Abstract
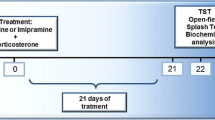
Although numerous studies have investigated the mechanisms underlying the fast and sustained antidepressant-like effects of ketamine, the contribution of the glucocorticoid receptor (GR) and dendritic branching remodeling to its responses remain to be fully established. This study investigated the ability of a single administration of ketamine to modulate the GR and dendritic branching remodeling and complexity in the hippocampus of mice subjected to chronic corticosterone (CORT) administration. CORT was administered for 21 days, followed by a single administration of ketamine (1 mg ∕kg, i.p.) or fluoxetine (10 mg ∕kg, p.o., conventional antidepressant) in mice. On 22nd, 24 h after the treatments, GR immunocontent in the hippocampus was analyzed by western blotting, while the dendritic arborization and dendrite length in the ventral and dorsal dentate gyrus (DG) of the hippocampus was analyzed by Sholl analysis. Chronic CORT administration downregulated hippocampal GR immunocontent, but this alteration was completely reversed by a single administration of ketamine, but not fluoxetine. Moreover, CORT administration significantly decreased dendritic branching in the dorsal and ventral DG areas and caused a mild decrease in dendrite length in both regions. Ketamine, but not fluoxetine, reversed CORT-induced dendritic branching loss in the ventral and dorsal DG areas, regions associated with mood regulation and cognitive functions, respectively. This study provides novel evidence that a single administration of ketamine, but not fluoxetine, rescued the impairments on GR and dendritic branching in the hippocampus of mice subjected to chronic CORT administration, effects that may be associated with its rapid antidepressant response.




Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Anacker C, Zunszain PA, Carvalho LA, Pariante CM (2011a) The glucocorticoid receptor: Pivot of depression and of antidepressant treatment? Psychoneuroendocrinology 36:415–425. https://doi.org/10.1016/j.psyneuen.2010.03.007
Anacker C, Zunszain PA, Cattaneo A et al (2011b) Antidepressants increase human hippocampal neurogenesis by activating the glucocorticoid receptor. Mol Psychiatry 16:738–750. https://doi.org/10.1038/mp.2011.26
Autry AE, Adachi M, Nosyreva E et al (2011) NMDA receptor blockade at rest triggers rapid behavioural antidepressant responses. Nature 475:91–96. https://doi.org/10.1038/nature10130
Bannerman DM, Rawlins JNP, McHugh SB et al (2004) Regional dissociations within the hippocampus - Memory and anxiety. Neurosci Biobehav Rev 28:273–283
Berman RM, Cappiello A, Anand A et al (2000) Antidepressant effects of ketamine in depressed patients. Biol Psychiatry 47:351–354. https://doi.org/10.1016/S0006-3223(99)00230-9
Bird CM, Burgess N (2008) The hippocampus and memory: insights from spatial processing. Nat Rev Neurosci 9:182–194. https://doi.org/10.1038/nrn2335
Camargo A, Dalmagro AP, Platt N et al (2020a) Cholecalciferol abolishes depressive-like behavior and hippocampal glucocorticoid receptor impairment induced by chronic corticosterone administration in mice. Pharmacol Biochem Behav 196:172971. https://doi.org/10.1016/j.pbb.2020.172971
Camargo A, Dalmagro AP, M. Rosa J, et al (2020b) Subthreshold doses of guanosine plus ketamine elicit antidepressant-like effect in a mouse model of depression induced by corticosterone: Role of GR/NF-κB/IDO-1 signaling. Neurochem Int 139:104797. https://doi.org/10.1016/j.neuint.2020.104797
Desmet SJ, De Bosscher K (2017) Glucocorticoid receptors: Finding the middle ground. J. Clin. Invest. 127:1136–1145. https://doi.org/10.1172/JCI88886
DiazGranados N, Ibrahim L, Brutsche N et al (2010) Rapid resolution of suicidal ideation after a single infusion of an NMDA Antagonist in patients with treatment-resistant major depressive disorder. J Clin Psychiatry 71:1605–1611. https://doi.org/10.4088/JCP.09m05327blu.Rapid
Fanselow MS, Dong HW (2010) Are the Dorsal and Ventral Hippocampus Functionally Distinct Structures? Neuron 65:7–19
Feldman S, Weidenfeld J (1999) Glucocorticoid receptor antagonists in the hippocampus modify the negative feedback following neural stimuli. Brain Res 821:33–37. https://doi.org/10.1016/S0006-8993(99)01054-9
Fraga DB, Costa AP, Olescowicz G et al (2020) Ascorbic acid presents rapid behavioral and hippocampal synaptic plasticity effects. Prog Neuropsychopharmacol Biol Psychiatry 96:109757. https://doi.org/10.1016/j.pnpbp.2019.109757
Freitas A, Heinrich I, Moura T et al (2020) Agmatine potentiates antidepressant and synaptic actions of ketamine: Effects on dendritic arbors and spines architecture and Akt/S6 kinase signaling. Exp Neurol 333:113398. https://doi.org/10.1016/j.expneurol.2020.113398
Gibb R, Kolb B (1998) A method for vibratome sectioning of Golgi-Cox stained whole rat brain. J Neurosci Methods 79:1–4. https://doi.org/10.1016/S0165-0270(97)00163-5
Jafari M, Seese RR, Babayan AH et al (2012) Glucocorticoid receptors are localized to dendritic spines and influence local actin signaling. Mol Neurobiol 46:304–315. https://doi.org/10.1007/s12035-012-8288-3
Jan YN, Jan LY (2010) Branching out: Mechanisms of dendritic arborization. Nat Rev Neurosci 11:316–328. https://doi.org/10.1038/nrn2836
Kannangara TS, Bostrom CA, Ratzlaff A et al (2014) Deletion of the NMDA receptor GluN2A subunit significantly decreases dendritic growth in maturing dentate granule neurons. PLoS ONE 9:1–10. https://doi.org/10.1371/journal.pone.0103155
Kaster MP, Moretti M, Cunha MP, Rodrigues ALS (2016) Novel approaches for the management of depressive disorders. Eur J Pharmacol 771:236–240. https://doi.org/10.1016/j.ejphar.2015.12.029
Koike H, Iijima M, Chaki S (2013) Effects of ketamine and LY341495 on the depressive-like behavior of repeated corticosterone-injected rats. Pharmacol Biochem Behav 107:20–23. https://doi.org/10.1016/j.pbb.2013.03.017
Kim EJ, Pellman B, Kim JJ (2015) Stress effects on the hippocampus: a critical review. Learn Mem 18:411–416. https://doi.org/10.1101/lm.037291.114
Leal RB, Cordova FM, Herd L et al (2002) Lead-stimulated p38MAPK-dependent Hsp27 phosphorylation. Toxicol Appl Pharmacol 178:44–51. https://doi.org/10.1006/taap.2001.9320
Lee AL, Ogle WO, Sapolsky RM (2002) Stress and depression: Possible links to neuron death in the hippocampus. Bipolar Disord 4:117–128. https://doi.org/10.1034/j.1399-5618.2002.01144.x
Lee MS, Kim YH, Park WS et al (2016) Temporal variability of glucocorticoid receptor activity is functionally important for the therapeutic action of fluoxetine in the hippocampus. Mol Psychiatry 21:252–260. https://doi.org/10.1038/mp.2014.137
Li N, Lee B, Liu RJ et al (2010) mTOR-dependent synapse formation underlies the rapid antidepressant effects of NMDA antagonists. Science 329:959–964. https://doi.org/10.1126/science.1190287
Li N, Liu RJ, Dwyer JM et al (2011) Glutamate N-methyl-D-aspartate receptor antagonists rapidly reverse behavioral and synaptic deficits caused by chronic stress exposure. Biol Psychiatry 69:754–761. https://doi.org/10.1016/j.biopsych.2010.12.015
Mizoguchi K, Ishige A, Aburada M, Tabira T (2003) Chronic stress attenuates glucocorticoid negative feedback: Involvement of the prefrontal cortex and hippocampus. Neuroscience 119:887–897. https://doi.org/10.1016/S0306-4522(03)00105-2
Modell S, Yassouridis A, Huber J, Holsboer F (1997) Corticosteroid receptor function is decreased in depressed patients. Neuroendocrinology 65:216–222. https://doi.org/10.1159/000127275
Morais M, Santos PAR, Mateus-Pinheiro A et al (2014) The effects of chronic stress on hippocampal adult neurogenesis and dendritic plasticity are reversed by selective MAO-A inhibition. J Psychopharmacol 28:1178–1183. https://doi.org/10.1177/0269881114553646
Morales-Medina JC, Sanchez F, Flores G et al (2009) Morphological reorganization after repeated corticosterone administration in the hippocampus, nucleus accumbens and amygdala in the rat. J Chem Neuroanat 38:266–272. https://doi.org/10.1016/j.jchemneu.2009.05.009
Neis VB, Bettio LB, Moretti M et al (2018) Single administration of agmatine reverses the depressive-like behavior induced by corticosterone in mice: Comparison with ketamine and fluoxetine. Pharmacol Biochem Behav 173:44–50. https://doi.org/10.1016/j.pbb.2018.08.005
Okret S, Poellinger L, Dong Y, Gustafson JA (1986) Down-regulation of glucocorticoid receptor mRNA by glucocorticoid hormones and recognition by the receptor of a specific binding sequence within a receptor cDNA clone. Proc Natl Acad Sci USA 83:5899–5903. https://doi.org/10.1073/pnas.83.16.5899
Okugawa O, Suzukawa, et al (2001) Long-term treatment with antidepressants increases glucocorticoid receptor binding and gene expression in cultured rat hippocampal neurones. J Neuroendocrinol 11:887–895. https://doi.org/10.1046/j.1365-2826.1999.00405.x
Olescowicz G, Neis VB, Fraga DB et al (2018) Antidepressant and pro-neurogenic effects of agmatine in a mouse model of stress induced by chronic exposure to corticosterone. Prog Neuropsychopharmacol Biol Psychiatry 81:395–407. https://doi.org/10.1016/j.pnpbp.2017.08.017
Olescowicz G, Sampaio T, Nascimento-Castro CP et al (2020) Protective effects of agmatine against corticosterone-induced impairment on hippocampal mTOR signaling and cell death. Neurotox Res 38:319–329. https://doi.org/10.1007/s12640-020-00212-1
Otte C, Gold S, Penninx B et al (2016) Major depressive disorder. Nat Rev Dis Primers 2:1–20. https://doi.org/10.1038/nrdp.2016.65
Papakostas GI, Ionescu DF (2015) Towards new mechanisms: An update on therapeutics for treatment-resistant major depressive disorder. Mol Psychiatry 20:1142–1150. https://doi.org/10.1038/mp.2015.92
Pariante CM, Alhaj HA, Arulnathan VE et al (2012) Central glucocorticoid receptor-mediated effects of the antidepressant, citalopram, in humans: A study using EEG and cognitive testing. Psychoneuroendocrinology 37:618–628. https://doi.org/10.1016/j.psyneuen.2011.08.011
Pariante CM, Lightman SL (2008) The HPA axis in major depression: classical theories and new developments. Trends Neurosci 31:464–468. https://doi.org/10.1016/j.tins.2008.06.006
Pazini FL, Cunha MP, Rosa JM et al (2016) Creatine, similar to ketamine, counteracts depressive-like behavior induced by corticosterone via PI3K/Akt/mTOR pathway. Mol Neurobiol 53:6818–6834. https://doi.org/10.1007/s12035-015-9580-9
Pazini FL, Cunha MP, Azevedo D et al (2017) Creatine prevents corticosterone-induced reduction in hippocampal proliferation and differentiation: Possible implication for its antidepressant effect. Mol Neurobiol 54:6245–6260. https://doi.org/10.1007/s12035-016-0148-0
Pedrazzoli M, Losurdo M, Paolone G et al (2019) Glucocorticoid receptors modulate dendritic spine plasticity and microglia activity in an animal model of Alzheimer’s disease. Neurobiol Dis 132:104568. https://doi.org/10.1016/j.nbd.2019.104568
Peterson GLA, simplification of the protein assay method of Lowry, et al (1977) which is more generally applicable. Anal Biochem 83:346–356. https://doi.org/10.1016/0003-2697(77)90043-4
Price RB, Nock MK, Charney DS, Mathew SJ (2009) Effects of intravenous ketamine on explicit and implicit measures of suicidality in treatment-resistant depression. Biol Psychiatry 66:522–526. https://doi.org/10.1016/j.biopsych.2009.04.029
Richter-Levin G, Xu L (2018) How could stress lead to major depressive disorder? IBRO Reports 4:38–43. https://doi.org/10.1016/j.ibror.2018.04.001
Smith SM, Vale WW (2006) The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues Clin. Neurosci. 8:383–395. https://doi.org/10.31887/DCNS.2006.8.4/ssmith.
Sterner EY, Kalynchuk LE (2010) Behavioral and neurobiological consequences of prolonged glucocorticoid exposure in rats: Relevance to depression. Prog Neuropsychopharmacol Biol Psychiatry 34:777–790. https://doi.org/10.1016/j.pnpbp.2010.03.005
Swanson AM, Shapiro LP, Whyte AJ, Gourley SL (2013) Glucocorticoid receptor regulation of action selection and prefrontal cortical dendritic spines. Commun Integr Biol 6:e26068. https://doi.org/10.4161/cib.26068
Treccani G, Ardalan M, Chen F et al (2019) S-Ketamine reverses hippocampal dendritic spine deficits in flinders sensitive line rats within 1 h of administration. Mol Neurobiol 56:7368–7379. https://doi.org/10.1007/s12035-019-1613-3
Wang G, Cheng Y, Gong M et al (2013) Systematic correlation between spine plasticity and the anxiety/depression-like phenotype induced by corticosterone in mice. NeuroReport 24:682–687. https://doi.org/10.1097/WNR.0b013e32836384db
Wang W, Liu L, Yang X et al (2019) Ketamine improved depressive-like behaviors via hippocampal glucocorticoid receptor in chronic stress induced- susceptible mice. Behav Brain Res 364:75–84. https://doi.org/10.1016/j.bbr.2019.01.057
Watson S, Mackin P (2006) HPA axis function in mood disorders. Psychiatry 5:166–170. https://doi.org/10.1383/psyt.2006.5.5.166
World Health Organization (2017) Depression and other common mental disorders: global health estimates. World Health Organization 1–24. CCBY-NC-SA3.0IGO
Yau SY, Li A, Bin TJ et al (2016) Chronic corticosterone administration reduces dendritic complexity in mature, but not young granule cells in the rat dentate gyrus. Restor Neurol Neurosci 34:849–857. https://doi.org/10.3233/RNN-160662
Yoshimura S, Sakamoto S, Kudo H et al (2003) Sex-differences in adrenocortical responsiveness during development in rats. Steroids 68:439–445. https://doi.org/10.1016/S0039-128X(03)00045-X
Zarate CA, Singh JB, Carlson PJ et al (2006) A randomized trial of an N-methyl-D-aspartate antagonist in treatment-resistant major depression. Arch Gen Psychiatry 63:856–864. https://doi.org/10.1001/archpsyc.63.8.856.10.3233/RNN-160662
Zeni ALB, Camargo A, Dalmagro AP (2019) Lutein prevents corticosterone-induced depressive-like behavior in mice with the involvement of antioxidant and neuroprotective activities. Pharmacol Biochem Behav 179:63–72. https://doi.org/10.1016/j.pbb.2019.02.004
Zhang K, Pan X, Wang F et al (2016) Baicalin promotes hippocampal neurogenesis via SGK1- and FKBP5-mediated glucocorticoid receptor phosphorylation in a neuroendocrine mouse model of anxiety/depression. Sci Rep 6:1–9. https://doi.org/10.1038/srep30951
Zhao Y, Xie W, Dai J et al (2009) The varying effects of short-term and long-term corticosterone injections on depression-like behavior in mice. Brain Res 1261:82–90. https://doi.org/10.1016/j.brainres.2008.12.083
Acknowledgments
The authors thank funding agencies CNPq and CAPES by the financial support, and the Laboratório Multiusuário de Estudos em Biologia (LAMEB) by technical support.
Funding
This study was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, #310113/2017–2), and Coordenação de Aperfeiçoamento de Pessoal de Ensino Superior (CAPES). JB, PSB, and ALSR are CNPq Research Fellows.
Author information
Authors and Affiliations
Contributions
Ana Lúcia S. Rodrigues and Daiane B. Fraga designed the study and wrote the protocol. Daiane B. Fraga, Gislaine Olescowicz, Dayane Azevedo, Francielle Mina, Patricia S. Brocardo, Josiane Budni, and Anderson Camargo performed the neurochemical and morphological analyses. Daiane B. Fraga and Anderson Camargo undertook the statistical analysis. All authors wrote the first draft of the manuscript, as well as approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All procedures were carried out in accordance with the National Institute of Health Guide for the Care and Use of Laboratory Animals and the protocols were approved by the Institutional Ethics Committee.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fraga, D.B., Camargo, A., Olescowicz, G. et al. Ketamine, but not fluoxetine, rapidly rescues corticosterone-induced impairments on glucocorticoid receptor and dendritic branching in the hippocampus of mice. Metab Brain Dis 36, 2223–2233 (2021). https://doi.org/10.1007/s11011-021-00743-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-021-00743-2