Abstract
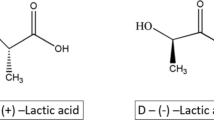
In this study, the hydrolytic and thermal oxidative degradation behavior of thermotropic aromatic–aliphatic copolyesters based on 4-hydroxybenzoic acid (HBA), 4-hydroxy-3-methoxybenzoic acid (VA), poly(butylene terephthalate) (PBT) and lactic acid (LA) have been investigated. The hydrophilicity and alkaline hydrolytic degradation rate of copolyesters increased with increasing LA contents. FTIR, DSC and TG results suggested that breakage of molecular chain mainly took place on aliphatic segments and also amorphous part. SEM analysis indicated the hydrolytic degradation followed surface corrosion mechanism. The dynamic TG results showed that all copolyesters exhibit total mass loss and two degradation stages under air atmosphere. The thermal oxidative degradation kinetics were evaluated by Kissinger and KAS method. The dependence of E values on conversion α has been observed in selected conversion range for all copolyesters, indicating complex degradation reaction existed.







Similar content being viewed by others
References
Inkinen S, Hakkarainen M, Albertsson A-C, Södergård A. From lactic acid to poly(lactic acid) (PLA): characterization and analysis of PLA and its precursors. Biomacromolecules. 2011;12(3):523–32.
Abebe DG, Kandil R, Kraus T, Elsayed M, Merkel OM, Fujiwara T. Three-layered biodegradable micelles prepared by two-step self-assembly of PLA–PEI–PLA and PLA–PEG–PLA triblock copolymers as efficient gene delivery system. Macromol Biosci. 2015;15(5):698–711.
Tabata Y, Abe H. Synthesis and properties of alternating copolymers of 3-hydroxybutyrate and lactate units with different stereocompositions. Macromolecules. 2014;47(21):7354–61.
Che H-L, Lee HJ, Uto K, Ebara M, Kim WJ, Aoyagi T, et al. Simultaneous drug and gene delivery from the biodegradable poly(-caprolactone) nanofibers for the treatment of liver cancer. J Nanosci Nanotechnol. 2015;15(10):7971–5.
Yu H, Yan C, Yao J. Fully biodegradable food packaging materials based on functionalized cellulose nanocrystals/poly(3-hydroxybutyrate-co-3-hydroxyvalerate) nanocomposites. RSC Adv. 2014;4(104):59792–802.
Felfel RM, Leander P, Miquel G-F, Tobias M, Gerhard H, Ifty A, et al. In vitro degradation and mechanical properties of PLA–PCL copolymer unit cell scaffolds generated by two-photon polymerization. Biomed Mater. 2016;11(1):015011.
Ortega-Toro R, Contreras J, Talens P, Chiralt A. Physical and structural properties and thermal behaviour of starch-poly(ɛ-caprolactone) blend films for food packaging. Food Packag Shelf Life. 2015;5:10–20.
Lavilla C, Alla A, Martínez de Ilarduya A, Benito E, García-Martín MG, Galbis JA, et al. Bio-based poly(butylene terephthalate) copolyesters containing bicyclic diacetalized galactitol and galactaric acid: influence of composition on properties. Polymer. 2012;53(16):3432–45.
Montes de Oca H, Wilson JE, Penrose A, Langton DM, Dagger AC, Anderson M, et al. Liquid-crystalline aromatic–aliphatic copolyester bioresorbable polymers. Biomaterials. 2010;31(30):7599–605.
Du J, Fang Y, Zheng Y. Synthesis, characterization and biodegradation of biodegradable-cum-photoactive liquid-crystalline copolyesters derived from ferulic acid. Polymer. 2007;48(19):5541–7.
Chen Y, Wombacher R, Wendorff JH, Visjager J, Smith P, Greiner A. Design, synthesis, and properties of new biodegradable aromatic/aliphatic liquid crystalline copolyesters. Biomacromolecules. 2003;4(4):974–80.
Prasad VS, Pillai CKS. Synthesis, characterization, and in vitro degradation of liquid-crystalline terpolyesters of 4-hydroxyphenylacetic acid/3-(4-hydroxyphenyl)propionic acid with terephthalic acid and 2,6-naphthalene diol. J Polym Sci Part A Polym Chem. 2002;40(11):1845–57.
Wei P, Wang L, Huang S, Wang X, Chen Y, Wang Y, et al. Synthesis and characterization of novel thermotropic aromatic–aliphatic biodegradable copolyesters containing d,l-lactic acid (LA), poly(butylene terephthalate) (PBT) and biomesogenic units. Polym Plast Technol Eng. 2014;53(16):1697–705.
Chao G, Fan L, Jia W, Qian Z, Gu Y, Liu C, et al. Synthesis, characterization and hydrolytic degradation of degradable poly(butylene terephthalate)/poly(ethylene glycol) (PBT/PEG) copolymers. J Mater Sci Mater Med. 2007;18(3):449–55.
Kullyakool S, Danvirutai C, Siriwong K, Noisong P. Determination of kinetic triplet of the synthesized Ni3(PO4)2·8H2O by non-isothermal and isothermal kinetic methods. J Therm Anal Calorim. 2014;115(2):1497–507.
Wei P, Wang L, Wang X, Chen Y, Wang Y, Wang Y. Nonisothermal and isothermal oxidative degradation behavior of thermotropic liquid crystal polyesters containing kinked bisphenol AF and bisphenol A units. High Perform Polym. 2014;26(8):935–45.
Wei P, Cakmak M, Chen Y, Wang X, Wang Y, Wang Y. The influence of bisphenol AF unit on thermal behavior of thermotropic liquid crystal copolyesters. Thermochim Acta. 2014;586:45–51.
Kissinger HE. Reaction kinetics in differential thermal analysis. Anal Chem. 1957;29(11):1702–6.
Kissinger HE. Variation of peak temperature with heating rate in differential thermal analysis. J Res Natl Bur Stand. 1956;57:217–21.
Sunose T, Akahira T. Method of determining activation deterioration constant of electrical insulating materials. Chiba Inst Technol (Sci Technol). 1971;16:22–31.
Vyazovkin S, Burnham AK, Criado JM, Pérez-Maqueda LA, Popescu C, Sbirrazzuoli N. ICTAC Kinetics Committee recommendations for performing kinetic computations on thermal analysis data. Thermochim Acta. 2011;520(1–2):1–19.
Chen Y, Tan L, Zhou W, Su J, Yang Y, Hu Y. Synthesis and structure of biodegradable hexylene terephthalate-co-lactide copolyesters. J Therm Anal Calorim. 2009;96(1):307–13.
Koo G-H, Jang J. Surface modification of poly(lactic acid) by UV/Ozone irradiation. Fibers Polym. 2009;9(6):674–8.
Wang X, Xia Y, Wei P, Chen Y, Wang Y, Wang Y. Nanocomposites of poly(propylene carbonate) reinforced with cellulose nanocrystals via sol-gel process. J Appl Polym Sci. 2014. doi:10.1002/app.40832.
Nagata M, Nakae M. Synthesis, characterization, and in vitro degradation of thermotropic polyesters and copolyesters based on terephthalic acid, 3-(4-hydroxyphenyl)propionic acid, and glycols. J Polym Sci Part A Polym Chem. 2001;39(18):3043–51.
Kricheldorf HR, Stukenbrock T. New polymer syntheses, 92. Biodegradable, thermotropic copolyesters derived from β-(4-hydroxyphenyl)propionic acid. Macromol. Chem Phys. 1997;198(11):3753–67.
Kijchavengkul T, Auras R, Rubino M, Alvarado E, Camacho Montero JR, Rosales JM. Atmospheric and soil degradation of aliphatic–aromatic polyester films. Polym Degrad Stab. 2010;95(2):99–107.
Bikiaris DN, Papageorgiou GZ, Achilias DS. Synthesis and comparative biodegradability studies of three poly(alkylene succinate)s. Polym Degrad Stab. 2006;91(1):31–43.
Tsuji H, Suzuyoshi K. Environmental degradation of biodegradable polyesters 1. Poly(ε-caprolactone), poly[(R)-3-hydroxybutyrate], and poly(l-lactide) films in controlled static seawater. Polym Degrad Stab. 2002;75(2):347–55.
Chen Y, Jia Z, Schaper A, Kristiansen M, Smith P, Wombacher R, et al. Hydrolytic and enzymatic degradation of liquid-crystalline aromatic/aliphatic copolyesters. Biomacromolecules. 2003;5(1):11–6.
Sato H, Kikuchi T, Koide N, Furuya K. Thermal degradation and combustion process of liquid crystalline polyesters studied by directly coupled thermal analysis-mass spectrometry. J Anal Appl Pyrolysis. 1996;37(2):173–83.
Acar I, Pozan GS, Özgümüş S. Thermal oxidative degradation kinetics and thermal properties of poly(ethylene terephthalate) modified with poly(lactic acid). J Appl Polym Sci. 2008;109(5):2747–55.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wei, P., Wang, Y., Xia, Y. et al. Hydrolytic and thermal oxidative degradation behavior of thermotropic aromatic–aliphatic copolyesters. J Therm Anal Calorim 128, 1067–1076 (2017). https://doi.org/10.1007/s10973-016-5975-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-016-5975-8