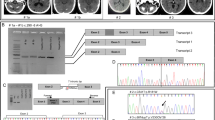
Abstract
Cerebral calcification may be caused by several potentially treatable conditions, however, in most cases it does not receive special attention in clinical practice. From the point of view of etiology, the diseases associated with cerebral calcification can be divided into two main groups: idiopathic (mostly incurable) and secondary (potentially treatable). The first group includes mainly the hereditary diseases identified before 2021 (primary familial brain calcification subtypes, previously known as Fahr’s disease or Fahr’s syndrome). In contrast, the second group includes diseases with cerebral calcification that develop generally as a consequence of metabolic/endocrine/autoimmune abnormalities. The aim of our research was to present hereditary and non-hereditary etiologies associated with extensive brain calcification. We compare the detailed clinical, radiological and laboratory results of 6 patients with prominent cerebral calcification identified in our clinic in the last 3 years (idiopathic and secondary etiologies as well). Our research draws attention to the complexity of the etiologies in the context of cerebral calcification. We recommend, beside NGS-based sequence analyses, the application of array comparative genomic hybridization as well, to identify potential genetic etiologies associated with brain calcification.

Similar content being viewed by others
Data availability
The datasets used and/or analyzed in the current study are available in this paper.
References
Andres RH, Schroth G, Remonda L (2009) Extensive cerebral calcification in a patient with systemic lupus erythematosus. BMJ case reports 2009:bcr2007125393. https://doi.org/10.1136/bcr.2007.125393
Bastepe M, Fröhlich LF, Hendy GN, Indridason OS, Josse RG, Koshiyama H, Körkkö J, Nakamoto JM, Rosenbloom AL, Slyper AH, Sugimoto T, Tsatsoulis A, Crawford JD, Jüppner H (2003) Autosomal dominant pseudohypoparathyroidism type Ib is associated with a heterozygous microdeletion that likely disrupts a putative imprinting control element of GNAS. J Clin Invest 112:1255–1263. https://doi.org/10.1172/JCI19159
Batla A, Tai XY, Schottlaender L, Erro R, Balint B, Bhatia KP (2017) Deconstructing Fahr's disease/syndrome of brain calcification in the era of new genes. Parkinsonism Relat Disord 37:1–10. https://doi.org/10.1016/j.parkreldis.2016.12.024
Bauer M, Rahat D, Zisman E, Tabach Y, Lossos A, Meiner V, Arkadir D (2019) MYORG mutations: a major cause of recessive primary familial brain calcification. Curr Neurol Neurosci Rep 19:70. https://doi.org/10.1007/s11910-019-0986-z
Bonazza S, Morgia CL, Martinelli P, Capellari S (2011) Strio-pallido-dentate calcinosis: a diagnostic approach in adult patients. Neurol Sci 32:537–545. https://doi.org/10.1007/s10072-011-0514-7
Cen Z, Chen Y, Chen S, Wang H, Yang D, Zhang H, Wu H, Wang L, Tang S, Ye J, Shen J, Wang H, Fu F, Chen X, Xie F, Liu P, Xu X, Cao J, Cai P, Pan Q, Li J, Yang W, Shan PF, Li Y, Liu JY, Zhang B, Luo W (2020) Biallelic loss-of-function mutations in JAM2 cause primary familial brain calcification. Brain 143:491–502. https://doi.org/10.1093/brain/awz392
Chelban V, Carecchio M, Rea G, Bowirrat A, Kirmani S, Magistrelli L, Efthymiou S, Schottlaender L, Vandrovcova J, Salpietro V, Salsano E, Pareyson D, Chiapparini L, Jan F, Ibrahim S, Khan F, Qarnain Z, Groppa S, Bajaj N, Balint B, Bhatia KP, Lees A, Morrison PJ, Wood NW, Garavaglia B, Houlden H (2020) MYORG-related disease is associated with central pontine calcifications and atypical parkinsonism. Neurol Genet 6:e399. https://doi.org/10.1212/NXG.0000000000000399
Chen WJ, Yao XP, Zhang QJ, Ni W, He J, Li HF, Liu XY, Zhao GX, Murong SX, Wang N, Wu ZY (2013) Novel SLC20A2 mutations identified in southern Chinese patients with idiopathic basal ganglia calcification. Gene 529:159–162. https://doi.org/10.1016/j.gene.2013.07.071
Cusano NE, Maalouf NM, Wang PY, Zhang C, Cremers SC, Haney EM, Bauer DC, Orwoll ES, Bilezikian JP (2013) Normocalcemic hyperparathyroidism and hypoparathyroidism in two community-based nonreferral populations. J Clin Endocrinol Metab 98:2734–2741. https://doi.org/10.1210/jc.2013-1300
De Oliveira MF, Silva EBE, de Oliveira JRM (2013) Prevalence of brain calcifications in a Brazilian cohort: a retrospective study in radiology services. Dement Neuropsychol 7:210–215. https://doi.org/10.1590/S1980-57642013DN70200012
Donzuso G, Mostile G, Nicoletti A, Zappia M (2019) Basal ganglia calcifications (Fahr's syndrome): related conditions and clinical features. Neurol Sci 40:2251–2263. https://doi.org/10.1007/s10072-019-03998-x
Elli FM, Sanctis LD, Peverelli E, Bordogna P, Pivetta B, Miolo G, Beck-Peccoz P, Spada A, Mantovani G (2014) Autosomal dominant pseudohypoparathyroidism type Ib: a novel inherited deletion ablating STX16 causes loss of imprinting at the a/B DMR. J Clin Endocrinol Metab 99:E724–E728. https://doi.org/10.1210/jc.2013-3704
Keller A, Westenberger A, Sobrido MJ, García-Murias M, Domingo A, Sears RL, Lemos RR, Ordoñez-Ugalde A, Nicolas G, da Cunha JEG, Rushing EJ, Hugelshofer M, Wurnig MC, Kaech A, Reimann R, Lohmann K, Dobričić V, Carracedo A, Petrović I, Miyasaki JM, Abakumova I, Mäe MA, Raschperger E, Zatz M, Zschiedrich K, Klepper J, Spiteri E, Prieto JM, Navas I, Preuss M, Dering C, Janković M, Paucar M, Svenningsson P, Saliminejad K, Khorshid HRK, Novaković I, Aguzzi A, Boss A, Le Ber I, Defer G, Hannequin D, Kostić VS, Campion D, Geschwind DH, Coppola G, Betsholtz C, Klein C, Oliveira JRM (2013) Mutations in the gene encoding PDGF-B cause brain calcifications in humans and mice. Nat Genet 45:1077–1082. https://doi.org/10.1038/ng.2723
Kıroğlu Y, Callı C, Karabulut N, Oncel C (2010) Intracranial calcifications on CT. Diagn Interv Radiol 16:263–269. https://doi.org/10.4261/1305-3825.DIR.2626-09.1
Kostić VS, Petrović IN (2017) Brain calcification and movement disorders. Curr Neurol Neurosci Rep 17:2. https://doi.org/10.1007/s11910-017-0710-9
Laspa E, Bastepe M, Jüppner H, Tsatsoulis A (2004) Phenotypic and molecular genetic aspects of pseudohypoparathyroidism type Ib in a Greek kindred: evidence for enhanced uric acid excretion due to parathyroid hormone resistance. J Clin Endocrinol Metab 89:5942–5947. https://doi.org/10.1210/jc.2004-0249
Legati A, Giovannini D, Nicolas G, López-Sánchez U, Quintáns B, Oliveira JRM, Sears RL, Ramos EM, Spiteri E, Sobrido MJ, Carracedo Á, Castro-Fernández C, Cubizolle S, Fogel BL, Goizet C, Jen JC, Kirdlarp S, Lang AE, Miedzybrodzka Z, Mitarnun W, Paucar M, Paulson H, Pariente J, Richard AC, Salins NS, Simpson SA, Striano P, Svenningsson P, Tison F, Unni VK, Vanakker O, Wessels MW, Wetchaphanphesat S, Yang M, Boller F, Campion D, Hannequin D, Sitbon M, Geschwind DH, Battini JL, Coppola G (2015) Mutations in XPR1 cause primary familial brain calcification associated with altered phosphate export. Nat Genet 47:579–581. https://doi.org/10.1038/ng.3289
Linglart A, Gensure RC, Olney RC, Jüppner H, Bastepe M (2005) A novel STX16 deletion in autosomal dominant pseudohypoparathyroidism type Ib redefines the boundaries of a cis-acting imprinting control element of GNAS. Am J Hum Genet 76:804–814. https://doi.org/10.1086/429932
Mantovani G, Bondioni S, Linglart A, Maghnie M, Cisternino M, Corbetta S, Lania AG, Beck-Peccoz P, Spada A (2007) Genetic analysis and evaluation of resistance to thyrotropin and growth hormone-releasing hormone in pseudohypoparathyroidism type Ib. J Clin Endocrinol Metab 92:3738–3742. https://doi.org/10.1210/jc.2007-0869
Manyam BV (2005) What is and what is not ‘Fahr’s disease’. Parkinsonism Relat Disord 11:73–80. https://doi.org/10.1016/j.parkreldis.2004.12.001
Miccoli P, Minuto MN, Miccoli M (2012) Incidence of morbidity following thyroid surgery. Thyroid surgery: preventing and managing complications. John Wiley & Sons, Ltd 2012:6–7
Nicolas G, Pottier C, Charbonnier C, Guyant-Maréchal L, Le Ber I, Pariente J, Labauge P, Ayrignac X, Defebvre L, Maltête D, Martinaud O, Lefaucheur R, Guillin O, Wallon D, Chaumette B, Rondepierre P, Derache N, Fromager G, Schaeffer S, Krystkowiak P, Verny C, Jurici S, Sauvée M, Vérin M, Lebouvier T, Rouaud O, Thauvin-Robinet C, Rousseau S, Rovelet-Lecrux A, Frebourg T, Campion D, Hannequin D, French IBGC Study Group (2013a) Phenotypic spectrum of probable and genetically-confirmed idiopathic basal ganglia calcification. Brain 136:3395–3407. https://doi.org/10.1093/brain/awt255
Nicolas G, Pottier C, Maltête D, Coutant S, Rovelet-Lecrux A, Legallic S, Rousseau S, Vaschalde Y, Guyant-Maréchal L, Augustin J, Martinaud O, Defebvre L, Krystkowiak P, Pariente J, Clanet M, Labauge P, Ayrignac X, Lefaucheur R, Le Ber I, Frébourg T, Hannequin D, Campion D (2013b) Mutation of the PDGFRB gene as a cause of idiopathic basal ganglia calcification. Neurology 80:181–187. https://doi.org/10.1212/WNL.0b013e31827ccf34
Petrarca M, Scipioni R, Di Giosia P, Giorgini P, Ferri C (2017) A case of brain calcifications in postsurgical hypoparathyroidism. Intern Emerg Med 12:113–115. https://doi.org/10.1007/s11739-016-1430-x
Ramos EM, Oliveira J, Sobrido MJ, Coppola G (2017) Primary familial brain calcification. In: Adam MP, Ardinger HH, Pagon RA, et al. (eds) GeneReviews® [internet]. Seattle (WA): University of Washington, Seattle, 1993–2021. Available from: https://www.ncbi.nlm.nih.gov/books/NBK1421/
Saade C, Najem E, Asmar K, Salman R, Achkar BE, Naffaa L (2019) Intracranial calcifications on CT: an updated review. J Radiol Case Rep 13:1–18. https://doi.org/10.3941/jrcr.v13i8.3633
Saleem S, Aslam HM, Anwar M, Anwar S, Saleem M, Saleem A, Rehmani MAK (2013) Fahr's syndrome: literature review of current evidence. Orphanet J Rare Dis 8:156. https://doi.org/10.1186/1750-1172-8-156
Savino E, Soavi C, Capatti E, Borrelli M, Vigna GB, Passaro A, Zuliani G (2016) Bilateral strio-pallido-dentate calcinosis (Fahr's disease): report of seven cases and revision of literature. BMC Neurol 16:165. https://doi.org/10.1186/s12883-016-0693-1
Taborda KNN, Wilches C, Manrique A (2017) A diagnostic algorithm for patients with intracranial calcifications. Rev Colomb Radiol 28:4732–4739
Tadic V, Westenberger A, Domingo A, Alvarez-Fischer D, Klein C, Kasten M (2015) Primary familial brain calcification with known gene mutations: a systematic review and challenges of phenotypic characterization. JAMA Neurol 72:460–467. https://doi.org/10.1001/jamaneurol.2014.3889
Wang C, Li Y, Shi L, Ren J, Patti M, Wang T, de Oliveira JRM, Sobrido MJ, Quintáns B, Baquero M, Cui X, Zhang XY, Wang L, Xu H, Wang J, Yao J, Dai X, Liu J, Zhang L, Ma H, Gao Y, Ma X, Feng S, Liu M, Wang QK, Forster IC, Zhang X, Liu JY (2012) Mutations in SLC20A2 link familial idiopathic basal ganglia calcification with phosphate homeostasis. Nat Genet 44:254–256. https://doi.org/10.1038/ng.1077
Westenberger A, Balck A, Klein C (2019) Primary familial brain calcifications: genetic and clinical update. Curr Opin Neurol 32:571–578. https://doi.org/10.1097/WCO.0000000000000712
Yamada M, Asano T, Okamoto K, Hayashi Y, Kanematsu M, Hoshi H, Akaiwa Y, Shimohata T, Nishizawa M, Inuzuka T, Hozumi I (2013) High frequency of calcification in basal ganglia on brain computed tomography images in Japanese older adults. Geriatr Gerontol Int 13:706–710. https://doi.org/10.1111/ggi.12004
Funding
GINOP-2.3.2–15-2 grant (TK and ZM) provided by the National Research, Development and Innovation Office (Hungary). Hungarian Brain Research Program [2017–1.2.1-NKP-2017-00002 NAP VI/4] (PK).
Author information
Authors and Affiliations
Contributions
AS and DZ examined the patients and wrote the manuscript. VLN and NSZ performed the neuropsychological examination of the patients. Genetic tests and consultations were performed by AU, ZM, EH, IB and TK. PK and CSB had important recommendations to the manuscript. LSZ, ML and BB provided essential assistance in patient involvement and investigation.
Corresponding author
Ethics declarations
Ethics approval
Written informed consent was obtained from all individual participants included in the study. Additional informed consent was obtained from all individual participants for whom identifying information is included in this article (Regional Human Biomedical Research Ethics Committee of the University of Szeged registration numbers are 150/2014. and 44/2016., respectively). All procedures performed in this study involving human participants were in accordance with the ethical standards of the Regional Human Biomedical Research Ethics Committee of the University of Szeged and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflicts of interest/competing interests
The authors declare that they have no competing/conflict of interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary video 1
Clinical phenotype of case 1 – mos 46,XY,der(11)t(X;11)(p21.1;q14.2)[152]/46,XY[48] (MP4 150,053 kb)
Supplementary video 2
Clinical phenotype of case 2 – SLC20A2 (MP4 76,142 kb)
Supplementary video 3
Clinical phenotype of case 3 – SLC20A2 (MP4 83,455 kb)
Supplementary video 4
Clinical phenotype of case 4 – STX16 (MP4 143,065 kb)
Supplementary video 5
Clinical phenotype of case 5 – Infiltration of the parathyroid gland (MP4 90,431 kb)
Supplementary video 6
Clinical phenotype of case 6 – Secondary hypoparathyroidism (thymectomy) (MP4 88,249 kb)
Supplementary file 1
The gene content of the genetic alteration mos 46,XY,der(11)t(X;11)(p21.1;q14.2)[152]/46,XY[48] found in Case 1 (PDF 488 kb)
Supplementary file 2
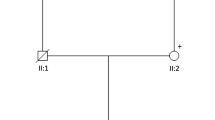
Pedigrees and chromatograms of Cases 1–4. (The generation are signed with Roman numbers. The deceased members are crossed. Individuals with symptoms are marked in black. Abbreviations: arrow – genetic test was performed; d. – age of death; E – genetic examination; p – proband; y – year; + or - – the result of the genetic test.) (PDF 1978 kb)
Rights and permissions
About this article
Cite this article
Salamon, A., Zádori, D., Ujfalusi, A. et al. Hereditary and non-hereditary etiologies associated with extensive brain calcification: case series. Metab Brain Dis 36, 2131–2139 (2021). https://doi.org/10.1007/s11011-021-00790-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-021-00790-9