Abstract
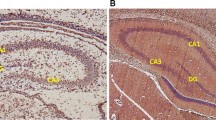
Pregnant women constitute about half the users of methamphetamine (MA), in whom the consumption may continue during breastfeeding. This study aimed to evaluate the effects of MA use during pregnancy and lactation on the hippocampus of pups. 35 pregnant Wistar rats were divided into seven groups, including three experimental groups daily administered with 5 mg/kg of MA (i.p.) during the prenatal and/or postnatal period (PND1–22). In addition, three sham control groups received normal saline at the same dose, and one normal control group received no interventions since early pregnancy until the end of lactation. After the interventions, two pups (aged one and 22 days) were randomly selected from each mother and their brain tissue sections were prepared to determine the expression of PSA-NCAM molecules and sialic acids using immunohistochemical and lectinhistochemical techniques, respectively. In one-day infant rats with MA exposure during pregnancy, a significant decrease was observed in the number of PSA-NCAM positive cells in the CA1 (P = 0.047), CA3 (P = 0.05) and DG (P = 0.006) hippocampus regions compared to control and expression intensity of these molecules in all the three regions (P ≤ 0.05). Moreover, in 22-day pups with MA exposure during pregnancy and lactation, number of PSA-NCAM positive cells and expression intensity of these molecules significantly reduced in all the three regions of the hippocampus (P ≤ 0.05). Findings regarding the intensity of sialic acid expression were aligned with PSA-NCAM expression. According to our results, MA administration during pregnancy and lactation may effect on polysialic acid-neural cell adhesion molecule expression in rat’s offspring hippocampus.








Similar content being viewed by others
Reference
Abrous DN, Adriani W, Montaron M-F, Aurousseau C, Rougon G, Le Moal M et al (2002) Nicotine self-administration impairs hippocampal plasticity. J Neurosci 22(9):3656–3662
Acuff-Smith KD, Schilling MA, Fisher JE, Vorhees CV (1996) Stage-specific effects of prenatal d-methamphetamine exposure on behavioral and eye development in rats. Neurotoxicol Teratol 18(2):199–215
Bartu A, Dusci LJ, Ilett KF (2009) Transfer of methylamphetamine and amphetamine into breast milk following recreational use of methylamphetamine. Br J Clin Pharmacol 67(4):455–459
Bideskan E, Reza A, Hassanzadeh Taheri MM, Nikravesh MR, Fazel AR (2011) Lectin histochemical study of vasculogenesis during rat pituitary morphogenesis. Iranian Journal of Basic Medical Sciences 14(1):35–41
Bowyer JF, Ali S (2006) High doses of methamphetamine that cause disruption of the blood-brain barrier in limbic regions produce extensive neuronal degeneration in mouse hippocampus. Synapse 60(7):521–532
Cho Dh, Lyu HM, Lee HB, Kim PY, Chin K (1991) Behavioral Teratogenicity of Methamphetamine. J Toxicol Sci 16(SupplementI):37–49
Cho AK, Melega WP, Kuczenski R, Segal DS (2001) Relevance of pharmacokinetic parameters in animal models of methamphetamine abuse. Synapse 39(2):161–166
Cui C, Sakata-Haga H (2006) Ohta Ki, Nishida M, Yashiki M, Sawada K, et al. histological brain alterations following prenatal methamphetamine exposure in rats. Congenital Anomalies 46(4):180–187
Ebrahimzadeh-Bideskan A-R, Hami J, Alipour F, Haghir H, Fazel A-R, Sadeghi A (2016) Protective effects of ascorbic acid and garlic extract against lead-induced apoptosis in developing rat hippocampus. Metab Brain Dis 31(5):1123–1132
Gao X, Yin S, Tang M, Chen J, Yang Z, Zhang W et al (2011) Effects of developmental exposure to TiO2 nanoparticles on synaptic plasticity in hippocampal dentate gyrus area: an in vivo study in anesthetized rats. Biol Trace Elem Res 143(3):1616–1628
Gascon E, Vutskits L, Jenny B, Durbec P, Kiss JZ (2007) PSA-NCAM in postnatally generated immature neurons of the olfactory bulb: a crucial role in regulating p75 expression and cell survival. Development 134(6):1181–1190
Gorman MC, Orme KS, Nguyen NT, Kent EJ, Caughey AB (2014) Outcomes in pregnancies complicated by methamphetamine use. Am J Obstet Gynecol 211(4):429 e1. e7
Heidari-Rarani M, Noori A, Ghodousi A (2014) Effects of methamphetamine on pituitary gonadal axis and spermatogenesis in mature male rats. Zahedan Journal of Research in Medical Sciences 16(12):37–42
Heidmets L-T, Kalda A, Zharkovsky A (2007) Acute amphetamine treatment decreases the expression of 180–200 kDa isoform of polysialic acid linked neural cell adhesion molecule in mouse hippocampus. Brain Res 1165:89–97
Kahn L, Alonso G, Normand E, Manzoni OJ (2005) Repeated morphine treatment alters polysialylated neural cell adhesion molecule, glutamate decarboxylase-67 expression and cell proliferation in the adult rat hippocampus. Eur J Neurosci 21(2):493–500
Khoradmehr A, Danafar A, Halvaei I, Golzadeh J, Hosseini M, Mirjalili T et al (2015) Effect of prenatal methamphetamine administration during gestational days on mice. Iranian Journal of Reproductive Medicine 13(1):41
Labarca R, Gajardo MI, Seguel M, Silva H, Jerez S, Ruiz A et al (1995) Effects of D-amphetamine administration on the release of endogenous excitatory amino acids in the rat nucleus accumbens. Prog Neuro-Psychopharmacol Biol Psychiatry 19(3):467–473
Ladhani NNN, Shah PS, Murphy KE, Births KSGDPL (2011) Prenatal amphetamine exposure and birth outcomes: a systematic review and metaanalysis. Am J Obstet Gynecol 205(3):219 e1-. e7
Leino O, Kiviranta H, Karjalainen A, Kronberg-Kippilä C, Sinkko H, Larsen EH et al (2013) Pollutant concentrations in placenta. Food Chem Toxicol 54:59–69
Maćkowiak M, Markowicz-Kula K, Fijał K, Wędzony K (2005) Acute and repeated administration of cocaine differentially regulates expression of PSA-NCAM-positive neurons in the rat hippocampus. Brain Res 1055(1):149–155
Maćkowiak M, Chocyk A, Markowicz-Kula K, Wędzony K (2007) Acute activation of CB1 cannabinoid receptors transiently decreases PSA-NCAM expression in the dentate gyrus of the rat hippocampus. Brain Res 1148:43–52
Macúchová E, Nohejlová-Deykun K, Slamberova R (2013) Effect of methamphetamine on cognitive functions of adult female rats prenatally exposed to the same drug. Physiol Res 62:S89
Manzi AE, Higa HH, Diaz S, Varki A (1994) Intramolecular self-cleavage of polysialic acid. J Biol Chem 269(38):23617–23624
Marwick C (2000) NIDA seeking data on effect of fetal exposure to methamphetamine. JAMA 283(17):2225–2226
Mazzetti S, Ortino B, Inverardi F, Frassoni C, Amadeo A (2007) PSA-NCAM in the developing and mature thalamus. Brain Res Bull 71(6):578–586
McDonnell-Dowling K, Kelly JP (2015) The consequences of prenatal and/or postnatal methamphetamine exposure on neonatal development and behaviour in rat offspring. Int J Dev Neurosci 47:147–156
McDonnell-Dowling K, Kelly JP (2016) Does route of methamphetamine exposure during pregnancy have an impact on neonatal development and behaviour in rat offspring? Int J Dev Neurosci 49:14–22
Minana R, Duran J, Tomas M, Renau-Piqueras J, Guerri C (2001) Neural cell adhesion molecule is endocytosed via a clathrin-dependent pathway. Eur J Neurosci 13(4):749–756
Mirjalili T, Kalantar SM, Lahijani MS, Sheikhha MH, Talebi A (2013) Congenital effects of methamphetamine on mice fetouses at histological, cellular and chromosomal levels. Iranian Journal of Reproductive Medicine 11(1):39–46
Mohammadzadeh E, Nikravesh MR, Jalali M, Fazel A, Ebrahimi V, Ebrahimzadeh-bideskan AR (2014) Immunohistochemical study of type III collagen expression during pre and post-natal rat skin morphogenesis. Iranian Journal of Basic Medical Sciences 17(3):196–200
Montoya ID, McCann DJ (2010) Drugs of abuse: management of intoxication and antidotes. Springer, Molecular, Clinical and Environmental Toxicology, pp 519–541
Rajabzadeh AA, Bideskan ARE, Haghir H, Fazel AR (2011) Morphometrical study of polysialylated neural cell adhesion molecule positive cells in rat pups hippocampus following induction of seizure during pregnancy. Iran Biomed J 15(4):157
Rambousek L, Kacer P, Syslova K, Bumba J, Bubenikova-Valesova V, Slamberova R (2014) Sex differences in methamphetamine pharmacokinetics in adult rats and its transfer to pups through the placental membrane and breast milk. Drug Alcohol Depend 139:138–144
Shingo A, Kito S (2005) Effects of nicotine on neurogenesis and plasticity of hippocampal neurons. J Neural Transm 112(11):1475–1478
Smith DE, Moser C, Wesson DR, Apter M, Buxton ME, Davison JV et al (1982) A clinical guide to the diagnosis and treatment of heroin-related sexual dysfunction. J Psychoactive Drugs 14(1–2):91–99
Smith L, Yonekura ML, Wallace T, Berman N, Kuo J, Berkowitz C (2003) Effects of prenatal methamphetamine exposure on fetal growth and drug withdrawal symptoms in infants born at term. J Dev Behav Pediatr 24(1):17–23
Vavrinkova B, Binder T, Zivný J (2001) Characteristics of a population of drug dependent pregnant women in the Czech Republic. Ceska gynekologie/Ceska lekarska spolecnost J Ev Purkyne 66(4):285–291
Williams MT, Morford LL, Wood SL, Wallace TL, Fukumura M, Broening HW et al (2003) Developmental D-methamphetamine treatment selectively induces spatial navigation impairments in reference memory in the Morris water maze while sparing working memory. Synapse 48(3):138–148
Won L, Bubula N, McCoy H, Heller A (2001) Methamphetamine concentrations in fetal and maternal brain following prenatal exposure. Neurotoxicol Teratol 23(4):349–354
Wouldes T, LaGasse L, Sheridan J, Lester B (2004) Maternal methamphetamine use during pregnancy and child outcome: what do we know. NZ Med J 117(1206):1–10
Acknowledgements
This study was financially supported by the Research Department of Mashhad University of Medical Sciences, Mashhad, Iran. Data presented in this paper were extracted from a master”s thesis. Hereby, we extend our gratitude to Dr. Sokhtanloo for providing the methamphetamine drug and Ms. F. Motejadded for technical and laboratory assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baei, F., Rajabzadeh, A., Bagheri, J. et al. Effect of methamphetamine exposure during pregnancy and lactation on polysialic acid-neural cell adhesion molecule expression in rat’s offspring hippocampus. Metab Brain Dis 32, 991–1002 (2017). https://doi.org/10.1007/s11011-017-9973-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-017-9973-8