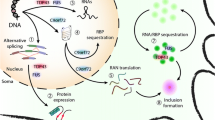
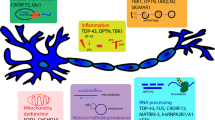
Abstract
Amyotrophic lateral sclerosis (ALS) is a deadly motor neuron disease (MND) and the most frequent MND in adults. ALS is recognized by degenerative alterations in both upper and lower motor neurons. This disorder is classified to familial and sporadic classes. Disease-causing mutations in SOD1, C9ORF72, FUS, and TARDBP have been recognized in familial ALS cases. However, in spite of conduction of several genetic association studies, heritable genetic risk elements in sporadic have not been identified completely. Several miRNAs have been dysregulated in the serum samples or brain tissues of ALS patients. Moreover, a number of miRNAs have been suggested as putative biomarkers for sporadic ALS. In the current manuscript, we review of miRNAs in the development of ALS.

Similar content being viewed by others
References
Alsultan AA, Waller R, Heath PR, Kirby J (2016) The genetics of amyotrophic lateral sclerosis: current insights. Degener Neurol Neuromuscul Dis 6:49
Boylan K (2015) Familial amyotrophic lateral sclerosis. Neurol Clin 33:807–830
Campos-Melo D, Droppelmann CA, He Z, Volkening K, Strong MJ (2013) Altered microRNA expression profile in Amyotrophic Lateral Sclerosis: a role in the regulation of NFL mRNA levels Mol Brain 6:26. https://doi.org/10.1186/1756-6606-6-26
Cardoso AL, Guedes JR, Pereira de Almeida L, Pedroso de Lima MC (2012) miR-155 modulates microglia-mediated immune response by down-regulating SOCS-1 and promoting cytokine and nitric oxide production. Immunology 135:73–88
Chen S, Sayana P, Zhang X, Le W (2013) Genetics of amyotrophic lateral sclerosis: an update. Mole Neurodegener 8:28
Chiò A, Logroscino G, Hardiman O, Swingler R, Mitchell D, Beghi E, Traynor BG (2009) Prognostic factors in ALS: A critical review Amyotrophic lateral sclerosis : official publication of the World Federation of Neurology Research Group. Motor Neuron Diseases 10:310–323. https://doi.org/10.3109/17482960802566824
Chio A et al (2009) Prognostic factors in ALS: a critical review. Amyotroph Lateral Scler 10:310–323
Chiò A, Logroscino G, Traynor B, Collins J, Simeone J, Goldstein L, White L (2013) Global epidemiology of amyotrophic lateral sclerosis: a systematic review of the published literature. Neuroepidemiology 41:118–130
De Felice B, Guida M, Guida M, Coppola C, De Mieri G, Cotrufo R (2012) A miRNA signature in leukocytes from sporadic amyotrophic lateral sclerosis. Gene 508:35–40. https://doi.org/10.1016/j.gene.2012.07.058
De Felice B, Manfellotto F, Fiorentino G, Annunziata A, Biffali E, Pannone R, Federico A (2018) Wide-Ranging Analysis of MicroRNA Profiles in Sporadic Amyotrophic Lateral Sclerosis Using Next-Generation Sequencing. Frontiers in genetics 9:310. https://doi.org/10.3389/fgene.2018.00310
Diekstra FP et al (2014) C9orf72 and UNC13A are shared risk loci for amyotrophic lateral sclerosis and frontotemporal dementia: a genome-wide meta-analysis. Annals Neurol 76:120–133. https://doi.org/10.1002/ana.24198
Dobrowolny G, Bernardini C, Martini M, Baranzini M, Barba M, Musaro A (2015) Muscle expression of SOD1(G93A) modulates microRNA and mRNA transcription pattern associated with the myelination process in the spinal cord of transgenic mice. Front Cell Neurosci 9:463. https://doi.org/10.3389/fncel.2015.00463
Dobrowolny G, Martone J., Lepore E., Casola I., Petrucci A., Inghilleri M., Morlando M., Colantoni A., Scicchitano B.M., Calvo A., Bisogni G., Chiò A., Sabatelli M., Bozzoni I., Musarò A. (2021) A longitudinal study defined circulating microRNAs as reliable biomarkers for disease prognosis and progression in ALS human patients Cell Death Discovery 7:4
Figueroa-Romero C, Hur J, Lunn JS, Paez-Colasante X, Bender DE, Yung R, Sakowski SA, Feldman EL (2016) Expression of microRNAs in human post-mortem amyotrophic lateral sclerosis spinal cords provides insight into disease mechanisms. Mol Cell Neurosci 71:34–45. https://doi.org/10.1016/j.mcn.2015.12.008
Greenway M et al (2004) A novel candidate region for ALS on chromosome 14q11. 2. Neurology 63:1936–1938
Guerrero EN, Wang H, Mitra J, Hegde PM, Stowell SE, Liachko NF, Kraemer BC, Garruto RM, Rao KS, Hegde ML (2016) TDP-43/FUS in motor neuron disease: complexity and challenges. Prog Neurobiol 145-146:78–97. https://doi.org/10.1016/j.pneurobio.2016.09.004
Hirano M, Quinzii CM, Mitsumoto H, Hays AP, Roberts JK, Richard P, Rowland LP (2011) Senataxin mutations and amyotrophic lateral sclerosis Amyotrophic lateral sclerosis : official publication of the World Federation of Neurology Research Group. Motor Neuron Dis 12:223–227. https://doi.org/10.3109/17482968.2010.545952
Joilin G et al (2020) Identification of a potential non-coding RNA biomarker signature for amyotrophic lateral sclerosis. Brain Comm 2:fcaa053. https://doi.org/10.1093/braincomms/fcaa053
Joilin G, Leigh PN, Newbury SF, Hafezparast M (2019) An Overview of MicroRNAs as Biomarkers of ALS. Front Neurol 10:186. https://doi.org/10.3389/fneur.2019.00186
Kamal MA, Mushtaq G, Greig NH (2015) Current Update on Synopsis of miRNA Dysregulation in Neurological Disorders CNS & neurological disorders. Drug Targets 14:492–501. https://doi.org/10.2174/1871527314666150225143637
Katsu M, Hama Y, Utsumi J, Takashina K, Yasumatsu H, Mori F, Wakabayashi K, Shoji M, Sasaki H (2019) MicroRNA expression profiles of neuron-derived extracellular vesicles in plasma from patients with amyotrophic lateral sclerosis. Neurosci Lett 708:134176. https://doi.org/10.1016/j.neulet.2019.03.048
Konovalova J, Gerasymchuk D, Parkkinen I, Chmielarz P, Domanskyi A (2019) Interplay between MicroRNAs and oxidative stress in neurodegenerative diseases. Int J Mole Sci 20:6055
Koval ED et al (2013) Method for widespread microRNA-155 inhibition prolongs survival in ALS-model micex. Human Mole Gene 22:4127–4135. https://doi.org/10.1093/hmg/ddt261
Li S, Sheng J, Hu JK, Yu W, Kishikawa H, Hu MG, Shima K, Wu D, Xu Z, Xin W, Sims KB, Landers JE, Brown RH Jr, Hu GF (2013) Ribonuclease 4 protects neuron degeneration by promoting angiogenesis, neurogenesis, and neuronal survival under stress. Angiogenesis 16:387–404. https://doi.org/10.1007/s10456-012-9322-9
Liu X, Cheng Y, Chen X, Yang J, Xu L, Zhang C (2011) MicroRNA-31 regulated by the extracellular regulated kinase is involved in vascular smooth muscle cell growth via large tumor suppressor homolog 2. J Biol Chem 286:42371–42380. https://doi.org/10.1074/jbc.M111.261065
Marcuzzo S et al (2014) Altered miRNA expression is associated with neuronal fate in G93A-SOD1 ependymal stem progenitor cells. Exp Neurol 253:91–101. https://doi.org/10.1016/j.expneurol.2013.12.007
Matamala JM, Arias-Carrasco R, Sanchez C, Uhrig M, Bargsted L, Matus S, Maracaja-Coutinho V, Abarzua S, van Zundert B, Verdugo R, Manque P, Hetz C (2018) Genome-wide circulating microRNA expression profiling reveals potential biomarkers for amyotrophic lateral sclerosis. Neurobiol Aging 64:123–138. https://doi.org/10.1016/j.neurobiolaging.2017.12.020
Mejzini R, Flynn LL, Pitout IL, Fletcher S, Wilton SD, Akkari PA (2019) ALS genetics, mechanisms, and therapeutics: where are we now? Front Neurosci 13
Narain P, Padhi AK, Dave U, Mishra D, Bhatia R, Vivekanandan P, Gomes J (2019) Identification and characterization of novel and rare susceptible variants in Indian amyotrophic lateral sclerosis patients. Neurogenetics 20:197–208. https://doi.org/10.1007/s10048-019-00584-3
Nguyen HP, Van Broeckhoven C, van der Zee J (2018) ALS genes in the genomic era and their implications for FTD Trends Genet 34:404–423
Padhi AK, Banerjee K, Gomes J, Banerjee M (2014) Computational and functional characterization of Angiogenin mutations, and correlation with amyotrophic lateral sclerosis. PLoS One 9:e111963. https://doi.org/10.1371/journal.pone.0111963
Padhi AK, Jayaram B, Gomes J (2013) Prediction of functional loss of human angiogenin mutants associated with ALS by molecular dynamics simulations Sci Rep 3:1225. https://doi.org/10.1038/srep01225
Padhi AK, Kumar H, Vasaikar SV, Jayaram B, Gomes J (2012) Mechanisms of loss of functions of human angiogenin variants implicated in amyotrophic lateral sclerosis. PloS one 7:e32479. https://doi.org/10.1371/journal.pone.0032479
Padhi AK, Narain P, Gomes J (2019) Rare Angiogenin and Ribonuclease 4 variants associated with amyotrophic lateral sclerosis exhibit loss-of-function: a comprehensive in silico study. Metab Brain Dis 34:1661–1677. https://doi.org/10.1007/s11011-019-00473-6
Padhi AK, Zhang KYJ (2020) Mechanistic insights into the loss-of-function mechanisms of rare human D-amino acid oxidase variants implicated in amyotrophic lateral sclerosis. Sci Rep 10:17146. https://doi.org/10.1038/s41598-020-74048-2
Paez-Colasante X, Figueroa-Romero C, Sakowski SA, Goutman SA, Feldman EL (2015) Amyotrophic lateral sclerosis: mechanisms and therapeutics in the epigenomic era. Nat Rev Neurol 11:266
Parisi C, Arisi I, D'Ambrosi N, Storti AE, Brandi R, D'Onofrio M, Volonte C (2013) Dysregulated microRNAs in amyotrophic lateral sclerosis microglia modulate genes linked to neuroinflammation. Cell Death Dis 4:e959. https://doi.org/10.1038/cddis.2013.491
Pesiridis GS, Lee VM, Trojanowski JQ (2009) Mutations in TDP-43 link glycine-rich domain functions to amyotrophic lateral sclerosis. Hum Mol Genet 18:R156–R162. https://doi.org/10.1093/hmg/ddp303
Renton AE et al (2011) A hexanucleotide repeat expansion in C9ORF72 is the cause of chromosome 9p21-linked ALS-FTD. Neuron 72:257–268. https://doi.org/10.1016/j.neuron.2011.09.010
Robinson R (2017) FDA approves Edaravone for ALS: phase 3 trial finds it slows progression. Neurol Today 17:1–30
Rosen DR et al (1993) Mutations in cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature 362:59–62. https://doi.org/10.1038/362059a0
Rowland LP, Shneider NA (2001) Amyotrophic lateral sclerosis New England. J Med 344:1688–1700
Russell AP, Wada S, Vergani L, Hock MB, Lamon S, Léger B, Ushida T, Cartoni R, Wadley GD, Hespel P, Kralli A, Soraru G, Angelini C, Akimoto T (2013) Disruption of skeletal muscle mitochondrial network genes and miRNAs in amyotrophic lateral sclerosis. Neurobiol Dis 49:107–117. https://doi.org/10.1016/j.nbd.2012.08.015
Si Y, Cui X, Crossman DK, Hao J, Kazamel M, Kwon Y, King PH (2018) Muscle microRNA signatures as biomarkers of disease progression in amyotrophic lateral sclerosis. Neurobiol Dis 114:85–94. https://doi.org/10.1016/j.nbd.2018.02.009
Taguchi YH, Wang H (2018) Exploring microRNA biomarker for amyotrophic lateral sclerosis. Int J Mole Sci 19. https://doi.org/10.3390/ijms19051318
Toivonen JM, Manzano R, Olivan S, Zaragoza P, Garcia-Redondo A, Osta R (2014) MicroRNA-206: a potential circulating biomarker candidate for amyotrophic lateral sclerosis. PLoS One 9:e89065. https://doi.org/10.1371/journal.pone.0089065
Wakabayashi K, Mori F, Kakita A, Takahashi H, Utsumi J, Sasaki H (2014) Analysis of microRNA from archived formalin-fixed paraffin-embedded specimens of amyotrophic lateral sclerosis. Acta Neuropathol Commun 2:173. https://doi.org/10.1186/s40478-014-0173-z
Wang L, Zhang L (2020) MicroRNAs in amyotrophic lateral sclerosis: from pathogenetic involvement to diagnostic biomarker and therapeutic agent development. Neurol Sci : Off J Italian Neurol Soc Italian Soc Clin Neurophysiol 41:3569–3577. https://doi.org/10.1007/s10072-020-04773-z
Yoshimura A, Naka T, Kubo M (2007) SOCS proteins, cytokine signalling and immune regulation. Nat Rev Immunol 7:454–465
Zoccolella S et al (2007) Riluzole and amyotrophic lateral sclerosis survival: a population-based study in southern Italy. Eur J Neurol 14:262–268. https://doi.org/10.1111/j.1468-1331.2006.01575.x
Zou Z-Y, Zhou Z-R, Che C-H, Liu C-Y, He R-L, Huang H-P (2017) Genetic epidemiology of amyotrophic lateral sclerosis: a systematic review and meta-analysis journal of neurology. Neurosurg Psychiatry 88:540–549
Acknowledgements
This study was financially supported by Shahid Beheshti University of Medical Sciences.
Availability of data and materials
The analyzed data sets generated during the study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Contributions
MT and SGF wrote the draft and revised it. NAD, SN and BMH collected the data, designed the tables and figures. All the authors contribute equally and read the submission.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent forms were obtained from all study participants. The study protocol was approved by the ethical committee of Shahid Beheshti University of Medical Sciences. All methods were performed in accordance with the relevant guidelines and regulations.
Consent for publication
Not applicable.
Competing interests
The authors declare they have nothing to report.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Akbari Dilmaghani, N., Hussen, B.M., Nateghinia, S. et al. Emerging role of microRNAs in the pathogenesis of amyotrophic lateral sclerosis. Metab Brain Dis 36, 737–749 (2021). https://doi.org/10.1007/s11011-021-00697-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-021-00697-5