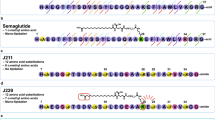
Abstract
The rapid degradation of native glucagon-like peptide 1 (GLP-1) by dipeptidyl peptidase-IV (DPP-IV) has advanced new approaches to the generation of degradation-resistant GLP-1 analogs. Chemical coupling of GLP-1 analog to HSA is innovatively achieved by solid-phase peptide synthesis in this study and shows prolonged glucose-lowering ability in vivo. GLP-1(7–37)(Ala8Aib)-Cys-HSA was constructed by solid-phase peptide synthesis through two levels of modification: mutation of Ala8 to aminoisobutyric acid (Aib) to decrease DPP-IV degradation, and conjugation to large serum protein HSA by chemical modification to decrease renal filtration. Glucose tolerance test and insulin secretion assay were performed to examine the biological activity of GLP-1(7–37)(Ala8Aib)-Cys-HSA in vivo in the present research. Long-lasting glucose-lowering and insulin-releasing effects were evaluated up to 4 weeks in T2DM rats. GLP-1(7–37)(Ala8Aib)-Cys-HSA lowered blood glucose in normal mice and T2DM rats. Twice administration of GLP-1(7–37)(Ala8Aib)-Cys-HSA to T2DM rats daily significantly reduced glycemic excursion following IP glucose challenge (P < 0.01 to 0.05) and greatly increased insulin secretion during the 4-week study period. These findings demonstrate that the albumin-conjugated GLP-1 analog mimics the function of native GLP-1 with prolonged activity.






Similar content being viewed by others
References
Atherton E, Fox H, Harkiss D, Logan CJ, Sheppard RC, Williams BJ (1978) A mild procedure for solid phase peptide synthesis: use of fluorenylmethoxycarbonylamino-acids. J Chem Soc Chem Commun.https://doi.org/10.1039/C39780000537
Briones M, Bajaj M (2006) Exenatide: a GLP-1 receptor agonist as novel therapy for Type 2 diabetes mellitus. Expert Opin Pharmacother 7:1055–1064
Chang CD, Meienhofer J (1978) Solid-phase peptide synthesis using mild base cleavage of N alpha-fluorenylmethyloxycarbonylamino acids, exemplified by a synthesis of dihydrosomatostatin. Int J Pept Protein Res 11:246–249. https://doi.org/10.1111/j.1399-3011.1978.tb02845.x
Davis PN, Ndefo UA, Oliver A, Payton E (2015) Albiglutide: a once-weekly glucagon-like peptide-1 receptor agonist for type 2 diabetes mellitus. Am J Health Syst Pharm 72:1097
Deacon CF, Plamboeck A, Mã¸Ller S, Holst JJ (2002) GLP-1-(9–36) amide reduces blood glucose in anesthetized pigs by a mechanism that does not involve insulin secretion. Am J Physiol Endocrinol Metab 282:E873
Drucker DJ (1998) Diabetes. Glucagon-like peptides 47:159–169
Drucker DJ, Dritselis A, Kirkpatrick P (2010) Liraglutide. Nat Rev Drug Discov 9:267–268
Duman JG, Miele RG, Liang H, Grella DK, Sim KL, Castellino FJ, Bretthauer RK (1998) O-Mannosylation of Pichia pastoris cellular and recombinant proteins. Biotechnol Appl Biochem 28:39–45. https://doi.org/10.1111/j.1470-8744.1998.tb00510.x
Fields GB, Noble RL (1990) Solid phase peptide synthesis utilizing 9-fluorenylmethoxycarbonyl amino acids International. Journal of Peptide Protein Research 35:161–214. https://doi.org/10.1111/j.1399-3011.1990.tb00939.x
Grassetti DR Jr (1969) The use of 2,2'-dithiobis-(5-nitropyridine) as a selective reagent for the detection of thiols. J Chromatogr 41:121
Hedrington MS, Davis SN (2019) Oral semaglutide for the treatment of type 2 diabetes. Expert Opin Pharmacother 20:133–141. doi:https://doi.org/10.1080/14656566.2018.1552258
Holst JJ (1997) Enteroglucagon. Annu Rev Physiol 59(1):257–271
Holst JJ (1999) Glucagon-like peptide 1 (GLP-1): an intestinal hormone, signalling nutritional abundance&nbsp;with an unusual therapeutic potential. Trends Endocrinol Metab 10:229–235
Huang XL, He Y, Ji LL, Wang KY, Wang YL, Chen DF, Geng Y, OuYang P, Lai WM (2017) Hepatoprotective potential of isoquercitrin against type 2 diabetes-induced hepatic injury in rats. Oncotarget 8:101545–101559
Jaradat DMM (2018) Thirteen decades of peptide synthesis: key developments in solid phase peptide synthesis and amide bond formation utilized in peptide ligation. Amino Acids 50:39–68. https://doi.org/10.1007/s00726-017-2516-0
Jaradat DMM, Saleh KKY, Za’arir BHM, Arafat T, Alzoubi KH, Al-Taweel SA, Mallah E, Haddad MA, Haimur BAK (2019) Solid-phase synthesis and antibacterial activity of an artificial cyclic peptide containing two disulfide bridges. Int J Pept Res Ther 25:1095–1102. https://doi.org/10.1007/s10989-018-9757-y
Kieffer TJ, Mcintosh CH, Pederson RA (1995) Degradation of glucose-dependent insulinotropic polypeptide and truncated glucagon-like peptide 1 in vitro and in vivo by dipeptidyl peptidase IV. Endocrinology 136:3585
Kim JG, Baggio LL, Bridon DP, Castaigne JP, Robitaille MF, Jetté L, Benquet C, Drucker DJ (2003) Development and characterization of a glucagon-like peptide 1-albumin conjugate: the ability to activate the glucagon-like peptide 1 receptor in vivo. Diabetes 52:751–759. https://doi.org/10.2337/diabetes.52.3.751
Li Y, Zheng X, Tang L, Xu W, Gong M (2011) GLP-1 analogs containing disulfide bond exhibited prolonged half-life in vivo than GLP-1. Peptides 32:1303
Manandhar B, Ahn JM (2015) Glucagon-like peptide-1 (GLP-1) analogs: recent advances, new possibilities, and therapeutic implications. J Med Chem 58:1020–1037
Mentlein R, Gallwitz B, Schmidt WE (1993) Dipeptidyl-peptidase IV hydrolyses gastric inhibitory polypeptide, glucagon-like peptide-1(7–36)amide, peptide histidine methionine and is responsible for their degradation in human serum. FEBS J 214:829
Nauck MA, Holst JJ, Willms B, Schmiegel W (1997) Glucagon-like peptide 1 (GLP-1) as a new therapeutic approach for type 2-diabetes. Exp Clin Endocrinol Diabetes 105:187–195
O'Halloran DJ, Nikou GC, Kreymann B, Ghatei MA, Bloom SR (1990) Glucagon-like peptide-1 (7–36)-NH2: a physiological inhibitor of gastric acid secretion in man. J Endocrinol 126:169–173
Parhofer KG (2007) Glucagon-like peptide 1 (GLP-1). MMW Fortschritte Der Medizin 149:41
Peters T Jr (1985) Serum albumin. Adv Protein Chem 37:161–245
Peng L, Turesky RJ (2011) Mass spectrometric characterization of 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine N-oxidized metabolites bound at Cys34 of human serum albumin. Chem Res Toxicol 24:2004–2017
Postma TM, Albericio F (2014) Disulfide formation strategies in peptide synthesis. Eur J Org Chem 2014:3519–3530. https://doi.org/10.1002/ejoc.201402149
Scheen AJ (2008) New therapeutic approaches in type 2 diabetes. Acta Clin Belg 63:402–407
Scheen AJ (2017) Dulaglutide for the treatment of type 2 diabetes. Expert Opin Biol Ther 17:485–496. https://doi.org/10.1080/14712598.2017.1296131
Schroll AL, Hondal RJ, Flemer S Jr (2012) 2,2′-Dithiobis(5-nitropyridine) (DTNP) as an effective and gentle deprotectant for common cysteine protecting groups. J Pept Sci 18:1
Stoffers DA, Kieffer TJ, Hussain MA, Drucker DJ, Bonnerweir S, Habener JF, Egan JM (2000) Insulinotropic glucagon-like peptide 1 agonists stimulate expression of homeodomain protein IDX-1 and increase islet size in mouse pancreas. Diabetes 49:741–748
Straub SG, Sharp GW (2002) Glucose-stimulated signaling pathways in biphasic insulin secretion. Diabetes Metab Res Rev 18:451–463
Turton MD, O’Shea D, Gunn I, Beak SA, Edwards CM, Meeran K, Choi SJ, Taylor GM, Heath MM, Lambert PD, Wilding JP, Smith DM, Ghatei MA, Herbert J, Bloom SR (1996) A role for glucagon-like peptide-1 in the central regulation of feeding. Nature 379:69–72
Vilsbøll T (2009) The effects of glucagon-like peptide-1 on the beta cell. Diabetes Obes Metab 11:11–18
Vishal G (2013) Glucagon-like peptide-1 analogues: an overview. Indian J Endocrinol Metab 17:413
Wettergren A, Schjoldager B, Mortensen PE, Myhre J, Christiansen J, Holst JJ (1993) Truncated GLP-1 (proglucagon 78–107-amide) inhibits gastric and pancreatic functions in man. Dig Dis Sci 38:665–673. https://doi.org/10.1007/bf01316798
Xu G, Stoffers DA, Habener JF, Bonner-Weir S (1999) Exendin-4 stimulates both beta-cell replication and neogenesis, resulting in increased beta-cell mass and improved glucose tolerance in diabetic rats. Diabetes 48:2270–2276
Yamato E, Noma Y, Tahara Y, Ikegami H, Yamamoto Y, Cha T, Yoneda H, Ogihara T, Ohboshi C, Hirota M, Shima K (1990) Suppression of synthesis and release of glucagon by glucagon-like peptide-1 (7–36 amide) without affect on mRNA level in isolated rat islets. Biochem Biophys Res Commun 167:431–437
Funding
This research was funded by the National Natural Science Foundation of China (No. 31571277, No. 31900906), Hunan Natural Science Foundation (No. 2018JJ2150, No. 17C0709, No. 2019JJ50207) and the Scientific Research Key Fund of Hunan Provincial Education Department (No. 18A318).
Author information
Authors and Affiliations
Contributions
YL, DN and YZ conceived and designed the experiment; QW and Y.Z performed the experiments. ZW and QW analyzed the data; YL, ZW and YZ wrote and revised the paper. All authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare no conflict of interest.
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, Y., Wang, Q., Nie, D. et al. Novel Modified GLP-1 Derivatives with Prolonged Glucose-Lowering Ability In Vivo. Int J Pept Res Ther 26, 1939–1947 (2020). https://doi.org/10.1007/s10989-019-09991-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-019-09991-4