Abstract
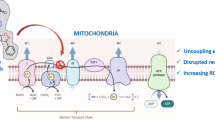
Modulation of mitochondrial bioenergetics and glycolysis in malignancies has recently emerged a potential chemotherapeutic strategy since numerous malignant cells have overcome inhibition of the glycolytic pathway by increasing mitochondrial ATP production. Quercetin is a flavonoid with antioxidant, antiangiogenic, and chemoprotective properties but the mitochondrial effects are less characterized. The present study was purported to assess the effects of quercetin on the bioenergetic profile of B164A5 murine melanoma cell line. The oxygen consumption rate (OCR) and extracellular acidification rate (ECAR) were measured at 24, 48, and 72 h of treatment of B164A5 murine melanoma cells with increasing concentrations (25, 50, 100, and 150 µM) of quercetin using the extracellular flux analyzer Seahorse XF24e (Seahorse Agilent). Analysis of mitochondrial function was performed in the presence of the classic modulators of the electron transport chain: oligomycin, FCCP, and rotenone. 72-h treatment with quercetin induced a dose-dependent decrease of all OCR parameters (basal respiration, proton leak, ATP turnover, maximal respiration, reserve capacity) as well as of ECAR. At variance, 48-h treatment induced a decrease of OCR and ECAR when quercetin was applied at 50, 100, and 150 µM, while the 24-h treatment induced a decrease of bioenergetic parameters only for the highest concentrations (100 and 150 µM) of the compound. Our data clearly demonstrated that quercetin elicited dose-dependent inhibitory effect on examined parameters of cellular bioenergetics that was most potent at 72 h of treatment. Thereby quercetin, modulating both glycolytic and mitochondrial pathways for ATP production, might be an efficient approach in killing cancer cells.



Similar content being viewed by others
References
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F (2015) Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 136:E359–E386. https://doi.org/10.1002/ijc.29210
Modica-Napolitano JS, Weissig V (2015) Treatment strategies that enhance the efficacy and selectivity of mitochondria-targeted anticancer agents. Int J Mol Sci 16:17394–17421. https://doi.org/10.3390/ijms160817394
Russo M, Spagnuolo C, Tedesco I, Russo GL (2010) Phytochemicals in cancer prevention and therapy: truth or dare? Toxins 2:517–551. https://doi.org/10.3390/toxins2040517
Farzaei MH, Bahramsoltani R, Rahimi R (2016) Phytochemicals as adjunctive with conventional anticancer therapies. Curr Pharm Des 22:4201–4218
Badalica M, Munteanu M, Sturza A, Noveanu L, Streian CG, Socaciu C, Muntean D, Timar R, Dragan S (2014) Characterization of the effects of two polyphenols-rich plant extracts on isolated diabetic human mammary arteries. Rev Chim 65:861–864
Chiarugi P, Cirri P (2016) Metabolic exchanges within tumor microenvironment. Cancer Lett 380:272–280. https://doi.org/10.1016/j.canlet.2015.10.027
Sak K (2014) Site-specific anticancer effects of dietary flavonoid quercetin. Nutr Cancer 66:177–193. https://doi.org/10.1080/01635581.2014.864418
Thangasamy T, Sittadjody S, Lanza-Jacoby S, Wachsberger PR, Limesand KH, Burd R (2007) Quercetin selectively inhibits bioreduction and enhances apoptosis in melanoma cells that overexpress tyrosinase. Nutr Cancer 59:258–268. https://doi.org/10.1080/01635580701499545
Anand David AV, Arulmoli R, Parasuraman S (2016) Overviews of biological importance of quercetin: a bioactive flavonoid. Pharmacogn Rev 10:84–89. https://doi.org/10.4103/0973-7847.194044
Kashyap D, Mittal S, Sak K, Singhal P, Tuli HS (2016) Molecular mechanisms of action of quercetin in cancer: recent advances. Tumour Biol 37:12927–12939. https://doi.org/10.1007/s13277-016-5184-x
Niedzwiecki A, Roomi MW, Kalinovsky T, Rath M (2016) Anticancer efficacy of polyphenols and their combinations. Nutrients. https://doi.org/10.3390/nu8090552
Brito AF, Ribeiro M, Abrantes AM, Pires AS, Teixo RJ, Tralhao JG, Botelho MF (2015) Quercetin in cancer treatment, alone or in combination with conventional therapeutics? Curr Med Chem 22:3025–3039
Zhang XM, Chen J, Xia YG, Xu Q (2005) Apoptosis of murine melanoma B16-BL6 cells induced by quercetin targeting mitochondria, inhibiting expression of PKC-alpha and translocating PKC-delta. Cancer Chemother Pharmacol 55:251–262. https://doi.org/10.1007/s00280-004-0863-5
Lowe GC, Saavedra A, Reed KB, Velazquez AI, Dronca RS, Markovic SN, Lohse CM, Brewer JD (2014) Increasing incidence of melanoma among middle-aged adults: an epidemiologic study in Olmsted County, Minnesota. Mayo Clin Proc 89:52–59. https://doi.org/10.1016/j.mayocp.2013.09.014
Horvathova K, Chalupa I, Sebova L, Tothova D, Vachalkova A (2005) Protective effect of quercetin and luteolin in human melanoma HMB-2 cells. Mutat Res 565:105–112. https://doi.org/10.1016/j.mrgentox.2004.08.013
Li Y, Yao J, Han C, Yang J, Chaudhry MT, Wang S, Liu H, Yin Y (2016) Quercetin, inflammation and immunity. Nutrients 8:167. https://doi.org/10.3390/nu8030167
Harris Z, Donovan MG, Branco GM, Limesand KH, Burd R (2016) Quercetin as an emerging anti-melanoma agent: a four-focus area therapeutic development strategy. Front Nutr 3:48. https://doi.org/10.3389/fnut.2016.00048
Duicu OM, Scurtu I, Popescu R, Sturza A, Coricovac D, Danila DM, Privistirescu A, Muntean MD (2015) Assessment of the effects of methylene blue on cellular bioenergetics in H9c2 cells. Rev Chim 66:519–522
Danciu C, Bojin F, Ambrus R, Muntean D, Soica C, Paunescu V, Cristea M, Pinzaru I, Dehelean C (2016) Physico-chemical and biological evaluation of flavonols: fisetin, quercetin and kaempferol alone and incorporated in beta cyclodextrins. Anticancer Agents Med Chem 17:615–626
Lee JC, Kim J, Park JK, Chung GH, Jang YS (2003) The antioxidant, rather than prooxidant, activities of quercetin on normal cells: quercetin protects mouse thymocytes from glucose oxidase-mediated apoptosis. Exp Cell Res 291:386–397
Arung ET, Furuta S, Ishikawa H, Tanaka H, Shimizu K, Kondo R (2011) Melanin biosynthesis inhibitory and antioxidant activities of quercetin-3′-O-beta-D-glucoside isolated from Allium cepa. Z Naturforsch C 66:209–214
Kim YJ (2012) Hyperin and quercetin modulate oxidative stress-induced melanogenesis. Biol Pharm Bull 35:2023–2027
Yang YM, Son YO, Lee SA, Jeon YM, Lee JC (2011) Quercetin inhibits alpha-MSH-stimulated melanogenesis in B16F10 melanoma cells. Phytother Res 25:1166–1173. https://doi.org/10.1002/ptr.3417
Braun RD, Beatty AL (2007) Modeling of oxygen transport across tumor multicellular layers. Microvasc Res 73:113–123
Brand Martin D, Nicholls David G (2011) Assessing mitochondrial dysfunction in cells. Biochem J 435:297–312. https://doi.org/10.1042/BJ20110162
Sharmila G, Bhat FA, Arunkumar R, Elumalai P, Raja Singh P, Senthilkumar K, Arunakaran J (2014) Chemopreventive effect of quercetin, a natural dietary flavonoid on prostate cancer in in vivo model. Clin Nutr 33:718–726. https://doi.org/10.1016/j.clnu.2013.08.011
Su Q, Peng M, Zhang Y, Xu W, Darko KO, Tao T, Huang Y, Tao X, Yang X (2016) Quercetin induces bladder cancer cells apoptosis by activation of AMPK signaling pathway. Am J Cancer Res 6:498–508
Hardie DG, Alessi DR (2013) LKB1 and AMPK and the cancer-metabolism link-ten years after. BMC Biol 11:36. https://doi.org/10.1186/1741-7007-11-36
Gutheil WG, Reed G, Ray A, Anant S, Dhar A (2012) Crocetin: an agent derived from saffron for prevention and therapy for cancer. Curr Pharm Biotechnol 13(1):173–179
Surh YJ (2003) Cancer chemoprevention with dietary phytochemicals. Nat Rev Cancer 3(10):768–780
Ramanujan VK (2015) Metabolic plasticity in cancer cells: reconnecting mitochondrial function to cancer control. J Cell Sci Ther. https://doi.org/10.4172/2157-7013.1000211
Acknowledgements
The research was funded by the university Grant PIII-C2-PCFI-2015/2016.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sturza, A., Pavel, I., Ancușa, S. et al. Quercetin exerts an inhibitory effect on cellular bioenergetics of the B164A5 murine melanoma cell line. Mol Cell Biochem 447, 103–109 (2018). https://doi.org/10.1007/s11010-018-3296-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-018-3296-x