Abstract
Context
Agricultural intensification and land-use patterns impact biodiversity and reduce ecosystem functions. Organic farming is often seen as a more biodiversity friendly way of farming. However, not all organic farming is equally supportive for biodiversity, structural diversity of the farms has a great impact and the often high intertwinement of organic and conventional farmland causes spill-over problems.
Objective
On organic farmland in central Italy, we analysed butterfly diversity and community structures in different land-use categories across a heterogeneous landscape. We aim to analyse the impact of the different land-use structures such as hedges and forest edges on this farmland, as well as the influence of adjoining conventionally farmed areas.
Methods
We studied butterflies on organic farmland including olive and hazelnut plantations, as well as meadows and fallow land, surrounded by hedges and forests. Hereby we considered the effects of land-use on the studied site and the adjoining land, e.g., semi-natural habitats (as hedges or forest edges), organic or conventional farmland. We counted butterflies along line transects and recorded habitat parameters for each transect. All encountered butterflies were classified according their ecological demands, behaviour, and life-history.
Results
We found highest mean butterfly species richness in fallow land and lowest in hazelnut plantations. Species community structures differed hardly between plantations and meadows, but considerably compared to fallow land. Butterfly abundances are reduced for olive plantations adjacent to agricultural fields treated with pesticides, compared to such surrounded by organic fields. Habitat para-meters such as the number of flowering plant species and shade impact butterfly diversity and abundance. Forest edges producing considerable shadow to the transects had a significantly negative effect on diversity and abundance, while the influence of hedges was positive.
Conclusions
Based on our findings, we conclude that also organic farmland has different degrees of biodiversity depending on a variety of land-use pattern. In particular, habitat heterogeneity and a high diversity of flowers positively influence butterfly diversity. The intensive mixing of organic and conventional agriculture must be seen critical for conservation as negative effects might spill over to organic fields, but might be compensable by landscape structures like hedges.
Similar content being viewed by others
Introduction
Biodiversity has declined significantly over the last decades (Dirzo et al. 2014). About 40% of all insect species worldwide are classified as threatened or are close to extinction (Sanchez-Bayo and Wyckhuys 2019). Recent studies documented severe decrease of insect diversity and abundance across major parts of Europe (Hallmann et al. 2017; Seibold et al. 2019; Widmer et al. 2021). Decreasing insect diversity and abundance also impact other taxa at higher trophic levels using invertebrates as food source (Hallmann et al. 2014). Furthermore, insects provide important ecosystem functions, such as pollination of plants (Ollerton et al. 2011). Phytophagous and detrivorous insects recycle nutrients for plants by decomposing dead material (Mattson and Addy 1975). Thus, the disappearance of insects affects a whole range of other organisms and cause a reduction of various ecosystem functions (Yang and Gratton 2014).
Drivers causing insect diversity decline are manifold (Widmer et al. 2021). As widely accepted, landscape scale drivers are most important for insect decline (Weibull et al. 2000; Rundölf and Smith 2006; Seibold et al. 2019). The transformation of natural ecosystems into agricultural land as well as agricultural intensification are identified as major factors leading to biodiversity loss (Maxwell et al. 2016). In this context, formerly heterogeneous landscapes have been transformed into homogeneous landscapes reducing habitat availability, niche diversity, and subsequently biodiversity (Maxwell et al. 2016). In order to increase yields of food crops, the use of artificial fertilizers and pesticides significantly increased since WWII (Robinson and Sutherland 2002). Meanwhile, pesticides are detectable in all ecosystems even beyond the areas where they were originally applied (Brühl and Zaller 2019). Thus, apart from a complete destruction of habitats, decreasing habitat quality is a major driver of biodiversity loss in Central Europe (Thomas 2016).
In general, heterogeneous agricultural landscapes, characterized by diverse structures such as hedges, wild flower strips and low intensity grassland, perform better in terms of biodiversity than those with less complexity (Estrada-Carmona et al. 2022; Fürst et al. 2022). The additional (semi-natural) habitats compensate for possible negative effects. Therefore, it is essential to consider these habitats providing structures in any study of agricultural effects on biodiversity. In addition, most studies on potential effects of pesticides on biodiversity are based on a comparative study setup, with areas with and without pesticide treatment (Geiger et al. 2010). Although drift of pesticides through landscapes is well documented (e.g., Rosi-Marshall et al. 2007; Hofmann et al. 2016), the harm of pesticides beyond the limits of their actual application needs further investigation, especially for better understanding the importance of the structure and context of organic farms for nature conservation.
In our study, we analysed butterfly species richness, abundance, and community structure along line transects. We observed butterflies in organic farmland (olive and hazelnut plantations) and adjoining meadows and fallow land, the latter two for comparing biodiversity (semi-natural vs. agricultural habitats) and classifying plantations in the landscape context. Although all transects were located on organic farmland, some of them were located directly adjacent to agricultural fields intensively treated with pesticides, while other transects were embedded in organic farmland.
We selected butterflies as model organism to test the importance of different land-use systems and landscape structures as well as the impact of pesticide applications on neighbouring sites on organic farmland. This species group is well suitable for our analyses because many butterflies react highly sensitive to a wide range of environmental parameters and also to toxins (Habel et al. 2019). Furthermore, the ecological demands, behavioural and life-history traits of butterflies are well-known (Bink 1992; Middleton-Welling et al. 2020). Moreover, in addition to their general popularity due to their beauty, butterflies also have high ecological value, as both pollinators (Rader et al. 2020) and food resources for other species such as birds. For all butterfly species found, we performed an ecological classification with respect to ecological specialisation, habitat use, and dispersal behaviour. Based on the results obtained, we address the following research questions:
-
1.
How do plantations differ from each other and from adjacent semi-natural habitats?
-
2.
Do structures like hedges, sub-Mediterranean shrubs, and forest edges positively contribute to butterfly diversity and abundance?
-
3.
Does the direct adjacency of pesticide-treated conventional agricultural areas lead to a decrease of butterfly diversity and abundance on organic farmland?
-
4.
Do the different land-use patterns impact the community structure and its ecological trait composition?
Materials and methods
Study area
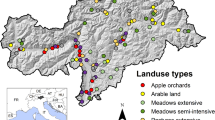
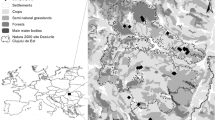
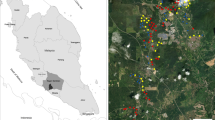
The study was conducted in the agricultural landscape of the municipalities of Grotte di Castro, San Lorenzo Nuovo, and Gradoli (Province Viterbo), near Lake Bolsena in central Italy (300 to 450 m above sea level). The study area covers an organic farm (180 hectares), where mainly olives and hazelnuts were cultivated. Distances between single rows of trees in plantations were 5 m, thus providing sufficient space for a partly flower-rich undergrowth. A fraction of the farmland was covered by meadows and fallow land. Most plantations were bordered by hedges and forest (Quercus and Pinus forest mainly), which—in some locations—may function as barriers between organic and pesticide-treated plantations. The organic farm operated according to biodynamic agricultural standards (no artificial fertilizers, no pesticides), but was surrounded by conventionally managed farmland. Most of the organic farm formed a mostly continuous area. The adjoining conventional farmland has been treated with herbicides and fertilizers during spring, with insecticides once or twice during summer depending on pest infestations, with herbicides again during autumn, and with fungicides throughout the season.
Transects
In total, we established 45 line transects at the organic farm, with each 50 m in length. These transects were set in the following habitats: Olive plantations (24 transects, 8 of which adjacent to pesticide-treated plantations, 16 embedded within continuous organic farmland), hazelnut plantations (13), meadows (4), and fallow land (4) (see Fig. 1). Mean distance to pesticide-treated plantations of all habitat types embedded organically was 50 m, mean distance of olive plantations adjacent to pesticides was 7 m. As the study was performed exclusively on organic farmland (borders are shown as dashed lines in the map), the distribution of the transects had to be arranged partly in a clustered design. Olive plantations had three clusters, located at three different sites (north, centre, and south), hazelnut plantations had two clusters at two sites (north and centre). All other habitat types are each located at one site. The directly adjacent areas are either forests or conventional agriculture. Minimum distance among single transects was 70 m to reduce potential effects from spatial autocorrelation; this should be sufficient as also smaller distances were applied in other studies (e.g., 50 m distance: Willott et al. 2000; Evans et al. 2013). All 45 transects were visited 9 times from 19 April to 9 June 2019 (in total 405 transect walks). Butterfly counts were conducted from 10 am to 5 pm when weather was sunny and warm (≥ 15 °C, no/little wind, according to Pollard and Yates 1993). All butterfly species and individuals were counted 2.50 m to the right and left and 5 m to the front and above the observer. All butterfly counts were conducted by the same person (LG). If possible, species identification was carried out in the field, but some representatives of the families Lycaenidae and Hesperiidae were caught with a butterfly net, photographed (wing upper and lower side), released, and subsequently identified using literature (Tshikolovets 2011; Settele et al. 2015).
Habitat parameters
For each transect, we collected habitat parameters hypothesised to influence butterfly diversity and abundance at the local scale: Degree of shading (estimated %), mean vegetation height (cm, by measuring 3 representative plants), coverage of flowering plants (estimated %), and number of flowering plant species (by counting). The entire area of each transect was considered for the plant survey. The recordings of all parameters were conducted by the same person (LG) in order to ensure consistent estimations. After collecting these values during each transect walk, we calculated mean values per transect for further analysis. In addition, we considered the surrounding ecosystems and their characteristics: the habitat type (olive and hazelnut plantation—the former divided into organically-embedded or adjacent to sites with pesticide applications, meadow, fallow land), context, i.e. surrounding structures (open area, plantation, hedge, or forest edge), disturbance by mowing (0 or 1), existence of sub-Mediterranean shrubs (0 or 1), and adjacency to areas with pesticide applications (0: distance ≥ 15 m, 1: distance ≤ 10 m, distances between 10 and 15 m did not occur).
Butterfly traits
We used traits for each butterfly species according its ecology, behaviour, and life-history and considered the following parameters with respective classifications (according to Bink 1992; Middleton-Welling et al. 2020): Host plant specificity, flight time over the year, generation time in the year, biology/diversity of habitats used, density, nutrient richness of respective habitat preferred, landscape setting, and dispersal (for details see Table 1).
Statistics
For each transect, we summed up the counted individuals for the nine transect walks. Based hereon, the number of species and individuals for each transect was calculated as well as Shannon and Evenness indices. To identify significant differences between different types of sites, i.e., hazelnut and olive plantations (the latter divided into “with” and “without pesticide” application sites adjoining), meadows, and fallow land, we carried out an analysis of variance. As the data did not show homogeneity of variance (Levene’s test, p < 0.05), we used the non-parametric Kruskal–Wallis test performed with p < 0.05 as level of significance. If so, a post-hoc test for a pairwise comparison was carried out using Wilcoxon rank sum test to detect those sites, which differ significantly. The conservative p value adjustment “Bonferroni” was used to avoid high type I errors. To show the direction of the difference, we calculated the means of the numbers of species and individuals, Shannon and Evenness indices and butterfly characteristics with standard deviation as error information. The same procedure was used for analysing differences of hazelnut and olive sites (“north”, “centre” and if applicable “south” and “adjacent to pesticides”). Only for butterfly characteristics, a weighted mean value was used, with the number of individuals for each species as weight.
Non-metric multidimensional scaling (NMDS) was applied to display similarity between sites (using Bray–Curtis dissimilarity), for which ellipses were calculated using the standard deviation with the “veganCovEllipse” function. The two axes of the NMDS were consecutively correlated with the habitat parameters as explanatory variables to determine how the habitat parameters influence the discrimination between sites in terms of their community structure. The habitat parameters “habitat type” and “surrounding structures” were used as factors and shown as ellipses, all other habitat parameters were used as metric predictors and are shown as arrows, in case they were significant.
Generalized linear models (GLMs) were constructed for both species richness and abundance of individuals as dependent variables. Only the data of the “habitat types” actively used for agriculture (i.e., hazelnut and both types of olive plantations) entered the GLMs to obtain an undistorted picture of the effect of these agricultural practices, which also reduces the “surrounding structure” to plantation, hedge, and forest edge. The potential effect of meadows and fallow land interspersed in agriculturally used areas is revealed by the above-mentioned analyses. Model selection was performed by applying multi-model inference and ranking all models by their corrected Akaike Information Criterion (AICc). So, all models within an AICc difference of two were considered (Burnham and Anderson 2001; Johnson and Omland 2004). This procedure resulted in several models for both response variables, which were all considered in the following. For individual addressing, each model was given an abbreviation: SR (species richness) or A (abundance) and a number that increases with its AICc, e.g., “SR 2” for the model with the response variable species richness and the second lowest AICc. Prior to model building, we tested for multicollinearity of habitat parameters without detecting critical bivariate correlations, all |rs| were < 0.7 (Schröder et al. 2009; Dormann et al. 2013). As no correlation with the surrounding structures was calculated, but a pre-analysis by boxplots showed a high connection to the degree of shading (forest edges had a significantly higher degree of shading than plantations), these two parameters were never used as predictors at the same time. The same applies for the predictors “habitat type” and “adjacency to pesticides”, as they contain partially identical information. An analysis of variance was performed, as described above, to quantify differences in the degree of shading between the types of surrounding structures used in GLM analysis. All habitat parameters were used as additive predictor variables, all two-way interactions (34 variations) have been checked, but did not lead to an improvement. GLMs of species richness were created using the log link function implying Poisson distributed error terms. Since this distribution led to overdispersion when abundance was modelled, we used the log link function for error terms with a negative-binomial distribution instead. Models were evaluated using the explained deviance. Model validation was performed using four-fold cross-validation. Additionally, we calculated spline correlograms of model residuals (Bjørnstad and Falck 2001), which did not detect any significant residual autocorrelation for all models.
Data were analysed and visualised using the R 4.0.0 environment (R Core Team 2020) using the packages vegan (Oksanen et al. 2019), car (Fox and Weisberg 2019), DHARMa (Harting 2020), MuMIn (Barton 2020), dplyr (Wickham et al. 2022), ggplot2 (Wickham 2016), ncf (Bjørnstad and Cai 2022) and MASS (Veneables and Ripley 2002).
Results
In total, we recorded 1,364 butterfly individuals representing 33 species across all transects. The most abundant species were Coenonympha pamphilus (476 individuals), and Maniola jurtina (337), followed by Aricia agestis (97), Ochlodes sylvanus (93), and Polyommatus icarus (59), a complete list of counted species is provided as electronic appendix. Four additional species were observed nearby transects (Zerynthia cassandra, Gonepteryx rhamni, Lycaena thersamon, Inachis io). Species richness and abundance did not differ significantly among the five habitat types, however, Shannon index values differed significantly between olive plantations and fallow land, Evenness between olive and hazelnut plantations (Table 2). While the highest values for species richness, Shannon index, and Evenness were obtained for the fallow land transects, the highest average number of individuals was found on the transects crossing olive plantations not adjoining areas with pesticide application.
NMDS analysis clearly separated the butterfly communities of most fallow land transects from all others. A moderate differentiation was obtained between olive and hazelnut plantation communities. Olive plantation communities, however, only exhibited a weak differentiation between such adjoining pesticide-treated sites and the ones embedded in organic farmland. The four meadow transects showed no distinguished butterfly community structure, but were most similar to some olive or hazelnut transects (Fig. 2a). The NMDS also seperates butterfly communities by their surrounding structure. Forest edges covered similar communities as fallow land and hazelnut plantations and were clearly seperated from the other groups, but slightly overlaped with hedges. Plantations, open areas, and hedges showed mostly overlapping community structures (Fig. 2b). Arrows of habitat parameters showed that butterfly communities on fallow land were determined by the presence of sub-Mediterranian shrubs and limited amounts of shadow, and communities of both types of olive plantations by a high number of flowering plant species. Hazelnut plantations and forest edges provided a higher degree of shading than the other habitat types and structures, which led to a clearly different community structure (Fig. 2c).
Commonalities in butterfly communities shown by an NMDS plot (stress = 0.164) with standard deviation ellipses showing a the habitat types (p < 0.05, olive a.p. = olive adjacent to pesticides), b the surrounding structure (p < 0.05), and c environmental gradients (p < 0.05, No. fps: number of flowering plant species, sub-Med. shrub: sub-Mediterranean shrub) given as arrows
A trait analysis referring to host plant specialisation and flight season aspects revealed diverse significant differences among the five habitat types (Table 3). Thus, host plant specificity of butterflies was strongest for butterflies observed at meadows and in olive plantations without contact to areas treated with pesticides, while the weakest specificities, differing significantly to these olive plantations, were obtained for fallow land. The mean number of flying months was significantly lower in olive plantations adjoining sites with pesticide application than at meadows. Similar results were observed for the mean onset and end of the flight season of the communities being latest and earliest, respectively, in olive plantations adjoining sites with pesticide applications.
In contrast, the means of the other assessed traits (i.e. biology, population density, nutrient richness, area requirement, dispersal behaviour) did not differ between olive plantations, irrespective whether adjoining sites were treated with pesticides or not (Table 4). The assemblies with lowest average population densities of the species were obtained on fallow land transects, and the mean area requirement was significantly lower in olive plantations than hazelnut plantations and fallow land. The mean dispersal behaviour was highest for butterflies observed on fallow land.
We estimated GLMs for the number of species and the abundance of individuals. For species numbers, four models were within the range of ∆AICc of two, for abundance three models (Table 5). Four out of eight potential predictor variables were relevant for species richness and five for abundance (Table 6). The predictor “number of flowering plant species” was present in all seven models, and had a strong positive effect. Species richness was also influenced by the surrounding structure, degree of shading, and vegetation height, the last two with a slight negative impact. Abundance was also explained by the surrounding structure and adjoining sites with pesticide applications in two out of three models. Furthermore, abundance was additionally explained in one out of three models by the degree of shading along with the coverage of flowering, plants; both variables promoted a slight negative impact. The explained deviance was good with a minimum of 22.7%, especially for the abundance models with about 2 to 3 times higher values (Table 5).
We used exemplary bivariate partial dependence plots to visualise the isolated effect of categorical habitat parameters in combination with the most important habitat parameter, the number of flowering plant species. This parameter had a strong, positive effect on both species richness and abundance (Fig. 3a, b). The effect of the surrounding structure (plantation, hedge, forest edge) varied between species richness and abundance. Species richness was positively influenced by hedges, abundance was positively associated with surrounding plantations. We had lowest species richness and abundance at forest edges. For later interpretation, it is important to note that the degree of shading was significantly higher at forest edges compared to within plantations (forest edge: 48.3% ± 23.5, hedge: 12.5% ± 15.8, plantation: 4.5% ± 7.3). The abundance and the positive effect of the number of flowering plant species was slightly lower at transects located adjacent to pesticide-treated plantations (Fig. 3c).
Exemplary partial dependence plots based on Generalized Linear Models, prediction (line) and 95% confidence intervals (shaded area, the darker the line, the darker the associated interval) of: a species richness (GLM SR1) in relation to the number of flowering plant species depending on the surrounding structure; abundance (GLM A1) in relation to the number of flowering plant species depending on b the surrounding structure and c whether the transect was adjacent to pesticides or not. The points represent the observations, coloured in the respective group shown
As most of the habitat types are located at one site, except for hazelnut and olive plantations, which were located at several sites, we compared species richness, abundance and, in case of significant differences, also habitat parameters between sites. No significant differences in species richness and abundance were found at hazelnut plantations. In contrast to species richness, abundance differed significantly, however only between the sites “south” and “adjacent to pesticides”, in olive plantations. Significantly more individuals were recorded in the south (south: n = 11, A = 53.1 ± 20.6; adjacent to pesticides: n = 8, A = 26.9 ± 7.2). At these sites, there was no significant difference between habitat parameters that were identified as important for abundance (see GLMs A1–3: number of flowering plant species, degree of shading, coverage of flowering plants, and surrounding structure), all olive transects in the south and adjacent to pesticides were surrounded by plantations. The only difference between these sites was the proximity to pesticide-treated plantations.
Discussion
In general, landscapes consisting of a mosaic of different habitats provide a large variety of ecological niches, larval food plants, and nectar sources. Such habitat mosaics are the prerequisite for the co-existence of many species and have the potential for high abundances of butterflies, and of arthropods in general (see Marini et al. 2009). In this context, it is consistent that we found significant differences in the composition of the butterfly species community among different habitat types. Particularly, there were strong differences between plantations (hazelnut, olive) and fallow land, but only marginal differences in butterfly community structures between the two types of plantations (hazelnut and olive). Butterfly community composition on meadows is very similar to the one found for olive and hazelnut plantations.
Most importantly, butterfly abundance and species richness increased with an increasing number of flowering plant species, which might attract butterfly individuals of different species with different flower-use habits. This leads to the accumulation of species and individuals at locations with high flower diversity and density. A positive relationship between the number of flowers and the number of butterflies has also been demonstrated in previous studies, and underlines the attraction effect of flowers on insects (Rundlöf et al. 2008; Nicholls and Altieri 2013; Kurylo et al. 2020). However, as one model even suggested a weak negative relationship between butterfly abundance and coverage of flowering plants, we assume that flowering plants are sufficiently available and in general not a limiting resource in our study area. Hence, the species number of flowering plant species from some point of density is apparently more important than the coverage. Furthermore, species richness might decrease slightly with increasing height of the vegetation because of the changing microclimate and higher competition of few plant species.
Our findings also underline that species richness and abundance decrease with increased shading. This relation was frequently observed for flying insects which strongly rely on warm and sunny conditions (Clark et al. 2007; Matteson and Langellotto 2010). When forest edges result in significantly higher levels of shading, as in this study, these structures can have a negative impact on diversity and abundance of butterflies. For the number of individuals, the plantation itself turned out to be the best surrounding structure as they form a heterogeneous landscape by covering several successional stages (ground vegetation with a variety of heights and trees). In addition, plantations have a limited amount of shadow, due to the even spacing of tree rows. This is even more the case for olive plantations than hazelnut plantations, because, due to their small leaves, they have a comparatively less dense leaf canopy and thus cast less shade. This difference is also noticeable in species composition. Species richness, on the other hand, was positively influenced by the presence of hedges because they considerably increase structural diversity important for butterfly diversity but not so much shadow.
We found highest butterfly species richness, abundance, and Shannon index on fallow land, and lowest on hazelnut plantations. The fallow land in this region was mainly characterized by little amounts of shadow and often adjoining sub-Mediterranean shrub, both of which had a visible influence on species composition. In parallel, lowest host plant specialisation and highest mean dispersal behaviour were identified for the butterflies on fallow land. Previous studies already indicated that fallow land is of high relevance for a large proportion of insects as this habitat type provides prerequisites for successful larval development for many species (Schmitt et al. 2008) because a comparatively low level of habitat disturbance creates excellent conditions for successful larval development (Kruess and Tscharntke 2002). Patches of fallow land interspersed across intensively used agricultural fields therefore may support the persistence of species and species´ mobility and exchange across landscapes (Aviron 2007), and thus support individual exchange among local populations and subsequently population and species persistence (Baguette et al. 2013). A large proportion of butterfly species found on fallow land had comparatively high Bink values for dispersal behaviour (Bink 1992). Previous studies also showed that most plant and animal communities on fallow land are composed by typical pioneer species with high dispersal, and consequently host a specific plant and animal community (Scalercio et al. 2007). In conclusion, patches of fallow land distributed within agricultural landscapes are generally accepted as being of high conservation relevance for the following reasons: (1) These habitats provide resources for a very unique community of taxa; (2) fallow land provides excellent conditions for undisturbed development of organisms; and (3) fallow land is an important stepping-stone connecting potential habitats increasing the permeability of landscapes, and thus guarantees the persistence of species (Tscharntke et al. 2002).
Furthermore, we found lower abundances of butterflies in olive plantations adjoining fields treated with pesticides compared to those embedded in organic farmland. More importantly, the comparison of individual olive sites shows significantly lower abundances in olive plantations near pesticide-treated plantations than those embedded in organic farmland in the south of the study area, despite having the same surrounding structures and not significantly varying habitat parameters. Furthermore, the communities’ flight season seems to be significantly reduced on the sites adjoining fields with pesticide applications. Negative effects on species diversity and individual abundance from pesticide applications have already been observed and reported (Braak et al. 2018). Insecticides might directly impact insect species on the sites of application, as well as beyond due to drift effects (Zaller and Brühl 2019; Zaller 2020; Brühl and Zaller 2021).
Herbicides at least indirectly impact insect species via the vanishing of a large variety of potential host plants. We found that host plant specialisation is second lowest for butterflies found on sites with adjoining fields treated with pesticides. This might directly correlate with the vanishing of a large number of plant species and subsequently the loss of potential food plants for larval development. While polyphagous species are able to use alternative food plants, monophagous insects strongly rely on very specific larval food plant species and thus show comparatively little plasticity.
The trends obtained from our study underline that pesticides have a rather negative effect on biodiversity, even beyond the areas where they were applied. Topography also plays a decisive role in this context. Thus, studies conducted in areas with frequent thermal winds revealed precipitation and accumulation of pesticides uphill (up to 300 m up-slope) and in particular on neighbouring southern slopes, with strong negative effects on butterfly diversity (Huemer 2001; Tarmann 2019). In conclusion, intermixing conventional and organic agriculture is critical as toxic substances might negatively impacts biodiversity—also beyond the fields, which have been treated. If not possible to disentangle both completely or at least in large separate blocks, at a minimum, buffer areas should be established between fields under conventional and organic management to minimize negative edge effects.
Nevertheless, this field study still has shortcomings that should be overcome in future studies. Thus, our data were collected during the spring season best suited for such an essay in central Italy, but excludes the other seasons. In addition, no year to year effects could be included as the study was conducted in a single year. Future studies therefore should if possible include more seasonal aspects and different years. Additionally, including other organic farms in a future extended study would be beneficial to archive more generally valid results. The increase in transect numbers, especially with more homogeneously distributed classes of habitat types, also might help detecting further significant relationships.
References
Aviron S, Kindlmann P, Burel F (2007) Conservation of butterfly populations in dynamic landscapes: the role of farming practices and landscape mosaic. Ecol Model 205(1–2):135–145
Baguette M, Blanchet S, Legrand D, Stevens VM, Turlure C (2013) Individual dispersal, landscape connectivity and ecological networks. Biol Rev 88:310–326
Barton K (2020) MuMIn: multi-model inference (Version 1.43.17)
Bink FA (1992) Ecologische atlas van de dagvlinders van Noordwest-Europa. Schuyt, Haarlem
Bjørnstad ON, Cai J (2022) ncf: Spatial covariance functions (Version 1.3-2)
Bjørnstad ON, Falck W (2001) Nonparametric spatial covariance functions: estimation and testing. Environ Ecol Stat 8:53–70
Braak N, Neve R, Jones AK, Gibbs M, Breuker CJ (2018) The effects of insecticides on butterflies—a review. Environ Pollut 242:507–518
Brühl CA, Zaller JG (2019) Biodiversity decline as a consequence of an inappropriate environmental risk assessment of pesticides. Front Environ Sci 7:177
Brühl CA, Zaller JG (2021) Indirect herbicide effects on biodiversity, ecosystem functions, and interactions with global changes. Herbicides. https://doi.org/10.1016/B978-0-12-823674-1.00005-5
Burnham KP, Anderson DR (2001) Kullback-Leibler information as a basis for strong inderence in ecological studies. Eur J Wildl Res 28:111–119
Clark PJ, Reed JM, Chew FS (2007) Effects of urbanization on butterfly species richness, guild structure, and rarity. Urban Ecosyst 10(3):321–337
Dirzo R, Young HS, Galetti M, Ceballos G, Isaac NJB, Collen B (2014) Defaunation in the Anthropocene. Science 345(6195):401–406
Dormann CF, Elith J, Bacher S, Buchmann C, Carl G, Carré G, Marquéz JRG, Gruber B, Lafourcade B, Leitão PJ, Münkemüller T, Mcclean C, Osborne PE, Reineking B, Schröder B, Skidmore AK, Zurell D, Lautenbach S (2013) Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography 36(1):27–46
Estrada-Carmona N, Sánchez AC, Remans R, Jones SK (2022) Complex agricultural landscapes host more biodiversity than simple ones: a global meta-analysis. Agricultural Sci 119(38):e2203385119
Evans TR, Muserts CJM, Cashatt ED, de Snoo GR (2013) Lepidoptera pest species response to mid-summer fire. Fire Ecol 9(3):25–32
Fox J, Weisberg S (2019) An R companion to applied regression (Version 3.1-0)
Fürst J, Bollmann K, Gossner MM, Duelli P, Obrist MK (2022) Increased arthropod biomass, abundance and species richness in an agricultural landscape after 32 years. J Insect Conserv. https://doi.org/10.1007/s10841-022-00445-9
Geiger F, Bengtsson J, Berendse F, Weisser WW, Emmerson M, Morales MB, Inchausti P (2010) Persistent negative effects of pesticides on biodiversity and biological control potential on european farmland. Basic Appl Ecol 11(2):97–105
Habel JC, Ulrich W, Biburger N, Seibold S, Schmitt T (2019) Agricultural intensification drives butterfly decline. Insect Conserv Divers. https://doi.org/10.1111/icad.12343
Hallmann CA, Foppen RPB, van Turnhout CAM, de Kroon H, Jongejans E (2014) Declines in insectivorous birds are associated with high neonicotinoid concentrations. Nature 511(7509):341–343
Hallmann CA, Sorg M, Jongejans E, Siepel H, Hofland N, Schwan H, de Kroon H (2017) More than 75% decline over 27 years in total flying insect biomass in protected areas. PLoS ONE 12(10):e0185809
Hartig F (2020) DHARMa: Residual diagnostics for hierarchical (multi-level/mixed) regression models (version 0.3.1)
Hofmann F, Kruse-Plass M, Kuhn U, Otto M, Schlechtriemen U, Vogel SB, Wosniok R W (2016) Accumulation and variability of maize pollen deposition on leaves of european Lepidoptera host plants and relation to release rates and deposition determined by standardised technical sampling. Environ Sci Europe 28:14
Huemer P (2001) Ökologische Bewertung nachtaktiver Schmetterlingsgemeinschaften (Lepidoptera) im Biotop Kalterer see (Südtirol). Gredleriana 1:419–447
Johnson JB, Omland KS (2004) Model selection in ecology and evolution. Trends Ecol Evol 19:101–108
Kruess A, Tscharntke T (2002) Grazing intensity and the diversity of grasshoppers, butterflies, and trap-nesting bees and wasps. Conserv Biol 16(6):1570–1580
Kurylo JS, Threlfall CG, Parris KM, Ossola A, Williams NSG, Evans KL (2020) Butterfly richness and abundance along a gradient of imperviousness and the importance of matrix quality. Ecol Appl. https://doi.org/10.1002/eap.2144
Marini L, Fontana P, Battisti A, Gaston KJ (2009) Agricultural management, vegetation traits and landscape drive orthopteran and butterfly diversity in a grassland-forest mosaic: a multi-scale approach. Insect Conserv Divers 2(3):213–220
Matteson KC, Langellotto GA (2010) Determinates of inner city butterfly and bee species richness. Urban Ecosyst 13(3):333–347
Mattson WJ, Addy ND (1975) Phytophagous Insects as regulators of forest primary production. Science 190(4214):515–522
Maxwell SL, Fuller RA, Brooks TM, Watson JEM (2016) Biodiversity: the ravages of guns, nets and bulldozers. Nature 536(7615):143–145
Middleton-Welling J, Dapporto L, García-Barros E, Wiemers M, Nowicki P, Plazio E, Shreeve T (2020) A new comprehensive trait database of european and Maghreb butterflies, Papilionoidea. Sci Data 7(1):351
Nicholls CI, Altieri MA (2013) Plant biodiversity enhances bees and other insect pollinators in agroecosystems: a review. Agron Sustain Dev 33(2):257–274
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Wagner H (2019) vegan: community ecology package (Version 2.5-5)
Ollerton J, Winfree R, Tarrant S (2011) How many flowering plants are pollinated by animals? Oikos 120(3):321–326
Pollard E, Yates TJ (1993) Monitoring butterflies for ecology and conservation: the british butterfly monitoring scheme, 1st edn. Chapman & Hall, New York
R Core Team (2020) R: a language and environment for statistical computing. Version R Foundation for Statistical Computing, Vienna
Rader R, Cunningham SA, Howlett BG, Inouye DW (2020) Non-bee insects as visitors and pollinators of crops: Biology, Ecology, and management. Ann Rev Entomol 65:391–407
Robinson RA, Sutherland WJ (2002) Post-war changes in arable farming and biodiversity in Great Britain. J Appl Ecol 39:157–176
Rosi-Marshall EJ, Tank JL, Royer TV, Whiles MR, Evans-White M, Chambers C, Griffiths A, Pokelsek J, Stephen ML (2007) Toxins in transgenic crop byproducts may affect headwater stream ecosystems. Proc Natl Acad Sci USA 104:16204–16208
Rundlöf M, Smith HG (2006) The effect of organic farming on butterfly diversity depends on landscape context: organic farming, landscape and butterflies. J Appl Ecol 43(6):1121–1127
Rundlöf M, Bengtsson J, Smith HG (2008) Local and landscape effects of organic farming on butterfly species richness and abundance: scale-dependent effects of organic farming. J Appl Ecol 45(3):813–820
Sánchez-Bayo F, Wyckhuys KAG (2019) Worldwide decline of the entomofauna: a review of its drivers. Biol Conserv 232:8–27
Scalercio S, Iannotta N, Brandmayr P (2007) The role of semi-natural and abandoned vegetation patches in sustaining lepidopteran diversity in an organic olive orchard. Bull Insectol 60:13
Schmitt T, Augenstein B, Finger A (2008) The influence of changes in viticulture management on the butterfly (Lepidoptera) diversity in a wine growing region of southwestern Germany. Europ J Entomol 105(2):249–255. https://doi.org/10.14411/eje.2008.035
Schröder B, Strauss B, Biedermann R, Binzenhöfer B, Settele J (2009) Predictive species distribution modelling in butterflies. In: Shreeve TG, Settele J, Konvicka M, van Dyck H (eds) Ecology of butterflies in Europe. Cambridge University Press, Cambridge, pp 62–78
Seibold S, Gossner MM, Simons NK, Blüthgen N, Müller J, Ambarlı D, Weisser WW (2019) Arthropod decline in grasslands and forests is associated with landscape-level drivers. Nature 574(7780):671–674
Settele J, Steiner R, Reinhardt R, Feldmann R, Hermann G (2015) Schmetterlinge: Die Tagfalter Deutschlands (3. aktualisierte Auflage). Stuttgart (Hohenheim): Ulmer
Tarmann GM (2019) Vergleich der historischen und aktuellen Verbreitung von Chazara briseis (Nymphalidae) und Zygaenidae (Lepidoptera) im oberen Vinschgau (Südtirol, Italien) zeigt ein komplettes Verschwinden der Zygaenidae in talnahen Gebieten. Gredleriana 19:109
Thomas JA (2016) Butterfly communities under threat. Science 353(6296):216–218
Tscharntke T, Steffan-Dewenter I, Kruess A, Thies C (2002) Contribution of small habitat fragments to conservation of insect communities of grassland-cropland landscapes. Ecol Appl 12(2):354–363
Tshikolovets V V (2011) Butterflies of Europe and the Mediterranean area. Tshikolovets Publications, Pardubice
Venables WN, Ripley BD, By WN, Venables, Ripley BD (2002) Random and mixed effects. In: Chambers J, Eddy W, Härdle W, Sheather S, Tierney L, (Series (eds) Modern applied statistics with S. Springer New York, New York, pp 271–300
Weibull A-C, Bengtsson J, Nohlgren E (2000) Diversity of butterflies in the agricultural landscape: the role of farming system and landscape heterogeneity. Ecography 23(6):743–750
Wickham H (2016) ggplot2: elegant graphics for data analysis (Version 3.3.6). Springer New York, New York
Wickham H, François R, Henry L, Müller K (2022) dplyr: a grammar of data manipulation (version 1.0.9)
Widmer I, Mühlethaler R, Baur B, Gonseth Y, Guntern J, Klaus G, Knop E, Lachat T, Moretti M, Pauli D, Pellissier L, Sattler T, Altermatt F (2021) Insektenvielfalt in der Schweiz: Bedeutung, Trends, Handlungsoptionen. Swiss Academies Reports 16: 108 https://doi.org/10.5281/ZENODO.5144739
Willott SJ, Lim DC, Compton SG, Sutton SL (2000) Effects of selective logging on the butterflies of a Bornean Rainforest. Conserv Biol 14:1055–1065
Yang LH, Gratton C (2014) Insects as drivers of ecosystem processes. Curr Opin Insect Sci 2:26–32
Zaller JG (2020) What is the Problem? Pesticides in our everyday life. In: Zaller JG (ed) Daily poison. Springer International Publishing, Cham, pp 1–125
Acknowledgements
We thank Claudia and Karsten Greve for providing their property as well as for financial and logistic support to collect these data. We thank Mechthild von Wolzogen and all employees of the Azienda Agricola AURORA for their support during field work. Mike Teucher (Halle) is acknowledged for help with GIS applications. We thank Dagmar Söndgerath (Braunschweig) for supervising the beginnings of this study and Anne-Kathrin Schneider (Braunschweig) and Swantje Löbel (Braunschweig) for the helpful discussions. We thank two anonymous reviewers for very valuable and constructive comments on draft versions of this article.
Funding
Open Access funding enabled and organized by Projekt DEAL. There are no sources of funding to be reported.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There exist no conflicts of interest (financial or non-financial).
Research involving human and animal rights
We guarantee the welfare of animals involved in this study; butterflies were immediately released after catching and taking photographs
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Guderjan, L., Habel, J.C., Schröder, B. et al. Land-use pattern and landscape structure impact butterfly diversity and abundance in organic agroecosystems. Landsc Ecol 38, 2749–2762 (2023). https://doi.org/10.1007/s10980-023-01731-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-023-01731-w