Abstract
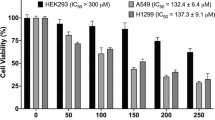
Lung cancer is a prevalent and life-threatening disease that requires potent and selective antitumor agents for effective treatment. Antimicrobial peptides (AMPs) have gained popularity as potential antitumor agents due to their reported ability to selectively target tumor cells. In this study, we investigated the anti-tumor activity of Retrocyclin-3, a synthetic AMP known for its beta-sheet structure. Our in vitro experiments focused on evaluating the cytotoxic effects of Retrocyclin-3 on A549 lung cancer cells. The results indicated that Retrocyclin-3 induced apoptotic cell death in the cancer cells, as confirmed by fluorescent staining techniques. Flow cytometry analysis also revealed that Retrocyclin-3 caused cell cycle arrest by inducing apoptosis. These findings suggest that Retrocyclin-3 preferentially targets cancer cells, causing necrosis, apoptosis, nucleic effect, and DNA damage. Based on these results, Retrocyclin-3 has promising potential as a therapeutic candidate for further development as an anti-cancer agent.





Similar content being viewed by others
References
Behera D, Balamugesh T (2004) Lung cancer in India. Indian J Chest Dis Allied Sci 46:269–281
Broggio J, Bannister N (2016) Cancer survival by stage at diagnosis for England (. experimental statistics) - Office for National Statistics, UK
Chen C, Chen Y, Yang C et al (2015) High selective performance of designed antibacterial and anticancer peptide amphiphiles. ACS Appl Mater Interfaces 7:17346–17355. https://doi.org/10.1021/ACSAMI.5B04547
Dennison SR, Harris F, Bhatt T et al (2009) The effect of C-terminal amidation on the efficacy and selectivity of antimicrobial and anticancer peptides. Mol Cell Biochem 332:43–50. https://doi.org/10.1007/S11010-009-0172-8
Deslouches B, Di YP, Deslouches B, Peter Di Y (2017) Antimicrobial peptides with selective antitumor mechanisms: prospect for anticancer applications. Oncotarget 8:46635–46651. https://doi.org/10.18632/ONCOTARGET.16743
DR G, JC R (1998) Mitochondria and apoptosis. Science 281:118. https://doi.org/10.1126/SCIENCE.281.5381.1309
Goldstraw P, Chansky K, Crowley J et al (2016) The IASLC Lung Cancer Staging Project: proposals for revision of the TNM Stage Groupings in the Forthcoming (Eighth) Edition of the TNM classification for Lung Cancer. J Thorac Oncol 11:39–51. https://doi.org/10.1016/J.JTHO.2015.09.009
Gross A, McDonnell JM, Korsmeyer SJ (1999) BCL-2 family members and the mitochondria in apoptosis. Genes Dev 13:1899–1911. https://doi.org/10.1101/GAD.13.15.1899
Herbst RS, Heymach JV, Lippman SM (2008) Lung cancer. N Engl J Med 359:1367–1380. https://doi.org/10.1056/NEJMRA0802714
Hoskin DW, Ramamoorthy A (2008) Studies on anticancer activities of antimicrobial peptides. Biochim Biophys Acta 1778:357–375. https://doi.org/10.1016/J.BBAMEM.2007.11.008
Kasibhatla S, Amarante-Mendes GP, Finucane D et al (2006) Acridine Orange/Ethidium bromide (AO/EB) staining to detect apoptosis. https://doi.org/10.1101/PDB.PROT4493. CSH Protoc 2006:pdb.prot4493
Knight SB, Crosbie PA, Balata H et al (2017) Progress and prospects of early detection in lung cancer. Open Biol 7. https://doi.org/10.1098/RSOB.170070
Latt SA, Wohlleb JC (1975) Optical studies of the interaction of 33258 Hoechst with DNA, chromatin, and metaphase chromosomes. Chromosoma 52(4):297–316
Lemjabbar-Alaoui H, Hassan OUI, Yang YW, Buchanan P (2015) Lung cancer: Biology and treatment options. Biochim Biophys Acta 1856:189–210. https://doi.org/10.1016/J.BBCAN.2015.08.002
Mishra SK, Kumar BSH, Khushu S et al (2017) Early monitoring and quantitative evaluation of macrophage infiltration after experimental traumatic brain injury: a magnetic resonance imaging and flow cytometric analysis. Mol Cell Neurosci 78:25–34. https://doi.org/10.1016/J.MCN.2016.11.008
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63. https://doi.org/10.1016/0022-1759(83)90303-4
Ohsaki Y, Gazdar A, Chen H, Johnson B (1992) Antitumor activity of magainin analogues against human lung cancer cell lines. Cancer Res 52(13), 3534–3538
Rajendiran V, Karthik R, Palaniandavar M et al (2007) Mixed-ligand copper(II)-phenolate complexes: effect of coligand on enhanced DNA and protein binding, DNA cleavage, and anticancer activity. Inorg Chem 46:8208–8221. https://doi.org/10.1021/IC700755P
Reers M, Smiley ST, Mottola-Hartshorn C et al (1995) Mitochondrial membrane potential monitored by JC-1 dye. Methods Enzymol 260:406–414. https://doi.org/10.1016/0076-6879(95)60154-6
Schwartzberg L, Kim ES, Liu D, Schrag D (2017) Precision Oncology: who, how, what, when, and when not? Am soc clin Oncol Educ book. Am Soc Clin Oncol Annu Meet 37:160–169. https://doi.org/10.1200/EDBK_174176
Schweizer F (2009) Cationic amphiphilic peptides with cancer-selective toxicity. Eur J Pharmacol 625:190–194. https://doi.org/10.1016/J.EJPHAR.2009.08.043
Shah R, Sabanathan S, Richardson J et al (1996) Results of surgical treatment of stage I and II lung cancer. J Cardiovasc Surg (Torino) 37:169–172. https://doi.org/10.1016/s0169-5002(97)89544-x
Sher T, Dy GK, Adjei AA (2008) Small cell lung cancer. Mayo Clin Proc 83:355–367. https://doi.org/10.4065/83.3.355
Siegel RL, Miller KD, Goding Sauer A et al (2020) Colorectal cancer statistics, 2020. CA Cancer J Clin 70:145–164. https://doi.org/10.3322/CAAC.21601
Spector DL, Goldman RD (2006) Basic Methods in Microscopy: Protocols and Concepts from Cells: A Laboratory Manual
Tice RR, Agurell E, Anderson D et al (2000) Single cell Gel/Comet Assay: guidelines for in Vitro and in vivo genetic toxicology testing. Environ Mol Mutagen 35:206–221
Wu D, Gao Y, Qi Y et al (2014) Peptide-based cancer therapy: opportunity and challenge. Cancer Lett 351:13–22. https://doi.org/10.1016/J.CANLET.2014.05.002
Acknowledgements
This work was supported by Indian Council of Medical Research (ISRM/11(65)/2019) and University Grants Commission, National Fellowship for SC, Award Letter No.: F1-17.1/2017-18/RGNF-2017-18-SC-TAM-45554. Dated: 16/08/2017.
Author information
Authors and Affiliations
Contributions
SKM, KN: performed experiments and drafted the whole manuscript, KB: performed statically analysis. SP: participated in manuscript written and study design. LS: conceived the original idea of this paper and also helped in writing part of this paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors have nothing to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Marimuthu, S.K., Nagarajan, K., Balakrishnan, K. et al. Potential Anticancer Activity of Retrocyclin-3: An in vitro Study. Int J Pept Res Ther 29, 58 (2023). https://doi.org/10.1007/s10989-023-10533-2
Accepted:
Published:
DOI: https://doi.org/10.1007/s10989-023-10533-2