Tuzcu M, Baydas G (2006) Effect of melatonin and vitamin E on diabetes-induced learning and memory impairment in rats. Eur J Pharmacol 537:106–110. https://doi.org/10.1016/j.ejphar.2006.03.024
Wang L, Du J, Zhao F, Chen Z, Chang J, Qin F, Wang Z, Wang F, Chen X, Chen N (2018) Trillium tschonoskii maxim saponin mitigates D-galactose-induced brain aging of rats through rescuing dysfunctional autophagy mediated by Rheb-mTOR signal pathway. Biomed Pharmacother 98:516–522. https://doi.org/10.1016/j.biopha.2017.12.046
Wang Q, Wang C, Shu Z, Chan K, Huang S, Li Y, Xiao Y, Wu L, Kuang H, Sun X (2014) Valeriana amurensis improves Amyloid-beta 1–42 induced cognitive deficit by enhancing cerebral cholinergic function and protecting the brain neurons from apoptosis in mice. J Ethnopharmacol 153:318–325. https://doi.org/10.1016/j.jep.2013.11.017
Wang R, Yan H, Tang XC (2006) Progress in studies of huperzine A, a natural cholinesterase inhibitor from Chinese herbal medicine. Acta Pharmacol Sin 27:1–26. https://doi.org/10.1111/j.1745-7254.2006.00255.x
Wang S, Wang F, Yang H, Li R, Guo H, Hu L (2017) Diosgenin glucoside provides neuroprotection by regulating microglial M1 polarization. Int Immunopharmacol 50:22–29. https://doi.org/10.1016/j.intimp.2017.06.008
West MJ, Coleman PD, Flood DG, Troncoso JC (1994) Differences in the pattern of hippocampal neuronal loss in normal ageing and Alzheimer’s disease. Lancet 344:769–72. https://doi.org/10.1016/s0140-6736(94)92338-8
Xu CJ, Wang JL, Jin WL (2016) The Emerging Therapeutic Role of NGF in Alzheimer’s Disease. Neurochem Res 41:1211–1218. https://doi.org/10.1007/s11064-016-1829-9
Yamada K, Tanaka T, Mamiya T, Shiotani T, Kameyama T, Nabeshima T (1999) Improvement by nefiracetam of β-amyloid-(1–42)-induced learning and memory impairments in rats. Br J Pharmacol 126:235–244. https://doi.org/10.1038/sj.bjp.0702309
Yang WS, Moon SY, Lee MJ, Lee EK, Park SK (2017) Diosgenin, an Activator of 1,25D3-MARRS Receptor/ERp57, Attenuates the Effects of TNF-α by Causing ADAM10-Dependent Ectodomain Shedding of TNF Receptor 1. Cell Physiol Biochem 43:2434–2445. https://doi.org/10.1159/000484396
Zhang B, Chen Y, Yang B, Jiang F, Zhao J, Ouyang Z (2017) Neuroprotective effects of diosgenin in rats with experimental spinal cord injury via promotion of autophagy. Int J Clin Exp Med 10:11655–11663
Zhang YW, Chen Y, Liu Y, Zhao Y, Liao FF, Xu H (2013) APP regulates NGF receptor trafficking and NGF-mediated neuronal differentiation and survival. PLoS One 8:e80571. https://doi.org/10.1371/journal.pone.0080571
Zhao WX, Wang PF, Song HG, Sun N (2017) Diosgenin attenuates neuropathic pain in a rat model of chronic constriction injury. Mol Med Rep 16:1559–1564. https://doi.org/10.3892/mmr.2017.6723
Acknowledgements
We express our gratitude to the Department of Pharmacognosy and Phytochemistry, Department of Pharmacology and Department of Pharmaceutical Biotechnology, JSS College of Pharmacy, Ooty for providing various chemicals and facilities used during the course of the study. This project was financially supported by the Lady Tata Memorial Trust, Mumbai, India with senior research fellowship (SRF).
Author information
Authors and Affiliations
Contributions
SS performed the experiments and was a major contributor in writing the manuscript. JA analyzed and interpreted the raw data regarding the Alzheimer’s disease. SP conceptualized, reviewed and edited the manuscript. SPD reviewed and edited the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
All experiments were conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals (National Research Council (US)) and was approved by the Institutional Animal Ethics Committee (IAEC) (JSSCP/IAEC/PhD/Pharmacy Practice/01/2018–19).
Competing Interest
All authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Som, S., Antony, J., Dhanabal, S. et al. Neuroprotective role of Diosgenin, a NGF stimulator, against Aβ (1–42) induced neurotoxicity in animal model of Alzheimer’s disease. Metab Brain Dis 37, 359–372 (2022). https://doi.org/10.1007/s11011-021-00880-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-021-00880-8