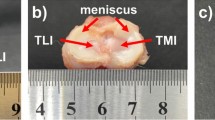
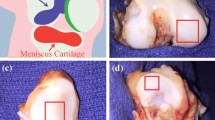
Abstract
This paper presents the results of a thermo-analytical study of the thermal expansion of rib cartilage used for autotransplantation. Deformations of rib cartilage implants caused by thermal expansion of tissue during Peltier cooling and laser heating in the temperature range from − 10 to + 40 °C. It is revealed that thermal expansion of the cartilage is influenced by the anisotropy of the internal structure of the tissue. Samples of rib cartilage, cut along the fiber bundle, are deformed four times stronger in length than in width. The anisotropy of the cartilaginous tissue structure affects the rate of heating of the samples at different conditions of heat transfer. Thus, the samples cut from the rib in two different ways, along or across the fiber bundle, have different heating rates and duration of the phase transition. Laser heating rate of frozen samples cut across the fiber bundle is 20% higher than along cut. Duration of ice melting for both types of the samples differs by 25%. These studies are important for planning operations of laser reshaping of cartilaginous tissue and for the preliminary selection of frozen implants.








Similar content being viewed by others
References
Thmford WW, Springfield DS, Mankin HJ. Fresh and frozen articular cartilage allografts. Orthopedics. 1992;15(10):1183–8.
Csönge L, Bravo D, Newman-Gage H, et al. Banking of osteochondral allografts, Part II. Preservation of chondrocyte viability during long-term storage. Cell Tissue Bank. 2002. https://doi.org/10.1023/A:1023687419152.
Helidonis E, Sobol E, Kavvalos G, Bizakis J, Christodoulou P, Velegrakis G, Bagratashvili V. Laser shaping of composite cartilage grafts. Am J Otolaryngol. 1993. https://doi.org/10.1016/0196-0709(93)90115-N.
Bagratashvili VN, Sobol EN, Sviridov AP, Popov VK, Omel’chenko AI, Howdle SM. Thermal and diffusion processes in laser-induced stress relaxation and reshaping of cartilage. J Biomech. 1997. https://doi.org/10.1016/S0021-9290(97)00028-6.
Changoor A, Fereydoonzad L, Yaroshinsky A, Buschmann MD. Effects of refrigeration and freezing on the electromechanical and biomechanical properties of articular cartilage. ASME J Biomech Eng. 2010. https://doi.org/10.1115/1.4000991.
Patczai B, Mintál T, Nőt LG, Wiegand N, Lőrinczy D. Effects of deep-freezing and storage time on human femoral cartilage. J Therm Anal Calorim. 2017. https://doi.org/10.1007/s10973-016-5365-2.
Assenmacher AT, Pareek A, Reardon PJ, Macalena JA, Stuart MJ, Krych AJ. Long-term outcomes after osteochondral allograft: a systematic review at long-term follow-up of 12.3 years. Arthrosc. 2016. https://doi.org/10.1016/j.arthro.2016.04.020.
Gracitelli GC, Moraes VY, Franciozi CES, Luzo MV, Belloti JC. Surgical interventions (microfracture, drilling, mosaicplasty and allograft transplantation) for treating isolated cartilage defects of the knee in adults. Cochrane Database Syst Rev. 2013. https://doi.org/10.1002/14651858.CD010675.
Baum OI, Soshnikova YM, Sobol EN, Korneychuk AY, Obrezkova MV, Svistushkin VM, Lunin VV. Laser reshaping of costal cartilage for transplantation. Lasers Surg Med. 2011. https://doi.org/10.1002/lsm.21077.
Baum OI, Alexandrovskaya YM, Svistushkin VM, Starostina SV, Sobol EN. New clinical application of laser correction of cartilage shape for implantation in otolaryngology. Laser Phys Lett. 2019. https://doi.org/10.1088/1612-202X/aafd21.
Sobol E, Vorobieva N, Baum O, Shekhter A, Guller A. Is it possible to perform laser reshaping without dramatic effect on chondrocytes? Lasers Surg Med. 2011;43:911–2.
Borodina K, Sorokina S, Blinova N, Boronenko M, Gulyaev P. Thermal analysis of reaction producing KXTiO2. J Therm Anal Calorim. 2018. https://doi.org/10.1007/s10973-017-6840-0.
Gulyaev PY, Kotvanova MK, Omelchenko AI. Nanotechnologies of the treatment and production of complex transition metal oxides with high photothermal effect. Inorg Mater Appl Res. 2018. https://doi.org/10.1134/S2075113318030140.
Soshnikova YM, Roman SG, Chebotareva NA, Baum OI, Obrezkova MV, Gillis RB, Harding SE, Sobol EN, Lunin VV. Starch-modified magnetite nanoparticles for impregnation into cartilage. J Nanopart Res. 2013. https://doi.org/10.1007/s11051-013-2092-5.
Farkas T, Bihari-Varga M, Biró T. Thermoanalytical and histological study of intra-articular papain-induced degradation and repair of rabbit cartilage. II. Mature animals. Ann Rheum Dis. 1976;35(1):23–6.
Toth K, Sohar G, Pallagi E, Szabo-Revesz P. Further characterization of degenerated human cartilage with differential scanning calorimetry. Thermochim Acta. 2007. https://doi.org/10.1016/j.tca.2007.07.013.
Sohar G, Szabo-Revesz P, Toth K, Aigner Z. Characterization of live and experimentally degenerated hyaline cartilage with thermal analysis. In: Qian C, editor. Osteoarthritis—diagnosis, treatment and surgery. London: InTech; 2012. p. 27–47.
Hemminger W, Höhne G. Calorimetry: fundamentals and practice. Basel: Vch; 1984.
Chae YS, Aguilar G, Lavernia EJ, Wong BJF. Characterization of temperature dependent mechanical behavior of cartilage. Lasers Surg Med. 2003. https://doi.org/10.1002/lsm.10167.
Soshnikova YM, Sobol EN, Baum OI, Keselman MM, Shults EV, Obrezkova MV, Lunin VV. Effect of anisotropy and drying of costal cartilage on its optical transmittance in laser reshaping of implants with 1, 2, and 3 mm in thickness. Lasers Surg Med. 2016. https://doi.org/10.1002/lsm.22575.
Bagratashvili VN, Sobol EN, Shekhter AB. Lasernaya engeneria khrashey. M. FizMatLit; 2006 (in Russian).
Lide David R, editor. Handbook of chemistry and physics. 84th ed. Boca Raton: CRC Press; 2003.
Maroudas A, Schneiderman R. “Free” and “exchangeable” or “trapped” and “non-exchangeable” water in cartilage. J Orthop Res. 1987. https://doi.org/10.1002/jor.1100050117.
Torzilli PA, Rose DE, Dethmers DA. Equilibrium water partition in articular cartilage. Biorheology. 1982. https://doi.org/10.3233/BIR-1982-19404.
Acknowledgements
This work was supported by the Ministry of Science and Higher Education of Russian Federation within the State assignment FSRC «Crystallography and Photonics» RAS in part “finding optimal regimes of laser action on cartilaginous tissue” and by the Russian Foundation for Basic Research (RFBR) (Project No. 18-29-02124) in part of “investigation of thermomechanical the behavior of cartilaginous tissue, taking into account its anisotropy with and without laser exposure.”
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kasianenko, E., Nushtaeva, R., Omelchenko, A. et al. Thermal expansion of rib cartilage implants at the non-isothermal cooling and heating. J Therm Anal Calorim 139, 3519–3526 (2020). https://doi.org/10.1007/s10973-019-08695-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-019-08695-7