Abstract
Context
The area-isolation paradigm of metapopulation theory predicts that larger and more connected patches have a higher probability of occupancy. Although it may be too simplistic for generalist terrestrial mammals, the area-isolation paradigm may be an effective tool for assessing patch-occupancy for habitat specialists.
Objectives
We tested predictions of the area-isolation paradigm for the endangered salt marsh harvest mouse (Reithrodontomys raviventris), a habitat specialist living in highly fragmented salt marsh habitat in the San Francisco Estuary (California, USA).
Methods
We surveyed for salt marsh harvest mice at 47 marsh patches throughout their range using a non-invasive genetic survey technique. We used occupancy modeling to estimate the effects of patch size, patch connectivity, matrix urbanization, and several habitat characteristics on occupancy probabilities. We evaluated occupancy at both coarse (e.g., among patches) and fine (e.g., within patches) spatial scales.
Results
Patch size, connectivity, and matrix urbanization had significant effects on patch-occupancy. Within patches, occupancy was positively related to the presence of high-tide escape vegetation. Our data also revealed the extirpation of several geographically distinct populations, consistent with expectations due to reduced patch sizes and connectivity over the past century.
Conclusions
Patterns of salt marsh harvest mouse patch-occupancy were consistent with the area-isolation paradigm. In addition, our models provide important guidelines of patch size and connectivity that can inform habitat conservation and restoration for this endangered species. Specifically, our data suggests that selecting restoration sites that are well-connected may be more beneficial than selecting larger, isolated sites.
Similar content being viewed by others
Introduction
Habitat fragmentation is one of the leading threats to wildlife worldwide (Haddad et al. 2015). Many endangered species are listed as such due to past and ongoing habitat fragmentation (Kerr and Cihlar 2004). Although the effects of habitat fragmentation ‘per se’ (the breaking apart of habitat, independent of habitat loss) may be confounded with the effects of habitat loss (Fahrig 2003), the two processes are highly correlated such that their effects are often indistinguishable (Koper et al. 2007; Hadley and Betts 2016; Liu et al. 2016; Fletcher et al. 2018). When endangered species live in patchy, fragmented distributions, identifying patch size and connectivity thresholds that facilitate population persistence can inform conservation actions. The “area-isolation paradigm” draws on insights from island biogeography (MacArthur and Wilson 1967; Diamond 1975) and metapopulation theory (Hanski 1991, 1998), and predicts that the probability that a species will occupy a given patch is related proportionally to patch size and inversely to patch isolation. However, it is less clear how species that evolved in more continuous patches of habitat will cope with an increased need for dispersal through a recently fragmented landscape.
Applying the area-isolation paradigm to nature can be challenging because it requires a binary delineation of the landscape into patches of suitable habitat and a matrix of non-habitat, which may be an oversimplification for most species (Hanski 1998; Prugh et al. 2008). However, when species are highly specialized, discrete habitat patches and non-habitat matrix may be a reasonable representation of the landscape (Hanski et al. 1995; Duggan et al. 2011). Additionally, habitat covariates can be incorporated into models to represent habitat patches more realistically in terms of variable habitat patch quality, which can be more important than the spatial configuration of habitat patches in some systems (Ye et al. 2013; Poniatowski et al. 2018). Thus, patch-occupancy models that integrate components of habitat and matrix quality with the area-isolation paradigm can be useful for assessing the value of specific habitat patches to some focal species (Wahlberg et al. 1996; Schultz and Crone 2005; Bollmann et al. 2011).
The salt marsh harvest mouse (Reithrodontomys raviventris) is an endangered species (USFWS 1970; CDFW 1971) endemic to the coastal wetlands of the San Francisco Bay and connecting tidal bodies, collectively, the San Francisco Estuary (SFE; USFWS 2013). Tidal wetlands in the SFE have been fragmented over the past century to the point where < 10% of historic tidal marsh habitat remains intact (Williams and Faber 2001). Given such levels of habitat loss, understanding minimum marsh patch size and connectivity needed to support salt marsh harvest mouse populations can be useful for predicting occupancy in remaining habitat and in guiding habitat restoration projects (Shellhammer and Duke 2010).
Although much attention has focused on vegetation characteristics of salt marsh harvest mouse habitat (Johnson and Shellhammer 1988; Bias and Morrison 2006; Sustaita et al. 2011; Smith et al. 2018a), as well as some abiotic factors (e.g., distance from roads—Marcot et al. 2020), spatial drivers of occupancy have been largely unexplored (Smith et al. 2018a). Despite a lack of research into patch dynamics and spatial habitat associations, their importance is highlighted by calls for maintenance or restoration of functionally connected habitat patches in the Recovery Plan for Tidal Marsh Ecosystems of Northern and Central California (USFWS 2013). Given the extensive history of habitat loss and threat of future habitat fragmentation from development and sea level rise (Thorne et al. 2018), identifying spatial thresholds that facilitate occupancy is an essential foundation for salt marsh harvest mouse conservation and management (Smith et al. 2018b).
Historically, the SFE was composed of large, continuous marsh habitat, contrasting with the modern landscape composed of a mosaic of small marsh patches surrounded most often by water, terrestrial grassland, or urban land cover (Williams and Faber 2001). Salt marsh harvest mice are primarily restricted to marsh habitat; upland and urban habitat edges are thought to be favorable to potential competitors, such as western harvest mice (R. megalotis), house mice (Mus musculus), and California voles (Microtus californicus) (Fisler 1965; Bias and Morrison 2006). Therefore, spatial attributes such as large patch size may buffer salt marsh harvest mice from negative edge effects. Because their habitat is inherently dynamic due to tidal action, salt marsh harvest mice must regularly negotiate tide cycles that can force them to take temporary refuge either in emergent vegetation (Johnston 1957; Smith et al. 2014) or by movement to non-inundated habitat (Hadaway and Newman 1971). If no refuge habitat is available, extreme high tide events have the potential to cause local extirpations. In such cases, long-term population persistence may rely on extinction-colonization dynamics as described in classical metapopulation theory (Levins 1969; Harrison 1991). Therefore, a landscape of many well-connected patches may be more resilient to extreme tidal events than a landscape characterized by one or a few large patches.
Microhabitat characteristics may also drive occupancy at finer (within-patch) scales. Pickleweed (Salicornia spp.) cover is assumed to be strongly associated with salt marsh harvest mouse presence (USFWS 2013), but diverse vegetation composition may be more beneficial than pickleweed monocultures (Smith et al. 2018a). Gumplant (Grindelia spp.) and other tall emergent vegetation may represent important high tide refuge for salt marsh harvest mice during extreme inundation events (Johnston 1957; Smith et al. 2014). Marsh patches with greater biomass of upland vegetation may be more likely to support competitors, such as western harvest mice (Fisler 1965).
Given that salt marsh harvest mice are restricted to marsh habitat (USFWS 2013; Statham et al. 2022), which is readily delineated from upland and urban habitats, assumptions of the area-isolation paradigm may be strongly applicable to this system. Our primary objective was to apply occupancy modeling (MacKenzie et al. 2002) to test the relationship between salt marsh harvest mouse occupancy and patch size, patch connectivity, patch vegetation characteristics, and matrix urbanization (i.e., the proportional composition of urban land cover within matrix habitat). Additionally, we assessed spatial and vegetation characteristics that influenced salt marsh harvest mouse occupancy at finer scales (i.e., within patches), and compared historical and modern habitat and occupancy data in a dynamic occupancy model (MacKenzie et al. 2003; i.e., multi-season occupancy model with historical and modern representing different “seasons”) to estimate relationships between local extinction probabilities and characteristics of habitat change over decades. Finally, occupancy modeling allowed us to quantify the detection probability, and determine covariates that influence detection probability, of a recently developed non-invasive genetic survey technique (Aylward et al. 2022). These objectives allowed us to assess the utility of the area-isolation paradigm as a tool for conservation and management of an endangered species in fragmented habitat.
Methods
Non-invasive genetic surveys
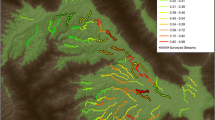
We conducted non-invasive genetic surveys at 47 patches (i.e., distinct marshes) from September 2020 to December 2021 (Fig. 1; Supplementary Table S1; Supplementary Fig. 1). We identified patches based on the San Francisco Estuary Institute Bay Area EcoAtlas Dataset (SFEI 1998). Patches were delineated on the basis of discontinuity (i.e., separation of marshes by non-marsh habitat), tidal regime (i.e., whether diked or tidal), and elevational strata (i.e., adjacent sections of low-elevation tidal marsh and high-elevation tidal marsh would be considered two distinct patches). In some cases, we used satellite imagery to manually add patches that were missing from the SFEI data set. Most surveys occurred during late fall and early winter (Oct–Feb) to limit potential seasonal variation in occupancy and/or detection. We placed bait stations within vegetation ~ 0.5–1 m above ground level along two transects in each marsh patch. We placed stations 20 m apart along transects with a maximum of 10 stations per transect, and curtailed the number of stations for transects in smaller marshes (range 4–10). We fitted bait stations with cotton batting and ~ 1 tbsp of seed mix (primarily oats, millet, and ground walnut). We checked and re-baited stations after 7 days and checked stations again after 14 days, providing two consecutive 7-day survey intervals. During each check, fecal pellets were collected in 2-ml ethanol (> 95%) vials; we pooled as many pellets as possible (without over-topping the ethanol) into a single vial for each station. At each station, we recorded several vegetation parameters within a five-meter radius: vegetation richness, presence/absence of pickleweed, presence/absence of high tide escape vegetation (HTEV; emergent vegetation with structural support and overall height great enough to provide refugial habitat during high tide, judged based on expert opinion), and presence/absence of terrestrial (upland) grasses. We conducted genetic species identification on fecal pellets following Aylward et al. (2022). Non-invasive genetic surveys were approved by UC Davis Institutional Animal Care and Use Committee and authorized by the California Department of Fish and Wildlife and United States Fish and Wildlife Service Cooperative Agreement.
Map of the study area and number of occupied/surveyed sites for salt marsh harvest mice (Reithrodontomys raviventris). Number of occupied and surveyed sites are listed for each region of the study area. The lower-right inset shows the detection status of specific patches of marsh habitat surveys in the Southwest region. Marsh habitat is colored purple in the large study area pane; detection status is only colored in the Southwest inset pane
Patch occupancy modeling
At the patch-scale, we considered a patch occupied if salt marsh harvest mice were detected at ≥ 1 station(s) within the patch. Next, we applied occupancy modeling (MacKenzie et al. 2002) to estimate the probabilities of detection (i.e., the probability of detecting salt marsh harvest mice given they are present) and occupancy (i.e., the probability of salt marsh harvest mouse occurrence) using the R package ‘unmarked’ (Fiske and Chandler 2011). Occupancy modeling accounts for imperfect detection of survey methods by estimating the probability of detection based on the number of survey sites in which a focal species was detected in ≥ 1, but not all, survey intervals (MacKenzie et al. 2002). We tested for effects of survey interval (i.e., first 7 days vs. second 7 days), survey effort (i.e., number of stations in the patch), and maximum high tide height during the survey interval on detection probabilities. We then estimated occupancy probability at the patch level as a function of five parameters: patch size, patch connectivity, matrix urbanization, patch vegetation characteristics, and the capture frequencies of three putative competitors (Supplementary Table S2). We defined patch size with and without edge effects of 50 and 200 m (i.e., subtracted 50 and 200 m buffers from the patch perimeter); patch connectivity as the proportion of marsh habitat within 50-m, 200-m, or 1-km buffers from the edge of the target patch; and matrix urbanization as the proportion of the matrix (i.e., non-marsh habitat within 50-m, 200-m, or 1-km of the patch) that was composed of urban land cover. Vegetation parameters included vegetation richness, pickleweed habitat, HTEV habitat, and terrestrial grass habitat. Vegetation richness was calculated at the patch level as the average richness at all stations within the patch. Pickleweed, HTEV, and terrestrial grass habitats were quantified as the proportion of stations where these habitat characteristics were recorded. Finally, the role of putative competitor species was characterized by the detection frequencies of western harvest mice, house mice, and California voles. We standardized all variables prior to occupancy modeling.
We assessed each of these predictors using univariable models and assessed model performance using AICc (Burnham and Anderson 2002). We considered predictors to be biologically informative if their fit to the data was better (i.e., at least 2 AICc units lower) than that of a model with no predictor covariates (i.e., the “null model”). These predictors were selected as candidates for multivariable modeling. For predictors that were calculated at multiple scales (i.e., different buffer sizes for patch size, connectivity, and matrix urbanization), we included only the scale-variant with the lowest AICc. We first constructed a model that included all candidate variables and then applied a backward-stepwise approach (e.g., Hosmer and Lemeshow 2000) to determine the top multivariable model. Each step involved removal of each individual predictor from the model and accepting the model with n-1 predictors with the greatest improvement in AICc compared to the model with n predictors. When none of the models with n-1 predictors represented an improvement in AICc over the model with n predictors, we accepted the latter as the top multivariable model. We also constructed a model using patch size and connectivity as the only two predictors, which provided a tool to help evaluate the suitability of any given patch of habitat based on the assumptions of the area-isolation paradigm.
To determine predicted threshold values of spatial variables with respect to salt marsh harvest mouse occupancy, we used our univariable occupancy models to predict occupancy probabilities across a range of predictor values, then identified the predictor values that corresponded to occupancy probabilities of 0.50 and 0.95 (i.e., 50% and 95% chance of being occupied; Schultz and Crone 2005; Shake et al. 2012). We conducted this analysis for patch size, patch connectivity, and urbanization predictors. We also calculated the conditional probabilities of patch size and patch connectivity using the multivariable patch size + patch connectivity model; the former was assessed holding patch connectivity equal to zero (i.e., to estimate the relationship between patch size and occupancy of a completely isolated marsh), while the latter was assessed holding patch size equal to 1 ha (i.e., to estimate the relationship between patch connectivity and occupancy of a very small marsh).
Fine-scale occupancy modeling
To better understand microhabitat use by salt marsh harvest mice we estimated the effects of covariates on fine-scale (e.g., within-patch) occupancy and detection. To estimate fine-scale occupancy patterns, we used station-level data from patches where salt marsh harvest mice were detected. We tested the effects of tide height and survey interval on detection probability. We also tested the effects of vegetation richness, dominant vegetation species (a categorical dummy variable using terrestrial grasses as the reference, due to their association with marginal habitat), presence/absence of pickleweed, presence/absence of HTEV, presence/absence of terrestrial grasses, and distance from the patch perimeter on occupancy (Supplementary Table S2). We used mixed-effects modeling in the R package ‘ubms’ (Kellner et al. 2021) with site as a random intercept (to account for potential non-independence of stations within sites) and fixed effects for all detection and occupancy predictors. We assessed model performance using leave-one-out information criterion (LOOIC), which is analogous to AICc but is derived from the expected predicted log density (ELPD) rather than the log-likelihood (Kellner et al. 2021).
Local extinction modeling
To evaluate factors associated with local extirpation of salt marsh harvest mice, we surveyed museum collections to document marshes where this species was known to occur historically, and we treated these earlier documentations as representing a first “season” of data in a dynamic occupancy model (MacKenzie et al. 2003). We documented 14 geographically distinct capture locations between 1938 and 1959 (Supplementary Table S3), for which species identity was verified genetically (by sequencing a small fragment of cytochrome b; sequencing methods in Statham et al. 2016). We used the SFEI EcoAtlas Historical Baylands dataset to determine the boundaries and calculate patch size of historical marsh patches (SFEI 1998). We used our non-invasive survey data from modern patches that fell within the boundary of a historical marsh patch to represent a second “season”. If salt marsh harvest mice were detected at ≥ 1 modern patch(es) within the boundary of a historical marsh patch, it was considered occupied in the modern season.
To facilitate compatibility with our modern data set using two survey intervals, we assumed perfect detection and full occupancy of two survey intervals for historical marshes (i.e., first-season detection history of “11”, implying two survey attempts both resulting in detection). We applied dynamic occupancy modeling (MacKenzie et al. 2003) to estimate the effects of four predictors on extinction probability: (1) percentage of remaining marsh habitat within the extent of a historical marsh patch, (2) area of the largest remaining marsh patch within the extent of a historical marsh patch, (3) proportion of a historical marsh patch converted to modern urban land cover, and (4) area of a historical marsh patch (Supplementary Table S2). Next, we constructed multivariable models based on hypotheses that salt marsh harvest mouse extinction was associated with combined effects of (1) historical patch size and the size of the largest modern patch, (2) historical patch size and the percentage of remaining marsh, (3) historical patch size and percentage of modern urban land cover, (4) percentage of remaining marsh and percentage of modern urban land cover, and (5) percentage of remaining marsh and the size of the largest modern patch. All dynamic occupancy models were constructed using R package ‘unmarked’.
Results
Patch occupancy modeling
Non-invasive sampling detected salt marsh harvest mice at 24 of 47 patches (Fig. 1; Supplementary Table S1). The single session detection probability at the patch level (p) was 0.949. Neither tide height, survey effort, nor survey interval substantially improved detection probabilities over the null detection model (ΔAICc < 2; Supplementary Table S4), so all patch-occupancy models used the null detection probability. Patch size was best modeled with no buffer, although incorporation of a 50-m edge effect was similarly supported (ΔAICc < 2; Supplementary Table S5). Patch connectivity and matrix urbanization were most predictive at the 1-km buffer size.
The best-ranked univariable occupancy model was connectivity (i.e., p[.]Ψ[Connectivity]), followed by patch size (Table 1A). Additionally, four other univariable models outperformed the null occupancy model (ΔAICc > 2). Among univariable models, connectivity accounted for ~ 97.0% of AICc weight, followed by patch size, accounting for 2.4% of AICc weight. Occupancy probability was positively related to patch connectivity and patch size, and negatively to matrix urbanization, terrestrial grass habitat, detection frequency of western harvest mice, and vegetation richness. When these variables were incorporated into backward-stepwise multivariable models the top-scoring model related the probability of occupancy positively to patch size and connectivity, and negatively to terrestrial grass habitat (i.e., p[.]Ψ[Patch Size + Connectivity + Terrestrial Grasses]); β coefficients of all three predictors did not overlap zero (Table 2A).
Based on univariable occupancy models, predicted patch occupancy exceeded 0.50 under the following conditions: (a) patch size exceeded 25 ha (Fig. 2A; Supplementary Table S6), (b) patches were surrounded by > 16% marsh (i.e., within a 1 km buffer; Fig. 2B), and (c) the matrix was composed of < 48% urban land cover (Fig. 2C). Predicted occupancy exceeded 0.95 (a) when patch size was > 72 ha, or (b) when patches were surrounded by > 37% marsh, but even as little as 1% matrix urbanization precluded an occupancy estimate ≥ 0.95 (Fig. 2).
Effects of A patch size, B connectivity, and C matrix urbanization on salt marsh harvest mouse (Reithrodontomys raviventris) occupancy probability based on univariable models derived from non-invasive genetic survey data. Conditional effects of D patch size of a completely isolated marsh patch (0% marsh within 1 km) and E connectivity of a small (1 ha) marsh patch on occupancy probability estimated from a bivariable model. Dashed lines represent 95% CIs and vertical dotted lines represent thresholds of 0.50 and 0.95 occupancy probability
When we assessed the conditional probability of patch size on occupancy of a completely isolated marsh (e.g., using the p[.]Ψ[Patch Size + Connectivity] model with connectivity held at zero), patches of 72 and 128 ha corresponded to 0.50 and 0.95 occupancy probability, respectively (Fig. 2D; Supplementary Table S6). However, confidence intervals were wide, with the lower 95% limit < 0.06 probability of occupancy even for the largest patches (Fig. 2D). Assessing conditional probability of patch connectivity on occupancy of a very small marsh patch (e.g., using the p[.]Ψ[Patch Size + Connectivity] model with patch size held at 1 ha), connectivity values of 26% and 46% corresponded to 0.50 and 0.95 occupancy probability, respectively (Fig. 2E). In this case, confidence intervals were narrower, suggesting that connectivity conditioned on small patch size was a more consistent predictor of occupancy than patch size conditioned on patch isolation.
Fine-scale occupancy modeling
We detected salt marsh harvest mice at 150 of 314 stations occurring within marshes occupied by salt marsh harvest mice. Station-level detection probability with the null detection model was 0.684. The best detection model according to AICc included tide height as a covariate, which had a slightly positive effect on detection (Supplementary Table S4). Therefore, we used tide height as a detection predictor in all fine-scale occupancy models.
The best-ranked univariable fine-scale occupancy model associated occupancy positively with high tide escape vegetation (HTEV; i.e., the p[Tide Height]Ψ[HTEV] model) and accounted for > 99.9% of the LOOIC weight among univariable fine-scale models (Table 1B). HTEV was the only covariate that had a significant β coefficient in fine-scale occupancy models, although four other univariable models showed improvement over the null model (ΔAICc > 2). Within these models, all predictors had 95% CIs that overlapped zero; the presence of pickleweed had a moderately positive effect, the presence of terrestrial grasses had a moderately negative effect, vegetation richness had a small positive effect, and dominant vegetation categories had variable but statistically non-significant effects (Supplementary Table S7). Among dominant vegetation categories, the largest positive effect sizes tended to be taller, emergent plants such as gumplant and alkali bulrush (Bolboschoenus maritima), whereas the largest negative effect sizes tended to be lower-lying plants, such as alkali heath (Frankenia salina) and marsh jaumea (Jaumea carnosa). When these variables were incorporated into backward-stepwise multivariable models the top-scoring model related fine-scale occupancy probability positively to the presence of HTEV and pickleweed, and variably among dominant vegetation types (i.e., p[Tide Height]Ψ[HTEV + Pickleweed + Dominant Vegetation]; Table 2B; Supplementary Table S8). HTEV was the only predictor in this model with a 95% CI that did not overlap zero.
Local extinction modeling
We detected salt marsh harvest mice within the boundaries of 10 of 14 historically occupied marshes (Supplementary Table S3) and used multi-season occupancy modeling to identify predictors associated with marshes that had experienced extinction between the historical and modern “seasons”. All extinction models that included a predictor covariate improved AICc relative to the null model, although small sample size limited the precision of our models (Table 3). All 95% confidence intervals surrounding β estimates were wide and overlapped zero. The top model, ranked according to AICc, associated extinction probability negatively with the size of the largest remaining patch of marsh within the extent of a historical marsh patch (i.e., modern landscapes comprised of smaller patches were more likely to go extinct). The percentage of the historical marsh converted to urban land cover was positively associated with extinction probability, and the percentage of remaining marsh and the historical patch size were negatively associated with extinction probability. The top univariable model (p[.]Ψ[.]ε[Largest Remaining Marsh]) outperformed all multivariable models, although two multivariable models resulted in ∆AICc < 2 (Table 3). All three top models included the largest remaining patch size as a predictor; one model included the size of the historical marsh patch and another included the percentage of remaining marsh habitat within the extent of the historical marsh patch.
Discussion
Salt marsh harvest mice and the area-isolation paradigm
Patterns of occupancy by salt marsh harvest mice in the San Francisco Estuary conformed well to the area-isolation paradigm. Our findings that patch size and connectivity were the two most important predictors of salt marsh harvest mouse occupancy contrasts with findings from other studies that found within-patch habitat quality to be more important to habitat specialists than spatial characteristics of patches (Ye et al. 2013; Poniatowski et al. 2018). Rather, our work adds to recent studies suggesting connectivity drives patch occupancy for other mammalian habitat specialists (Gardiner et al. 2018; Zimbres et al. 2018). Connectivity was the strongest univariable predictor and the only predictor in the best-performing multivariable model with confidence intervals not overlapping zero. Salt marsh harvest mouse gene flow and movement is constrained by non-wetland habitat, such as open water and upland habitat (> 2 m elevation; Statham et al. 2022). Therefore, an interconnected network of marsh habitat may be particularly important for coping with extreme inundation events on fine temporal scales (e.g., to provide temporary refuge) and broad temporal scales (e.g., to facilitate recolonization and metapopulation dynamics). Patch size also influenced occupancy, which agrees with previous work suggesting that salt marsh harvest mice respond negatively to edge habitat (Bias and Morrison 2006; Marcot et al. 2020). The effect of urban land cover in the matrix had a sufficiently negative impact on salt marsh harvest mouse occupancy that any such habitat precluded our models from predicting ≥ 0.95 probability of occupancy. A meta-analysis of patch-occupancy studies found that characteristics of the matrix, rather than patch size and connectivity per se, were often stronger predictors of occupancy across numerous taxa with different life history traits (Prugh et al. 2008), including some habitat specialist rodents (Pita et al. 2007). Taken together, our results suggest a landscape of well-connected, large patches of marsh, with natural intervening matrix (e.g., grasslands, rather than urban land cover) represents optimal conditions for salt marsh harvest mouse persistence. Given the potential for sea level rise to change the marsh landscape in the San Francisco Estuary, our model predictions could be useful for exploring patch connectivity and occupancy under future climate scenarios.
Within-patch habitat variables
The influence of habitat on occupancy was scale-dependent. At the broader (patch-level) scale, terrestrial grass was the only habitat variable that had a statistically significant effect on salt marsh harvest mouse occupancy; in contrast, the only such variable at the finer (station-level) scale was high tide escape vegetation (HTEV). The failure of HTEV to emerge in broad-scale models likely reflected the presence of HTEV at many sites both with and without salt marsh harvest mice, suggesting that HTEV alone does not guarantee occupancy but that it is an important feature when other criteria for occupancy are met. Johnston (1957) observed salt marsh harvest mice hiding in tall gumplant bushes during extreme high tide events, and we observed multiple occasions of mice hiding in bait stations placed in gumplant and other tall emergent vegetation during surveys that aligned with high tides. Together, the importance of emergent high tide escape habitat at fine scales and patch connectivity at broad scales implies that refuge (HTEV) and rescue from dynamic tidal conditions (connectivity) are important drivers of long-term persistence, which may become increasingly important in light of rising sea levels (Thorne et al. 2018). Fisler (1965) hypothesized that intrusion of terrestrial grasses into marsh habitat favored the occupancy of sympatric generalist rodents, such as the western harvest mouse, over that of salt marsh harvest mice. Our findings support this hypothesis at the patch level but did not support fine-scale avoidance of terrestrial grasses.
Management implications
We evaluated thresholds of spatial parameters that corresponded to 0.50 and 0.95 occupancy probabilities in univariable models to provide some guidance for management (Fig. 2; Supplementary Table S6). However, these thresholds should be interpreted critically and applied to restoration projects on a case-by-case basis. For example, although the univariable patch size model estimated that 72 ha corresponded with 0.95 occupancy probability, if we assumed no connectivity among patches (i.e., using our conditional multivariable patch size + connectivity model), then 72 ha corresponded to just 0.50 occupancy probability, with a lower 95% CI of just 0.06. Broad uncertainty in our conditional model suggests that large patches do not guarantee salt marsh harvest mouse occupancy; rather, our conditional models suggest that small patch size effectively guarantees the absence of salt marsh harvest mice if not compensated by sufficient connectivity to nearby suitable habitat. Supporting this, a significant finding from our patch size + connectivity model was that even very small patches of marsh (e.g., 1 ha) can have high occupancy probability with relatively high confidence if they are well connected to nearby marsh habitat. Thus, our findings suggest that small scale restoration projects have a high probability of success if they are surrounded by a sufficient proportion of (occupied) marsh habitat, and that even large-scale restoration projects may have uncertain outcomes if they are isolated from other occupied marsh habitat.
Local extinction
Although the small number of sites with genetically verified historical presence of salt marsh harvest mice limited statistical power for our extinction models, we believe it is important that all predictors improved model performance relative to the null model. Moreover, the trends we observed were broadly consistent with those based on our contemporary occupancy models. Extinction models suggested that loss of large patches of habitat were associated with higher likelihood of extirpation. The top performing predictor was the size of the largest remaining patch of marsh habitat, which may imply that the four marshes we found to be likely extirpated had crossed below a minimum patch size threshold. Extinction modeling also suggested that larger historical marshes and marshes that retained a greater proportion of historical marsh habitat were less likely to become extirpated. With regard to modern efforts to conserve salt marsh harvest mouse populations, our results emphasize that smaller extant patches are more vulnerable to extinction.
Non-invasive monitoring of salt marsh harvest mice
Ongoing monitoring of salt marsh harvest mice should integrate regular surveys in small and isolated patches of habitat, which are likely the most vulnerable, but have not been regularly surveyed in the past (USFWS 2013). The non-invasive genetic survey approach we used in this study has the potential to significantly improve survey efficiency, but its efficacy had not been quantified prior to our study. Based on data presented in this study, bait station surveys were extremely effective at detecting salt marsh harvest mice at the patch level. Of note, we conducted the majority of our surveys in winter, and we do not yet know whether high summer temperatures would affect detection. Future work is needed to determine seasonal effects on detection probability using this approach, but our results from the cold season are promising.
Conclusions
Salt marsh harvest mouse occupancy conformed to the area-isolation paradigm of metapopulation theory. Specifically, larger patch size and greater patch connectivity were associated with higher occupancy probability. Our models predicted that small, well-connected marshes were more likely to be occupied than larger, isolated marshes. The abundance of upland vegetation in marsh patches negatively affected occupancy at broad scales, and the presence of high tide escape vegetation positively affected occupancy at fine scales. Furthermore, local extirpation at four historically occupied sites was associated with measures of habitat loss, particularly the loss of large patches of habitat. Our findings highlight important spatial and habitat considerations for future marsh preservation and restoration projects targeting salt marsh harvest mouse habitat.
References
Aylward CM, Grahn RA, Barthman-Thompson L, Kelt DA, Sacks BN, Statham MJ (2022) A novel non-invasive genetic survey technique for small mammals. J Mammal 103:1441–1447
Bias MA, Morrison ML (2006) Habitat selection of the salt marsh harvest mouse and sympatric rodent species. J Wildl Manag 70:732–742
Bollmann K, Graf RF, Suter W (2011) Quantitative predictions for patch occupancy of capercaillie in fragmented habitats. Ecography 34:276–286
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach. Springer, New York
CDFW [California Department of Fish and Wildlife] (1971) State and federally listed endangered and threatened animals of California. Accessed 20 April 2021
Diamond JM (1975) The island dilemma: Lessons of modern biogeographic studies for the design of natural reserves. Biol Conserv 7:129–146
Duggan JM, Schooley RL, Heske EJ (2011) Modeling occupancy dynamics of a rare species, Franklin’s ground squirrel, with limited data: are simple connectivity metrics adequate? Landsc Ecol 26:1477–1490
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Evol Syst 34:487–515
Fiske I, Chandler R (2011) unmarked: an R package for fitting hierarchical models of wildlife occurrence and abundance. J Stat Softw 43:1–23
Fisler GF (1965) Adaptations and speciation in harvest mice of the marshes of San Francisco Bay. Univ Calif Publ Zool 77:1–108
Fletcher RJ, Didham RK, Banks-Leite C, Barlow J, Ewers RM, Rosindell J, Holt RD, Gonzalez A, Pardini R, Damschen EI, Melo FPL, Ries L, Prevedello JA, Tscharntke T, Laurance WF, Lovejoy T, Haddad NM (2018) Is habitat fragmentation good for biodiversity? Biol Conserv 226:9–15
Gardiner R, Bain G, Hamer R, Jones ME, Johnson CN (2018) Habitat amount and quality, not patch size, determine persistence of a woodland-dependent mammal in an agricultural landscape. Landsc Ecol 33:1837–1849
Hadaway HC, Newman JR (1971) Differential responses of five species of salt marsh mammals to inundation. J Mammal 52:818–820
Haddad NM, Brudvig LA, Clobert J, Davies KF, Gonzalez A, Holt RD, Lovejoy TE, Sexton JO, Austin MP, Collins CD, Cook WM, Damschen EI, Ewers RM, Foster BL, Jenkins CN, King AJ, Laurance WF, Levey DJ, Margules CR, Melbourne BA, Nicholls AO, Orrock JL, Song DX, Townshend JR (2015) Habitat fragmentation and its lasting impact on Earth’s ecosystems. Sci Adv 1:e1500052
Hadley AS, Betts MG (2016) Refocusing habitat fragmentation research using lessons from the last decade. Curr Landsc Ecol Rep 1:55–66
Hanski I (1991) Single-species metapopulation dynamics: concepts, models, and observations. Biol J Lin Soc 42:17–38
Hanski I (1998) Metapopulation dynamics. Nature 396:41–49
Hanski I, Pakkala T, Kuussaari M, Lei G (1995) Metapopulation persistence of an endangered butterfly in a fragmented landscape. Oikos 72:21–28
Harrison S (1991) Local extinction in a metapopulation context: an empirical evaluation. Biol J Lin Soc 42:73–88
Hosmer DW, Lemeshow S (2000) Applied logistic regression, 2nd edn. Wiley, New York
Johnson V, Shellhammer HS (1988) The ecology of the salt marsh harvest mouse (Reithrodontomys raviventris) in a diked salt marsh and adjacent grass-lands in Palo Alto. California, US Fish and Wildlife Service, Sacramento
Johnston RF (1957) Adaptation of salt marsh mammals to high tides. J Mammal 38:529–531
Kellner KF, Fowler NL, Petroelje TR, Kautz TM, Beyer DE, Belant JL (2021) ubms: An R package for fitting hierarchical occupancy and N-mixture abundance models in a Bayesian framework. Methods Ecol Evol 13:577–584
Kerr JT, Cihlar J (2004) Patterns and causes of species endangerment in Canada. Ecol Appl 14:743753
Koper N, Schmiegelow FKA, Merrill EH (2007) Residuals cannot distinguish between ecological effects of habitat amount and fragmentation: implications for the debate. Landsc Ecol 22:811–820
Levins RA (1969) Some demographic and genetic consequences of environmental heterogeneity for biological control. Bull Entomol Soc Am 15:237–240
Liu Z, He C, Wu J (2016) The relationship between habitat loss and fragmentation during urbanization: an empirical evaluation from 16 world cities. PLoS ONE 11:e0154613
MacArthur RH, Wilson EO (1967) The theory of island biogeography. Princeton University Press, Princeton, p 203
MacKenzie DI, Nichols JD, Lachman GB, Droege S, Royle JA, Langtimm CA (2002) Estimating site occupancy rates when detection probabilities are less than one. Ecology 83:2248–2255
MacKenzie DI, Nichols JD, Hines JE, Knutson MG, Franklin AB (2003) Estimating site occupancy, colonization, and extinction when a species is detected imperfectly. Ecology 84:2200–2207
Marcot BG, Woo I, Thorne KM, Freeman CM, Guntenspergen GR (2020) Habitat of the endangered salt marsh harvest mouse (Reithrodontomys raviventris) in San Francisco Bay. Ecol Evol 10:662–677
Pita R, Beja P, Mira A (2007) Spatial population structure of the Cabrera vole in Mediterranean farmland: the relative role of patch and matrix effects. Biol Conserv 134:383–392
Poniatowski D, Stuhldreher G, Löffler F, Fartmann T (2018) Patch occupancy of grassland specialists: habitat quality matters more than habitat connectivity. Biol Conserv 225:237–244
Prugh LR, Hodges KE, Sinclair ARE, Brashares JS (2008) Effect of habitat area and isolation on fragmented animal populations. Proc Natl Acad Sci 105:20770–20775
Schultz CB, Crone EE (2005) Patch size and connectivity thresholds for butterfly habitat restoration. Conserv Biol 19:887–896
SFEI [San Francisco Estuary Institute] (1998) Bay Area EcoAtlas v1.50b4 1998: Geographic Information System of wetland habitats past and present. http://www.sfei.org/content/ecoatlas-version-150b4-1998
Shake CS, Moorman CE, Riddle JD, Burchell MR (2012) Influence of patch size and shape on occupancy by shrubland birds. Condor 114:268–278
Shellhammer HS, Duke RR (2010) Salt marsh harvest mice and width of salt marshes in the south San Francisco Bay. Calif Fish Game 96:165–170
Smith KR, Barthman-Thompson L, Gould WR, Mabry KE (2014) Effects of natural and anthropogenic change on habitat use and movement of endangered salt marsh harvest mice. PLoS ONE 9:e3108739
Smith KR, Riley MK, Barthman-Thompson L, Woo I, Statham MJ, Estrella S, Kelt DA (2018a) Toward salt marsh harvest mouse recovery: a review. San Francisco Estuary Watershed Sci 16:1–24
Smith KR, Riley MK, Barthman-Thomspon L, Statham MJ, Estrella S, Kelt DA (2018b) Toward salt marsh harvest mouse recovery: research priorities. San Francisco Estuary Watershed Sci 16:1–6
Statham MJ, Aamoth S, Barthman-Thompson L, Estrella S, Fresquez S, Hernandez LD, Tertes R, Sacks BN (2016) Conservation genetics of the endangered San Francisco Bay endemic salt marsh harvest mouse. Conserv Genet 17:1055–1066
Statham MJ, Aylward CM, Barthman-Thompson L, Kierepka EM, Sacks BN (2022) Landscape genetics of an endangered salt marsh endemic: identifying population continuity and barriers to dispersal. Online Early. https://doi.org/10.1007/s10592-022-01446-6
Sustaita D, Quickert PF, Patterson L, Barthman-Thompson L, Estrella S (2011) Salt marsh harvest mouse demography and habitat use in the Suisun Marsh, California. J Wildl Manag 75:1498–1507
Thorne K, MacDonald G, Guntenspergen G, Ambrose R, Buffington K, Dugger B, Freeman C, Janousek C, Brown L, Rosencranz J, Holmquist J, Smol J, Hargan K, Takekawa J (2018) US Pacific coastal wetland resilience and vulnerability to sea-level rise. Sci Adv 4:ea003270
USFWS [U.S. Fish and Wildlife Service] (1970) United States list of endangered native fish and wildlife. Fed Reg 35:16047–16048
USFWS [U.S. Fish and Wildlife Service] (2013) Recovery plan for tidal marsh ecosystems of Northern and Central California. U.S. Fish and Wildlife Service, Sacramento
Wahlberg N, Miolanen A, Hanski I (1996) Predicting the occurrence of endangered species in fragmented landscapes. Science 273:1536–1538
Williams P, Faber P (2001) Salt marsh restoration experience in San Francisco Bay. J Coastal Res 23:203–211
Ye X, Skidmore AK, Wang T (2013) Within-patch habitat quality determines the resilience of specialist species in fragmented landscapes. Landsc Ecol 28:135–147
Zimbres B, Peres CA, Penido G, Machado RB (2018) Thresholds of riparian forest use by terrestrial mammals in a fragmented Amazonian deforestation frontier. Biodivers Conserv 27:2815–2836
Acknowledgements
We thank John Krause (California Department of Fish and Wildlife; CDFW), Karen Taylor (CDFW), Terris Kasteen (CDFW), Orlando Rocha (CDFW), Dave Reinsche (East Bay Regional Park District), Rachel Tertes (United States Fish and Wildlife Service), Lisa Myers (Palo Alto Baylands Nature Preserve), Veronica Pearson (Marin County Parks), Julian Skinner (City of Larkspur), Craig Murray (City of Richmond), and Barbara Salzman (Marin Audubon Society) for access to field sites. This work was funded by the California Department of Water Resources (contract 4600011551 to DAK and 4600012662 to MJS), the USDA National Institute of Food and Agriculture (Hatch project CA-D-WFB-6126-H to DAK), and a Christine Stevens Wildlife Award to CMA. We thank the Museum of Vertebrate Zoology (University of California, Berkeley), Natural History Museum of Los Angeles County, and the UC Davis Museum of Wildlife and Fish Biology for providing historical museum specimens.
Funding
This work was funded by the California Department of Water Resources (contract 4600011551 to Douglas A. Kelt and 4600012662 to Mark Statham), the USDA National Institute of Food and Agriculture (Hatch project CA-D-WFB-6126-H to Douglas A. Kelt), and a Christine Stevens Wildlife Award to Cody Aylward.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Data collection was performed by CMA, MJS, and LB-T. Data analysis was performed by CMA. The first draft of the manuscript was written by CMA and all authors commented on drafts of the manuscript. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Open research statement
Data and code are available from the authors upon request.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Appendix 1
Appendix 1
The following museum specimens were used to verify the historical presence of salt marsh harvest mice. Museum of Vertebrate Zoology (University of California, Berkeley): MVZ:MAMM:101731, MVZ:MAMM:101732, MVZ:MAMM:101733, MVZ:MAMM:102484, MVZ:MAMM:102485, MVZ:MAMM:104412, MVZ:MAMM:109726, MVZ:MAMM:109731, MVZ:MAMM:109732, MVZ:MAMM:113406, MVZ:MAMM:121096, MVZ:MAMM:121097, MVZ:MAMM:124117, MVZ:MAMM:124121, MVZ:MAMM:124123, MVZ:MAMM:124137, MVZ:MAMM:124296, MVZ:MAMM:124423, MVZ:MAMM:126083, MVZ:MAMM:126088, MVZ:MAMM:128656, MVZ:MAMM:130008, MVZ:MAMM:130010, MVZ:MAMM:130013, MVZ:MAMM:130014, MVZ:MAMM:130020, MVZ:MAMM:130023, MVZ:MAMM:130030, MVZ:MAMM:130042, MVZ:MAMM:130044, MVZ:MAMM:130048, MVZ:MAMM:130063, MVZ:MAMM:130064, MVZ:MAMM:130065, MVZ:MAMM:130067, MVZ:MAMM:130069, MVZ:MAMM:130071, MVZ:MAMM:130074, MVZ:MAMM:130077, MVZ:MAMM:130078, MVZ:MAMM:130079, MVZ:MAMM:130082, MVZ:MAMM:130083, MVZ:MAMM:130084, MVZ:MAMM:130087, MVZ:MAMM:130088, MVZ:MAMM:130092, MVZ:MAMM:130096, MVZ:MAMM:135350, MVZ:MAMM:135351, MVZ:MAMM:80671, MVZ:MAMM:80673, MVZ:MAMM:80674, MVZ:MAMM:80833, MVZ:MAMM:80836, MVZ:MAMM:80837, MVZ:MAMM:80838, MVZ:MAMM:80839, MVZ:MAMM:81295, MVZ:MAMM:81298, MVZ:MAMM:81300, MVZ:MAMM:81301, MVZ:MAMM:81302, MVZ:MAMM:81303, MVZ:MAMM:87901, MVZ:MAMM:87902, MVZ:MAMM:87903, MVZ:MAMM:87904, MVZ:MAMM:94752, MVZ:MAMM:94753, MVZ:MAMM:94754, MVZ:MAMM:94834, MVZ:MAMM:96241, MVZ:MAMM:96242, MVZ:MAMM:96243.
Natural History Museum of Los Angeles County: LACM:Mamm:5219, LACM:Mamm:5220, LACM:Mamm:5221, LACM:Mamm:5222, LACM:Mamm:5223, LACM:Mamm:5224, LACM:Mamm:5225, LACM:Mamm:5226, LACM:Mamm:5227, LACM:Mamm:5228, LACM:Mamm:5229, LACM:Mamm:5230, LACM:Mamm:5231, LACM:Mamm:5232, LACM:Mamm:5233, LACM:Mamm:5234, LACM:Mamm:5235, LACM:Mamm:5252, LACM:Mamm:5253, LACM:Mamm:5254, LACM:Mamm:5255, LACM:Mamm:5256, LACM:Mamm:5257, LACM:Mamm:5258, LACM:Mamm:5259, LACM:Mamm:5260, LACM:Mamm:5261, LACM:Mamm:5264, LACM:Mamm:5265, LACM:Mamm:5266, LACM:Mamm:5271, LACM:Mamm:5272, LACM:Mamm:5273, LACM:Mamm:5274, LACM:Mamm:5275, LACM:Mamm:5276, LACM:Mamm:5277, LACM:Mamm:5278, LACM:Mamm:5279, LACM:Mamm:5280, LACM:Mamm:5293, LACM:Mamm:5294, LACM:Mamm:5295, LACM:Mamm:5296, LACM:Mamm:5297, LACM:Mamm:5298, LACM:Mamm:5299, LACM:Mamm:5301, LACM:Mamm:5302, LACM:Mamm:5303, LACM:Mamm:27653, LACM:Mamm:27656, LACM:Mamm:27658, LACM:Mamm:27659, LACM:Mamm:27660, LACM:Mamm:27662, LACM:Mamm:27663, LACM:Mamm:27666, LACM:Mamm:27667, LACM:Mamm:27668, LACM:Mamm:27806, LACM:Mamm:27807, LACM:Mamm:27808, LACM:Mamm:27809, LACM:Mamm:27810, LACM:Mamm:27811, LACM:Mamm:27813, LACM:Mamm:27814, LACM:Mamm:27815, LACM:Mamm:27816.
UC Davis Museum of Wildlife and Fish Biology: MWFB-1614Z, MWFB-1616Z, MWFB-5607Z, MWFB-4512Z, MWFB-2781, MWFB-2782, MWFB-3465, MWFB-3466, MWFB-4542, MWFB-4693.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Aylward, C.M., Barthman-Thompson, L., Bean, W.T. et al. Patch size and connectivity predict remnant habitat occupancy by an endangered wetland specialist, the salt marsh harvest mouse. Landsc Ecol 38, 2053–2067 (2023). https://doi.org/10.1007/s10980-023-01683-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-023-01683-1