Abstract
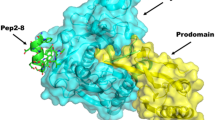
Wnt signaling pathway plays a major role in the regulation of cell proliferation, migration, tissue homeostasis, tumor progression and cancer. This pathway can be antagonized by different proteins such as DKK proteins, which disrupt the initiatory complex (Frizzled–LRP6 complex). Therefore, interruption of its formation could be a promising strategy for the design of Low-density lipoprotein receptor-Related Protein 6 (LRP6) inhibitors. A computational study was conducted in order to assist in the design of inhibitory peptides against LRP6 as co-receptor of frizzled. Twelve fragments as peptide derivatives of natural ligand of LRP6 receptor (DKK1) were designed using the information from the analysis of the DKK1_C/LRP6 complex, hot spot residues and the secondary structure. These fragments were based on cys2 domain of DKK1. The designed peptides were energy minimized by molecular dynamics simulations in the presence and absence of LRP6 receptor and their binding affinities were investigated via molecular docking using ClusPro, HADDOCK and PRODIGY webservers. Finally, the stability and free energy of binding in peptides were calculated by FoldX software. The results showed that four designed peptides had the highest affinity (the interaction energy: −10.2867, −10.1388, −7.94339 and −7.57536 kcal/mol) to interact with the receptor which showed the most interacting residues and the lowest free energy of binding. Also, the RMSD, RMSF and RoG of the protein–peptide complex exhibited less structural fluctuations which can be linked to the stability of peptides associated to the receptor. These peptides may be considered as candidates for inhibiting Wnt signaling pathway through LRP6 receptor.





Similar content being viewed by others
References
Abraham MJ, van der Spoel D, Lindahl E, Hess B, GROMACS Development team (2015) GROMACS User Manual version 5.0.6
Acar M, Çora T, Toy H, Acar H (2012) Analysis of promoter methylation of Dickkopf1 (DKK1) gene in breast cancer. Turk J Med Sci 42:1379–1387
Ahn VE, Chu ML, Choi HJ, Tran D, Abo A, Weis WI (2011) Structural basis of Wnt signaling inhibition by Dickkopf binding to LRP5/6. Dev Cell 21:862–873. doi:10.1016/j.devcel.2011.09.003
Akhoon BA, Gupta SK, Verma V, Dhaliwal G, Srivastava M, Ahmad RF (2010) In silico designing and optimization of anti-breast cancer antibody mimetic oligopeptide targeting HER-2 in women. J Mol Graph Model 28:664–669. doi:10.1016/j.jmgm.2010.01.002
Bafico A, Liu G, Yaniv A, Gazit A, Aaronson SA (2001) Novel mechanism of Wnt signalling inhibition mediated by Dickkopf-1 interaction with LRP6/Arrow. Nat Cell Biol 3:683–686. doi:10.1038/35083081
Bao J, Zheng JJ, Wu D (2013) The structural basis of DKK-mediated inhibition of Wnt/LRP signaling. Sci Signal 5(224):22. doi:10.1126/scisignal.2003028
Berendsen HJC, van der Spoel D, van Drunen R (1995) GROMACS: a message-passing parallel molecular dynamics implementation. Comput Phys Commun 91:43–56
Binnerts ME et al (2009) The first propeller domain of LRP6 regulates sensitivity to DKK1. Mol Biol Cell 20:3552–3560. doi:10.1091/mbc.E08-12-1252
Bourhis E et al (2011) Wnt antagonists bind through a short peptide to the first beta-propeller domain of LRP5/6. Structure 19:1433–1442. doi:10.1016/j.str.2011.07.005
Chen S et al (2011) Structural and functional studies of LRP6 ectodomain reveal a platform for Wnt signaling. Dev Cell 21:848–861. doi.10.1016/j.devcel.2011.09.007
Cheng Z, Biechele T, Wei Z, Morrone S, Moon RT, Wang L, Xu W (2011) Crystal structures of the extracellular domain of LRP6 and its complex with DKK1. Nat Struct Mol Biol 18:1204–1210. doi:10.1038/nsmb.2139
Comeau SR, Gatchell DW, Vajda S, Camacho CJ (2004a) ClusPro: a fully automated algorithm for protein–protein docking. Nucleic Acids Res 32. doi:10.1093/nar/gkh354
Comeau SR, Gatchell DW, Vajda S, Camacho CJ (2004b) ClusPro: a fully automated algorithm for protein–protein docking. Nucleic Acids Res 32:W96–W99. doi:10.1093/nar/gkh354
Dahmen RP et al (2001) Deletions of AXIN1, a component of the WNT/wingless pathway, in sporadic medulloblastomas. Cancer Res 61:7039–7043
DeLano WL (2002) The PyMOL Molecular Graphics System. DeLano Scientific, Palo Alto, CA
Dominguez C, Boelens R, Bonvin AM (2003) HADDOCK: a protein–protein docking approach based on biochemical or biophysical information. J Am Chem Soc 125:1731–1737. doi:10.1021/ja026939x
Fatima S, Lee NP, Luk JM (2011) Dickkopfs and Wnt/β-catenin signalling in liver cancer World J Clin Oncol 10; 2(8):311–325
Giles RH, van Es JH, Clevers H (2003) Caught up in a Wnt storm: Wnt signaling in cancer. Biochim Biophys Acta 1653:1–24
Glinka A WW, Delius H, Monaghan AP, Blumenstock C, Niehrs C (1998) Dickkopf-1 is a member of a new family of secreted proteins and functions in head induction. Nature 391:357–362
Gregory CA, Perry AS, Reyes E, Conley A, Gunn WG, Prockop DJ (2005) Dkk-1-derived synthetic peptides and lithium chloride for the control and recovery of adult stem cells from bone marrow. J Biol Chem 280:2309–2323. doi:10.1074/jbc.M406275200
He X, Semenov M, Tamai K, Zeng X (2004) LDL receptor-related proteins 5 and 6 in Wnt/beta-catenin signaling: arrows point the way. Development 131:1663–1677 doi:10.1242/dev.01117
Heinz DW, Baase WA, Dahlquist FW, Matthews BW (1993) How amino-acid insertions are allowed in an alpha-helix of T4 lysozyme. Nature 361:561–564. doi:10.1038/361561a0
Jorgensen WL, Chandrasekhar J, Madura JD, Impey RW, Klein ML (1983) Comparison of simple potential functions for simulating liquid water. J Chem Phys 79:926–935 doi. doi:10.1063/1.445869
Kato M et al (2002) Cbfa1-independent decrease in osteoblast proliferation, osteopenia, and persistent embryonic eye vascularization in mice deficient in Lrp5, a Wnt coreceptor. J Cell Biol 157:303–314. doi:10.1083/jcb.200201089
Khan Z, Vijayakumar S, de la Torre TV, Rotolo S, Bafico A (2007) Analysis of endogenous LRP6 function reveals a novel feedback mechanism by which Wnt negatively regulates its receptor. Mol Cell Biol 27:7291–7301. doi:10.1128/MCB.00773-07
Kongkham PN, Northcott PA, Croul SE, Smith CA, Taylor MD, Rutka JT (2010) The SFRP family of WNT inhibitors function as novel tumor suppressor genes epigenetically silenced in medulloblastoma Oncogene 29:3017–3024 doi:10.1038/onc.2010.32
Kortemme T, Baker D (2002) A simple physical model for binding energy hot spots in protein–protein complexes. Proc Natl Acad Sci USA 99:14116–14121. doi:10.1073/pnas.202485799
Kortemme T, Kim DE, Baker D (2004) Computational alanine scanning of protein–protein interfaces. Sci STKE 2004(219):pl2. doi:10.1126/stke.2192004pl2
Kozakov D, Brenke R, Comeau SR, Vajda S (2006) PIPER: an FFT-based protein docking program with pairwise potentials. Proteins 65:392–406. doi:10.1002/prot.21117
Kozakov D, Beglov D, Bohnuud T, Mottarella SE, Xia B, Hall DR, Vajda S (2013) How good is automated protein docking? Proteins 81:2159–2166. doi:10.1002/prot.24403
Laskowski RA, Swindells MB (2011) LigPlot+: multiple ligand–protein interaction diagrams for drug discovery. J Chem Inf Model 51:2778–2786. doi:10.1021/ci200227u
Laskowski RA, MacArthur MW, Moss DS, Thornton JM (1993) PROCHECK: a program to check the stereochemical quality of protein structures. J Appl Crystallogr 26(2):283–291
Li Y, Lu W, He X, Schwartz AL, Bu G (2004) LRP6 expression promotes cancer cell proliferation and tumorigenesis by altering beta-catenin subcellular distribution. Oncogene 23:9129–9135. doi:10.1038/sj.onc.1208123
Lindorff-Larsen K, Piana S, Palmo K, Maragakis P, Klepeis JL, Dror RO, Shaw DE (2010) Improved side-chain torsion potentials for the Amber ff99SB protein force field. Proteins 78:1950–1958. doi:10.1002/prot.22711
Luthy R, Bowie JU, Eisenberg D (1992) Assessment of protein models with three-dimensional profiles. Nature 356:83–85. doi:10.1038/356083a0
Ma B, Elkayam T, Wolfson H, Nussinov R (2003) Protein–protein interactions: structurally conserved residues distinguish between binding sites and exposed protein surfaces. Proc Natl Acad Sci USA 100:5772–5777. doi:10.1073/pnas.1030237100
MacDonald BT, Tamai K, He X (2009) Wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev Cell 17:9–26. doi:10.1016/j.devcel.2009.06.016
MacDonald BT, Semenov MV, Huang H, He X (2011) Dissecting molecular differences between Wnt coreceptors LRP5 and LRP6. PLoS ONE 6:e23537. doi:10.1371/journal.pone.0023537
Moal IH, Agius R, Bates PA (2011) Protein–protein binding affinity prediction on a diverse set of structures. Bioinformatics 27:3002–3009. doi:10.1093/bioinformatics/btr513
Moal IH, Jimenez-Garcia B, Fernandez-Recio J (2015) CCharPPI web server: computational characterization of protein–protein interactions from structure. Bioinformatics 31:123–125. doi:10.1093/bioinformatics/btu594
Moreira IS, Fernandes PA, Ramos MJ (2006) Unraveling the importance of protein–protein interaction: application of a computational alanine-scanning mutagenesis to the study of the IgG1 streptococcal protein G (C2 fragment) complex. J Phys Chem B 110:10962–10969. doi:10.1021/jp054760d
Morin PJ, Sparks AB, Korinek V, Barker N, Clevers H, Vogelstein B, Kinzler KW (1997) Activation of beta-catenin-Tcf signaling in colon cancer by mutations in beta-catenin or APC. Science 275:1787–1790
Moult J (2005) A decade of CASP: progress, bottlenecks and prognosis in protein structure prediction. Curr Opin Struct Biol 15:285–289. doi:10.1016/j.sbi.2005.05.011
Pinson KI, Brennan J, Monkley S, Avery BJ, Skarnes WC (2000) An LDL-receptor-related protein mediates Wnt signalling in mice. Nature 407:535–538. doi:10.1038/35035124
Rubinfeld B, Robbins P, El-Gamil M, Albert I, Porfiri E, Polakis P (1997) Stabilization of beta-catenin by genetic defects in melanoma cell lines. Science 275:1790–1792
Schneider WJ, Nimpf J (2003) LDL receptor relatives at the crossroad of endocytosis and signaling. Cell Mol Life Sci 60:892–903. doi:10.1007/s00018-003-2183-Z
Schymkowitz J, Borg J, Stricher F, Nys R, Rousseau F, Serrano L (2005) The FoldX web server: an online force field. Nucleic Acids Res 33:W382–W388. doi:10.1093/nar/gki387
Sharma A et al. (2013) Computational approach for designing tumor homing peptides. Sci Rep 3:1607 doi:10.1038/srep01607
Swanson JAaJ (2013) Advances in the prediction of protein–peptide binding affinities: implications for peptide-based drug discovery. Chem Biol Drug Des 81:50–60
Takagi J, Yang Y, Liu JH, Wang JH, Springer TA (2003) Complex between nidogen and laminin fragments reveals a paradigmatic beta-propeller interface. Nature 424:969–974. doi:10.1038/nature01873
Tamai K et al (2000) LDL-receptor-related proteins in Wnt signal transduction. Nature 407:530–535. doi:10.1038/35035117
Vangone A, Bonvin AM (2015) Contacts-based prediction of binding affinity in protein–protein complexes. Elife 4:e07454 doi:10.7554/eLife.07454
Wang K et al (2008) Characterization of the Kremen-binding site on Dkk1 and elucidation of the role of Kremen in Dkk-mediated Wnt antagonism. J Biol Chem 283:23371–23375. doi:10.1074/jbc.M802376200
Wu G, Han K, Lv F (2013) Use of fast conformational sampling to improve the characterization of VEGF A-peptide interactions. J Theor Biol 317:293–300. doi:10.1016/j.jtbi.2012.10.021
Xiao YF, Jie MM, Li BS, Hu CJ, Xie R, Tang B, Yang SM (2015) Peptide-based treatment: a promising cancer therapy. J Immunol Res 2015:761820. doi:10.1155/2015/761820
Xue LC, Rodrigues JP, Kastritis PL, Bonvin AM, Vangone A (2016) PRODIGY: a web server for predicting the binding affinity of protein–protein complexes. Bioinformatics. doi:10.1093/bioinformatics/btw514
Yang ZR, Dong WG, Lei XF, Liu M, Liu QS (2012) Overexpression of Dickkopf-3 induces apoptosis through mitochondrial pathway in human colon cancer. World J Gastroenterol 18:1590–1601. doi:10.3748/wjg.v18.i14.1590
Zhang Y et al (2004) The LRP5 high-bone-mass G171V mutation disrupts LRP5 interaction with Mesd. Mol Cell Biol 24:4677–4684. doi:10.1128/MCB.24.11.4677-4684.2004
Acknowledgements
This work was funded as Ph.D. thesis project by Pasteur Institute of Iran.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
All authors declare that there is no conflict of interest in this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rismani, E., Rahimi, H., Arab, S.S. et al. Computationally Design of Inhibitory Peptides Against Wnt Signaling Pathway: In Silico Insight on Complex of DKK1 and LRP6. Int J Pept Res Ther 24, 49–60 (2018). https://doi.org/10.1007/s10989-017-9589-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-017-9589-1