Abstract
Background
Chronic hepatitis-B virus (HBV) infection due to mother-to-child transmission (MTCT) during the perinatal period is an important global health concern. Chile is a low-prevalence country with an increasing migratory inflow from Latin- American countries, with intermediate to high endemic rates of HBV infection, and until 2021, there is no universal maternal screening. This study aimed to evaluate infant outcomes using a risk-based strategy of maternal screening to prevent MTCT of hepatitis B virus (HBV) in a low-prevalence country.
Methods
This prospective study included infants born to HBsAg-positive women detected using a local risk-based strategy. The exposed infants received immunoprophylaxis (IP) and follow-up to evaluate their clinical outcomes and immune responses through post-serological vaccine testing (PSVT) after completing the three- dose schedule of the HBV vaccine.
Results
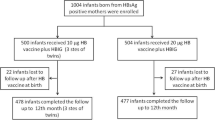
A total of 99 HBsAg-positive mothers were detected. Seventy-six (82%) infants completed the follow-up and had PSVT between 9 and 12 months of age. 55.2% female, the median gestational age was 39 weeks (25–41) and the median birth weight was 3,130g (816–4,400 g). All patients received IP with recombinant HBV vaccine plus hepatitis-B virus immunoglobulin (HBIG) and three doses of the HBV vaccine. There were no cases of HBV infection, and 96% (72) responded to immunization with HBsAg antibodies (anti-HBsAg) >10 UI/ml, with a median level of 799 IU/ml.
Conclusions
A high-risk strategy can be implemented in countries with non-universal screening for VHB. Timely IP plus high-uptake VHB vaccination in infants born to HBsAg-positive mothers was associated with a high immunogenic response and absence of MTCT.
Significance
There is robust evidence that immunoprophylaxis is recommended in all infants born to a HBsAg positive mother like one of different strategies to control VHBtransmission.
AbstractSection What this study adds?In countries with non-universal screening of VHB a risk based strategy could be considered to prevent MTCT. Timely immunoprophylaxis plus high uptake VHB vaccination in infants born to HBsAg positive mothers is associated with a high immunogenic response and no MTCT. PVST should be considered in populations with lower immunogenic response such as infants born with <2,000 g of weight. PVST should be performed 1 or 2 months after the priming vaccine doses, helping to avoid unnecessary revaccination due to expected declining seroprotecting levels through time.



Similar content being viewed by others
References
Alarcon, J., Alarcón, Y., Hering, E., & Buccioni, R. (2008). Curvas antropométricas de recién nacidos chilenos. Revista Chilena De pediatría, 79(4), 364–372. https://doi.org/10.4067/S0370-41062008000400003.
Brown, R. S., McMahon, J., Lok, B. J., Wong, A. S., Ahmed, J. B., Mouchli, A. T., Wang, M. A., Prokop, Z., Murad, L. J., M. H., & Mohammed, K. (2016). Antiviral therapy in chronic hepatitis B viral infection during pregnancy: A systematic review and meta-analysis. Hepatology (Baltimore Md), 63(1), 319–333. https://doi.org/10.1002/hep.28302.
Cheung, K. W., Seto, M. T. Y., & Lao, T. T. (2019). Prevention of perinatal hepatitis B virus transmission. Archives of Gynecology and Obstetrics, 300(2), 251–259. https://doi.org/10.1007/s00404-019-05190-0.
Del Canho, R., Grosheide, P. M., Schalm, S. W., de Vries, R. R., & Heijtink, R. A. (1994). Failure of neonatal hepatitis B vaccination: The role of HBV-DNA levels in hepatitis B carrier mothers and HLA antigens in neonates. Journal of Hepatology, 20(4), 483–486. https://doi.org/10.1016/s0168-8278(05)80494-5.
Espinosa, C. M., & Jhaveri, R. (2018). Update on the management of hepatitis B and C infections in the neonatal period. Seminars in Perinatology, 42(3), 185–190. https://doi.org/10.1053/j.semperi.2018.02.006.
Franco, E., Meleleo, C., Serino, L., Sorbara, D., & Zaratti, L. (2012). Hepatitis A: Epidemiology and prevention in developing countries. World Journal of Hepatology, 4(3), 68–73. https://doi.org/10.4254/wjh.v4.i3.68.
Fuster, F., Peirano, F., Vargas, J. I., Zamora, F. X., López-Lastra, M., Núñez, R., Soza, J., González, K., Estay, D., Barchiesi, B., Fuster, A., López, I., Utrera, N., Landeros, J., Chandía, J., Paredes, A., Reyes, D., Arias, R., Padilla, L., Suárez, H., & Soza, A. (2020). Infectious and non-infectious diseases burden among Haitian immigrants in Chile: A cross-sectional study. Scientific Reports, 10(1), 22275. https://doi.org/10.1038/s41598-020-78970-3.
Health Ministry Chile (2022). Norma Técnica de Prevención de Transmisión Vertical de Hepatitis B 2022 Subsecretaría de Salud Pública, División de Prevención y Control de Enfermedades, Ministerio de Salud, Chile. https://diprece.minsal.cl/wp-content/uploads/2022/08/NORMA-TECNICA-N°-224-DECRETO-N°-55-SSP-2022.pdf [accessed 15 October 2022].
INE, Instituto nacional de estadística (2020). Estimación de personas extranjeras residentes habituales en Chile al 31 de diciembre de 2020. Instituto Nacional de estadísticas. Departamento de Extranjería y Migración. Ministerio del Interior y Seguridad Pública, Gobierno de Chile. https://www.ine.cl/docs/default-source/demografia-y-migracion/publicaciones-y-anuarios/migraci?n-internacional/estimaci?n-poblaci?n-extranjera-en-chile-2018/estimaci?n-poblaci?n-extranjera-en-chile-2020-s%C3%ADntesis.pdf?sfvrsn=5bdc44de_4.
Izquierdo, G., Bustos, S., González, Á., Córdova, L., Riquelme, P., Liendo, F., Villalobos, C., Guajardo, M., Araneda, A., Campoverde, P., Piñera, C., Pino, E., Guerra, C., & Zamora, F. (2019). Cribado De virus de Hepatitis B en mujeres embarazadas: Inmigrantes, y chilenas con conductas de riesgo. Manejo Del Binomio madre-hijo: Plan piloto [Screening of hepatitis B in high-risk Chilean and immigrant pregnant women: Management of mother to child transmission]. Revista chilena de infectología: órgano oficial de la Sociedad Chilena De Infectología, 36(5), 576–584. https://doi.org/10.4067/S0716-10182019000500576.
Kumar, M., Singh, T., & Sinha, S. (2012). Chronic hepatitis B virus infection and pregnancy. Journal of Clinical and Experimental Hepatology, 2(4), 366–381. https://doi.org/10.1016/j.jceh.2012.09.001.
Ma, L., Alla, N. R., Li, X., Mynbaev, O. A., & Shi, Z. (2014). Mother-to-child transmission of HBV: Review of current clinical management and prevention strategies. Reviews in Medical Virology, 24(6), 396–406. https://doi.org/10.1002/rmv.1801.
Pereira, S., Ana, Valenzuela, B., María Teresa, Mora, J., & Vera, L. (2008). Situación actual de la hepatitis B en Chile. Revista Médica De Chile, 136(6), 725–732. https://doi.org/10.4067/S0034-98872008000600006.
Pittaluga, P., Enrica, D. A., Verónica, Mena, N., Patricia, & Corvalán, V., Sergio (2002). Curva de crecimiento intrauterino para prematuros entre 23 a 36 semanas de edad gestacional. Revista Chilena De pediatría, 73(2), 135–141. https://doi.org/10.4067/S0370-41062002000200005.
Schillie, S., Harris, A., Link-Gelles, R., Romero, J., Ward, J., & Nelson, N. (2018). Recommendations of the advisory committee on immunization practices for use of a hepatitis B vaccine with a novel adjuvant. MMWR Morbidity and Mortality Weekly Report, 67(15), 455–458. https://doi.org/10.15585/mmwr.mm6715a5.
The World Bank Data. (n.d) https://data.worldbank.org/country/chile [accessed 13 October 2022].
Tohme, R. A., Andre-Alboth, J., Tejada-Strop, A., Shi, R., Boncy, J., François, J., Domercant, J. W., Griswold, M., Hyppolite, E., Adrien, P., & Kamili, S. (2016). Hepatitis B virus infection among pregnant women in Haiti: A cross-sectional serosurvey. Journal of Clinical Virology: The Official Publication of the Pan American Society for Clinical Virology, 76, 66–71. https://doi.org/10.1016/j.jcv.2016.01.012.
UK Health Security Agency (2013). Hepatitis B: the green book, Chap. 18. https://www.gov.uk/government/publications/hepatitis-b-the-green-book-chapter-18 [accessed December 20th, 2022].
Valdés, R., Sepúlveda, E., Candia, M. A. P, P., & Lattes, A., K (2010). Hepatitis aguda viral durante El embarazo [Acute viral hepatitis during pregnancy] (Vol. 27, pp. 505–512). órgano oficial de la Sociedad Chilena de Infectologia. 6Revista chilena de infectología:.
Vincent, J. P., Nyamasege, C., Wang, S., Madec, Y., & Shimakawa, Y. (2023). Prevalence of hepatitis B, C, and D virus infection in Haiti: A systematic review and meta-analysis. Frontiers in Public Health, 10, 1099571. https://doi.org/10.3389/fpubh.2022.1099571.
Waitz, M., Hopfner, R., Hummler, H. D., & Heininger, U. (2015). Hepatitis B Postexposure Prophylaxis in Preterm and low-birth-weight infants. AJP Reports, 5(1), e67–e72. https://doi.org/10.1055/s-0035-1547329.
Wang, J., Zhu, Q., & Zhang, X. (2002). Effect of delivery mode on maternal-infant transmission of hepatitis B virus by immunoprophylaxis. Chinese Medical Journal, 115(10), 1510–1512.
WHO (2016). World Health Organization. Global Health Sector Strategies on Viral Hepatitis 2016–2021. Available from: http://apps.who.int/gb/ebwha/pdf_files/WHA69/A69_32-en.pdf?ua=1.
WHO (2017). World Health Organization, Global Hepatitis Report 2017. Geneva: Licence: CC BY-NC-SA 3.0 IGO. https://www.who.int/publications/i/item/9789241565455.
WHO (2020a). World Health Organization. https://www.who.int/campaigns/world-hepatitis-day/2020 [accessed 12 December 2022].
WHO (2020b). Prevention of mother-to-child transmission of hepatitis B virus: guidelines on antiviral prophylaxis in pregnancy. Geneva: World Health Organization; 2020. Licence: CC BY-NC-SA 3.0 IGO. https://apps.who.int/iris/bitstream/handle/10665/333391/9789240002708-eng.pdf.
Funding
No source of funding.
Author information
Authors and Affiliations
Contributions
G.I., R.V., L.C., and E.P. contributed to the design and implementation of the research; G.I., C.I., C.P., P.L., and R.V. analyzed the results and wrote the manuscript. G.I. conceived and supervised the project.
Corresponding author
Ethics declarations
Ethical Approval
This study was approved by the Ethics Committee of the South Metropolitan Health Department for Clinical Investigation in Humans, Santiago, Chile.
Consent to Participate
After obtaining signed informed consent, the data were collected and confidentially managed by the research team.
Conflict of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Copiz, G.I., Ibañez, C., Piñera, C. et al. Outcome of Infants Born to Women with Chronic Hepatitis B: A Local Risk-Based Strategy in a Low Prevalence Country. Matern Child Health J 28, 767–774 (2024). https://doi.org/10.1007/s10995-024-03909-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10995-024-03909-3