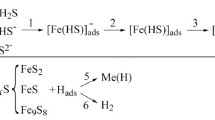
The effect of different concentrations of CO2 and H2S in a chloride-acetate solution on corrosion-mechanical properties of 17G1S-U steel was studied. In a solution saturated with CO2, the corrosion rate of steel was lower than in the presence of H2S, but increased over time due to the absence of protective carbonate films on the surface, plasticity parameters were 2–2.7 times lower than in air due to dimple surface damage. The corrosion rate and hydrogenation of steel was determined primarily by the hydrogen sulfide concentration in the environment. At a concentration of 100 mg/dm3, dense films of the troilite-mackinavite composition were formed, which inhibit corrosion. At higher concentrations, the corrosion rate increased due to the sulfides transformation and the formation of surface layers with defects. With an increase in the H2S concentration from 100 mg/dm3, the strength characteristics of steel decreased in three times, and plasticity decreased in 3–5 times.





Similar content being viewed by others
References
P. Sui, J. Sun, Y. Hua, H. Liu, M. Zhou, Y. Zhang, J. Liu, and Y. Wang, “Effect of temperature and pressure on corrosion behavior of X65 carbon steel in water-saturated CO2 transport environments mixed with H2S,” Int. J. of Greenhouse Gas Control, 73, 60–69 (2018); https://doi.org/10.1016/j.ijggc.2018.04.003
C. Ren, D. Liu, Z. Bai, and T. Li, “Corrosion behavior of oil sulfide in simulant solution with hydrogen sulfide and carbon dioxide,” Mater. Chemistry and Physics, 93, Iss. 2–3, 305–309 (2005); https://doi.org/10.1016/j.matchemphys.2005.03.010
X. H. Zhao, Y. Feng, S. Tang, and J. Zhang, “Electrochemical corrosion behavior of 15Cr–6Ni–2Mo stainless steel with/without stress under the coexistence of CO2 and H2S,” Int. J. Electrochem. Sci., 13, Is. 7, 6296–6309 (2018); https://doi.org/10.20964/2018.07.59
M. Khoma, V. Vynar, M. Chuchman, and C. Vasyliv, “Corrosion-mechanical failure of pipe steels in hydrogen sulfide environments,” in: Degradation Assessment and Failure Prevention of Pipeline Systems, Springer, Cham (2021), pp. 231–239; https://doi.org/10.1007/978-3-030-58073-5_18
M. S. Khoma, S. A. Korniy, V. A. Vynar, B. M. Datsko, Yu. Ya. Maksishko, O. V. Dykha, and R. L. Bukliv, “Influence of hydrogen sulfide on the carbon-dioxide corrosion and the mechanical characteristics of high-strength pipe steel,” Mater. Sci., 57, No. 6, 805–812 (2022); https://doi.org/10.1007/s11003-022-00610-0
M. M. Ivaniuta (Ed.), Atlas of Oil and Gas Deposits of Ukraine: in 6 volumes [in Ukrainian], Tsentr Yevropy, Lviv (1998). ISBN 966-7022-04-8.
C. Plennevaux, J. Kittel, M. Frégonèse, B. Normand, F. Ropital, F. Grosjean, and T. Cassagne, “Contribution of CO2 on hydrogen evolution and hydrogen permeation in low alloy steels exposed to H2S environment,” Electrochem. Communic., 26, Is. 1, 17–20 (2013); https://doi.org/10.1016/j.elecom.2012.10.010
G. A. Zhang, Y. Zeng, X. P. Guo, F. Jiang, D. Y. Shi, and Z. Y. Chen, “Electrochemical corrosion behavior of carbon steel under dynamic high pressure H2S/CO2 environment,” Corros. Sci., 65, 37–47 (2012); https://doi.org/10.1016/j.corsci.2012.08.007
W. Sun, S. Nesic, and S. Papavinasam, “Kinetics of iron sulfide and mixed iron sulfide/carbonate scale precipitation in CO2/H2S corrosion,” NACE – Int. Corrosion Conference Series, 06644\1–06644\26 (2006).
X. Wen, P. Bai, B. Luo, S. Zheng, and C. Chen, “Review of recent progress in the study of corrosion products of steels in a hydrogen sulphide environment,” Corros. Sci., 139, 124–140 (2018); https://doi.org/10.1016/j.corsci.2018.05.002
NACE Standard SP0110-2010, Wet Gas Internal Corrosion Direct Assessment Methodology for Pipelines, NACE, Houston, TX (2010).
M. Khoma, V. Vynar, C. Vasyliv, M. Chuchman, B. Datsko, V. Ivashkiv, and O. Dykha, “Tribo-corrosion of 17Mn1Si steel in chloride-acetate environments at the different concentrations of hydrogen sulfide,” J. of Bio- and Tribo-Corrosion, 8, Is. 2, art. no. 53 (2022); https://doi.org/10.1007/s40735-022-00655-3
M. S. Khoma, V. R. Ivashkiv, N. B. Ratska, B. M. Datsko, and M. R. Chuchman, “Corrosion-electrochemical properties of 17G1SU steel in chloride-acetate solutions with different concentrations of hydrogen sulfide,” Mater. Sci., 56, No. 4, 544–549 (2021); https://doi.org/10.1007/s11003-021-00462-0
M. S. Khoma, S. A. Holovei, V. R. Ivashkiv, and Kh. B. Vasyliv, “Effect of sulfides on the hydrogen overvoltage and hydrogenation of U8 steel in chloride-hydrogen-sulfide media,” Mater. Sci., 53, No. 6, 761–768 (2018); https://doi.org/10.1007/s11003-018-0133-z
S. K. Dwivedi, and M. Vishwakarma, “Hydrogen embrittlement in different materials: A review,” Int. J. of Hydrogen Energy, 43, Is. 46, 21603–21616 (2018); https://doi.org/10.1016/j.ijhydene.2018.09.201
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Fizyko-Khimichna Mekhanika Materialiv, Vol. 59, No. 2, pp. 80–87, March–April, 2023.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Khoma, M.S., Pokhmurskii, V.I., Chuchman, M.R. et al. Corrosion-Mechanical Properties and Susceptibility to Hydrogenetaion of Pipe Steel in the Presence of Carbon Dioxide Gas and Hydrogen Sulphide in Environment. Mater Sci 59, 205–212 (2023). https://doi.org/10.1007/s11003-024-00764-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11003-024-00764-z