Abstract
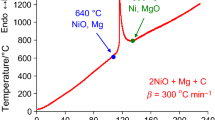
The most popular method for production of metallic uranium is by magnesiothermic reduction of UF4. Efficient production of good quality uranium metal chunk in the reaction vessel requires multiple parameter constraints. To theoretically optimize these process parameters, many basic properties, i.e. particle size, heat dissipation, packing density, heating rate, kinetic parameters of reaction, etc., are required. There was lack of information on kinetic parameters of this reaction; therefore, kinetic triplets of UF4 and magnesium metal reaction were determined by non-isothermal DTA method, at 5–20 K min−1 heating rates. The analysis of data was done by model-less methods and model-fitting methods. It was found that the reaction takes place through two parallel competitive mechanisms, bulk diffusion and first-order nucleation and growth of reaction sites, and the corresponding kinetic parameters, frequency factors and activation energies, were 440 min−1, 43.9 kJ mol−1, 420 min−1 and 49.03 kJ mol−1, respectively.










Similar content being viewed by others
References
Nininger RD. Minerals of atomic energy. 2nd ed. Princeton: D Van Nostrand Co., Inc.; 1956.
Harper J, Williams AE. Extraction and refining of rarer metals. London: The Institute of Mining and Metallurgy; 1957. p. 143–62.
Katz JJ, Rabinowitch E. Part 1—the elements, its binary and related compounds. The Chemistry of Uranium. New York: McGraw-Hill; 1951.
Barin I. Thermochemical data of pure substances. Weinheim: VCH Verlagsgesellschaft mbH; 1995.
Manna S, Chowdhury S, Satpati SK, Roy SB. Magnesio thermic reduction (MTR) reaction an study of relative importance of the dependent parameters using artificial neural network. Indian Chem Eng. 2007;49(3):221–32.
Chiotti P, Shoemaker HE. Nuclear technology in the chemical and petroleum industries pyrometallurgical separation of uranium from thorium. Ind Eng Chem. 1958;50(2):137–40.
Sutar H, Sahoo A. Computational simulation of unsteady state heat transfer in externally heated magnesio thermic reduction reactor; an overview (CSUHEMTRR). Int J Chem Eng Appl. 2011;2(3):212–5.
Saliba-Silva AM, Aguiar B, Carvalho EU, Da Silva DG, Gracher Riella H, Durazzo M. Analysis of slag formation during UF4 magnesiothermic reduction. In: RERTR 2011—33rd international meeting on reduced enrichment for research and test reactors, Santiago, Chile, October 23–27, 2011.
Paine RM, Ruehle AE, Lewis GW. USAEC Report, NYO-1335 (1957).
Domingues JTH. The kinetics of the reduction of uranium fluoride by magnesium. Ph.D. thesis, University of London, London (May 1964).
Vyazovkin S, Chrissafis K, Lorenzo MD, Koga N, Pijolat M, Roduit B, Sbirrazzouli N, Sunol JJ. ICTAC kinetics committee recommendations for collecting experimental thermal analysis data for kinetic computations. Thermochim Acta. 2014;590:1–23.
PDF Card No. 35-0821 (Mg), International Centre for Diffraction Data, Newtown Square, USA.
PDF Card No. 71-1962 (UF4), International Centre for Diffraction Data, Newtown Square, USA.
PDF Card No. 73-2293 (UO2), International Centre for Diffraction Data, Newtown Square, USA.
PDF Card No. 75-0944 (U4O9), International Centre for Diffraction Data, Newtown Square, USA.
PDF Card No. 74-2103 (U3O8), International Centre for Diffraction Data, Newtown Square, USA.
PDF Card No. 11-0628 (U), International Centre for Diffraction Data, Newtown Square, USA.
PDF Card No. 16-0160 (MgF2), International Centre for Diffraction Data, Newtown Square, USA.
Scott KY, Harrison KT. Some studies of oxidation of uranium dioxide. J Nucl Mater. 1963;8(3):307–19.
Tempest PA, Tucker PM, Tyler JW. Oxidation of UO2 fuel pellets in air at 503 and 543 K studied using X-ray photoelectron spectroscopy and X-ray diffraction. J Nucl Mater. 1988;151:269–74.
Campbell TK, Gilbert ER, White GD, Plepel GF, Wrona BJ. Oxidation behaviour of nonirradiated UO2. Nucl Technol. 1989;85(2):160–71.
McEachern RJ, Taylor P. A review of the oxidation of uranium dioxide at temperatures below 400 °C. AECL-11335, COG-95-281-1, Whiteshell Laboratories, Pinawa, Manitoba R0E 1L0; 1997.
Friedman H. Kinetics of thermal degradation of char-forming plastics from thermogravimetry. Application to a phenolic plastic. J Polym Sci C. 1964;6(1):183–95.
Kissinger HE. Reaction kinetics in differential thermal analysis. Anal Chem. 1957;29(11):1702–6.
Akahira T, Sunose T. Method of determining activation deterioration constant of electrical insulating materials. Res Rep CHIBA Inst Technol. 1971;16:22–31.
Flynn JH, Wall LA. A quick, direct method for the determination of activation energy from thermogravimetric data. J Polym Sci B Polym Lett. 1966;4:323–8.
Ozawa T. A new method of analyzing thermogravimetric data. Bull Chem Soc Jpn. 1965;38:1881.
Starink MJ. The determination of activation energy from linear heating rate experiments: a comparison of the accuracy of isoconversion methods. Thermochim Acta. 2003;404:163–76.
Starink MJ. A new method for the derivation of activation energies from experiments performed at constant heating rate. Thermochim Acta. 1996;288:97–104.
Vyazovkin S, Burnham AK, Criado JM, Perez-Maqueda LA, Popescu C, Sbirrazzuoli N. ICTAC kinetic committee recommendations for performing kinetic computations on thermal analysis data. Thermochim Acta. 2011;520:1–19.
Fan R-H, Lu H-L, Sun K-N, Wang W-X, Yi X-B. Kinetics of thermite reaction in Al-Fe2O3 system. Thermochim Acta. 2006;440:129–31.
Guida MY, Bouaik H, El Mouden L, Moubarik A, Aboulkas A. El harfi K and Hannioui A, Utilization of Starink approach and Avrami theory to evaluate the kinetic parameters of the pyrolysis of olive mill solid waste and olive mill wastewater. J Adv Chem Eng. 2016;7(1):10. https://doi.org/10.4172/2090-4568.1000155.
Farjas J, Butchosa N, Roura P. A simple kinetic method for the determination of the reaction model from non-isothermal experiments. J Therm Anal Calorim. 2010;102:615–25.
Malek J. The kinetic analysis of non-isothermal data. Thermochim Acta. 1992;200:257–69.
Aboulkas A, El Harfi K. Study of the kinetics and mechanisms of thermal decomposition of Moroccan tarfaya oil shale and its kerogen. Oil Shale. 2008;25(4):426–43.
Heydari M, Rahman M, Gupta R. Kinetic study and thermal decomposition behaviour of lignite coal. Int J Chem Eng 2015, Article ID 481739.
Coats AW, Redfern JP. Kinetic parameters from thermogravimetric data II. J Polym Sci B Polym Lett. 1965;3:917–20.
Coats AW, Redfern JP. Kinetic parameters from thermogravimetric data. Nature. 1964;201:68–9.
Pomerantsev AL, Kutsenova AL, Rodionova OY. Kinetic analysis of non-isothermal solid-state reactions: multistage modeling without assumptions in the reaction mechanism. Phys Chem Chem Phys. 2017;19:3606–15.
Perez-Maqueda LA, Criado JM, Sanchez-Jimenez PE. Combined kinetic analysis of solid-state reactions: a powerful tool for the simultaneous determination of kinetic parameters and the kinetic model without previous assumptions on the reaction mechanism. J Phys Chem A. 2006;110:12456–62.
Colmenares CA. The oxidation of thorium, uranium, and plutonium. Prog Solid State Chem. 1975;9:139–239.
Schnizlein JG, Woods JD, Bingle JD, Vogel RC. Identification of the diffusing species in uranium oxidation. J Electrochem Soc. 1960;107:783–5.
Olander DR. The chemical diffusivity of UO2−x. J Nucl Mater. 1982;110:352.
Lidiard A. Self diffusion of uranium in UO2. J Nucl Mater. 1966;19(1):106–8.
Tomlinson L, Morrow SA, Graves S. Kinetics of the hydrofluorination of uranium oxide. Trans Faraday Soc. 1961;57:1008–18.
Sinitsyn VV, Lips O, Privalov AF, Fusjara F, Murin IV. Transport properties of LaF3 fast ionic conductor studied by field gradient NMR and impedance spectroscopy. J Phys Chem Solids. 2003;64(7):1201–5.
Sorokin NI, Karimov DN, Sobolev BP. Anisotropy of ionic conductivity of TbF3 crystals. Crystallogr Rep. 2019;64(4):621–5.
Labaton VY, Johnson KDB. The fluorides of uranium—III kinetic studies of the fluorination of uranium tetrafluoride by flourine. J Inorg Nucl Chem. 1959;10(1):74–85.
Labaton VY. The fluorides of uranium—IV Kinetic studies of the fluorination of uranium tetrafluoride by chlorine trifluoride. J Inorg Nucl Chem. 1959;10(1):86–93.
Kanellakopulos B, Parthey H. The oxidation of uranium tetrachloride. J Inorg Nucl Chem. 1959;28:2541–9.
Quesnel E, Dumas L, Jacob D, Peiro F. Optical and microstructural properties of MgF2 UV coatings grown by ion beam sputtering process. J Vac Sci Technol A. 2000;18(6):2869–76.
Catlow CRA, James R, Norgett MJ. The defect structure of compounds with rutile structure. J Phys Colloq. 1976;37(C7):443–8.
Milicka K, Cadek J, Rys P. High temperature creep mechanism in magnesium. Acta Metall. 1970;18:1071–82.
Khawam A, Flanagan DR. Solid-state kinetic models: basics and mathematical fundamentals. J Phys Chem B. 2006;110:17315–28.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Agarwal, R., Raina, A. & Gupta, N.K. Magnesiothermic reduction kinetics of UF4. J Therm Anal Calorim 143, 3353–3363 (2021). https://doi.org/10.1007/s10973-020-10359-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-020-10359-w