Abstract
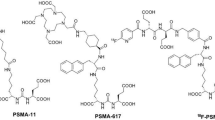
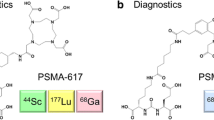
68Ga-radiolabeled small molecules targeting prostate-specific membrane antigen (PSMA) have been extensively investigated as 68Ga-positron emission tomography (68Ga-PET) tracers. Nevertheless, current 68Ga-labeled radiotracers suffer from either fair detection rates for metastatic prostate cancer lesions or a high uptake by the kidneys. This study synthesizes new PSMA-targeted radiotracers, [68Ga]SC691-DOTA, [68Ga]SC691-NOTA, and [68Ga]SC691-HBEDCC, and explores the in vitro and in vivo properties. These three radiotracers enabled PSMA-positive LNCaP tumors to be visualized by micro-PET/CT as early as 5 min post-injection. Biodistribution studies revealed that all three compounds could accumulate in the tumor with high affinity. The kidney’s uptake of [68Ga]SC691-DOTA and [68Ga]SC691-NOTA was comparable but lower than [68Ga]SC691-HBEDCC. [68Ga]SC691-HBEDCC demonstrated the most increased uptake and retention in the tumor over 2 h post-injection and a lower tumor-to-tissue ratio than [68Ga]SC691-DOTA, and [68Ga]SC691-NOTA.











Similar content being viewed by others
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71(3):209–249. https://doi.org/10.3322/caac.21660
Siegel RL, Miller KD, Fuchs HE, Jemal A (2022) Cancer statistics, 2022. CA Cancer J Clin 72(1):7–33. https://doi.org/10.3322/caac.21708
Siegel RL, Miller KD, Fuchs HE, Jemal A (2021) Cancer statistics, 2021. CA Cancer J Clin. 71(1):7–33.https://doi.org/10.3322/caac.21654. Erratum In: CA Cancer J Clin. 2021Jul; 71(4):359
Sweat SD, Pacelli A, Murphy GP, Bostwick DG (1998) Prostate-specific membrane antigen expression is greatest in prostate adenocarcinoma and lymph node metastases. Urology 52(4):637–640. https://doi.org/10.1016/s0090-4295(98)00278-7
Chang SS, Reuter VE, Heston WD, Bander NH, Grauer LS et al (1999) Five different anti-prostate-specific membrane antigen (PSMA) antibodies confirm PSMA expression in tumor-associated neovasculature. Cancer Res 59(13):3192–3198
Silver DA, Pellicer I, Fair WR, Heston WD, Cordon-Cardo C (1997) Prostate-specific membrane antigen expression in normal and malignant human tissues. Clin Cancer Res 3(1):81–85
Bostwick DG, Pacelli A, Blute M, Roche P, Murphy GP (1998) Prostate specific membrane antigen expression in prostatic intraepithelial neoplasia and adenocarcinoma: a study of 184 cases. Cancer 82(11):2256–2261. https://doi.org/10.1002/(sici)1097-0142(19980601)82:11%3c2256::aid-cncr22%3e3.0.co;2-s
Cunha AC, Weigle B, Kiessling A, Bachmann M, Rieber EP (2006) Tissue-specificity of prostate specific antigens: comparative analysis of transcript levels in prostate and non-prostatic tissues. Cancer Lett 236(2):229–238. https://doi.org/10.1016/j.canlet.2005.05.021
Donin NM, Reiter RE (2018) Why targeting PSMA Is a game changer in the management of prostate cancer. J Nucl Med 59(2):177–182. https://doi.org/10.2967/jnumed.117.191874
Baratto L, Song H, Duan H, Hatami N, Bagshaw HP, Buyyounouski M et al (2021) PSMA- and GRPR-Targeted PET: results from 50 patients with biochemically recurrent prostate cancer. J Nucl Med 62(11):1545–1549. https://doi.org/10.2967/jnumed.120.259630
Boellaard R, Delgado-Bolton R, Oyen WJ, Giammarile F, Tatsch K, Eschner W et al., European Association of Nuclear Medicine (EANM) (2015) FDG PET/CT: EANM procedure guidelines for tumour imaging: version 20. Eur J Nucl Med Mol Imaging 42(2):328–354. https://doi.org/10.1007/s00259-014-2961-x
Liu T, Liu C, Liu F, Xu X, Guo X, Xia L et al (2019) Synthesis and preclinical evaluation of 68Ga-PSMA-BCH for prostate cancer imaging. Bioorg Med Chem Lett 29(7):933–937. https://doi.org/10.1016/j.bmcl.2019.01.013
Liu T, Liu C, Xu X, Liu F, Guo X, Li N et al (2019) Preclinical evaluation and pilot clinical study of Al18F-PSMA-BCH for prostate cancer PET imaging. J Nucl Med 60(9):1284–1292. https://doi.org/10.2967/jnumed.118.221671
Xia L, Meng X, Wen L, Zhou N, Liu T, Xu X et al (2021) A highly specific multiple enhancement theranostic nanoprobe for PET/MRI/PAI image-guided radioisotope combined photothermal therapy in prostate cancer. Small 17(21):e2100378. https://doi.org/10.1002/smll.202100378
Ma X, Wang M, Wang H, Zhang T, Wu Z, Sutton MV et al (2019) Development of bispecific NT-PSMA heterodimer for prostate cancer imaging: a potential approach to address tumor heterogeneity. Bioconjug Chem 30(5):1314–1322. https://doi.org/10.1021/acs.bioconjchem.9b00252
Liu H, Moy P, Kim S, Xia Y, Rajasekaran A, Navarro V et al (1997) Monoclonal antibodies to the extracellular domain of prostate-specific membrane antigen also react with tumor vascular endothelium. Cancer Res 57(17):3629–3634
Jones W, Griffiths K, Barata PC, Paller CJ (2020) PSMA theranostics: review of the current status of PSMA-Targeted imaging and radioligand therapy. Cancers 12(6):1367. https://doi.org/10.3390/cancers12061367
Barakat A, Yacoub B, Homsi ME, Saad Aldine A, El Hajj A, Haidar MB (2020) Role of early PET/CT imaging with 68Ga-PSMA in staging and restaging of prostate cancer. Sci Rep 10(1):2705. https://doi.org/10.1038/s41598-020-59296-6
Lu M, Lindenberg L, Mena E, Turkbey B, Seidel J, Ton A et al (2022) A pilot study of dynamic 18F-DCFPyL PET/CT imaging of prostate adenocarcinoma in high-risk primary prostate cancer patients. Mol Imaging Biol 24(3):444–452. https://doi.org/10.1007/s11307-021-01670-5
Meijer D, Jansen BHE, Wondergem M, Bodar YJL, Srbljin S, Vellekoop AE et al (2020) Clinical verification of 18F-DCFPyL PET-detected lesions in patients with biochemically recurrent prostate cancer. PLoS ONE 15(10):e0239414. https://doi.org/10.1371/journal.pone.0239414
Afshar-Oromieh A, Zechmann CM, Malcher A, Eder M, Eisenhut M, Linhart HG et al (2014) Comparison of PET imaging with a (68)Ga-labelled PSMA ligand and (18)F-choline-based PET/CT for the diagnosis of recurrent prostate cancer. Eur J Nucl Med Mol Imaging 41(1):11–20. https://doi.org/10.1007/s00259-013-2525-5
Kuten J, Fahoum I, Savin Z, Shamni O, Gitstein G, Hershkovitz D et al (2020) Head-to-head comparison of 68Ga-PSMA-11 with 18F-PSMA-1007 PET/CT in staging prostate cancer using histopathology and immunohistochemical analysis as a reference standard. J Nucl Med 61(4):527–532. https://doi.org/10.2967/jnumed.119.234187
von Eyben FE, Bauman G, von Eyben R, Rahbar K, Soydal C, Haug AR et al (2020) Optimizing PSMA radioligand therapy for patients with metastatic castration-resistant prostate cancer: a systematic review and meta-analysis. Int J Mol Sci 21(23):9054. https://doi.org/10.3390/ijms21239054
Afshar-Oromieh A, Haberkorn U, Schlemmer HP, Fenchel M, Eder M, Eisenhut M et al (2014) Comparison of PET/CT and PET/MRI hybrid systems using a 68Galabelled PSMA ligand for the diagnosis of recurrent prostate cancer: initial experience. Eur J Nucl Med Mol Imaging 41(5):887–897. https://doi.org/10.1007/s00259-013-2660-z
Afshar-Oromieh A, Holland-Letz T, Giesel FL, Kratochwil C, Mier W, Haufe S et al (2017) Diagnostic performance of 68Ga-PSMA-11 (HBED-CC) PET/CT in patients with recurrent prostate cancer: evaluation in 1007 patients. Eur J Nucl Med Mol Imaging 44(8):1258–1268. https://doi.org/10.1007/s00259-017-3711-7
Carlucci G, Ippisch R, Slavik R, Mishoe A, Blecha J, Zhu S (2021) 68Ga-PSMA-11 NDA approval: a novel and successful academic partnership. J Nucl Med 62(2):149–155. https://doi.org/10.2967/jnumed.120.260455
Zamboglou C, Schiller F, Fechter T, Wieser G, Jilg CA, Chirindel A et al (2016) (68)Ga-HBED-CC-PSMA PET/CT versus histopathology in primary localized prostate cancer: a voxel-wise comparison. Theranostics 6(10):1619–1628. https://doi.org/10.7150/thno.15344
Fendler WP, Eiber M, Beheshti M, Bomanji J, Ceci F, Cho S et al (2017) 68Ga-PSMA PET/CT: joint EANM and SNMMI procedure guideline for prostate cancer imaging: version 1.0. Eur J Nucl Med Mol Imaging 44(6):1014–1024. https://doi.org/10.1007/s00259-017-3670-z
Szigeti F, Schweighofer-Zwink G, Meissnitzer M, Hauser-Kronberger C, Hitzl W, Kunit T et al (2022) Incremental impact of [68Ga]Ga-PSMA-11 PET/CT in primary N and M staging of prostate cancer prior to curative-intent surgery: a prospective clinical trial in comparison with mpMRI. Mol Imaging Biol 24(1):50–59. https://doi.org/10.1007/s11307-021-01650-9
Chen H, Cai P, Feng Y, Sun Z, Wang Y, Chen Y et al (2021) In vitro and in vivo comparative study of a novel 68Ga-labeled PSMA-targeted inhibitor and 68Ga-PSMA-11. Sci Rep 11(1):19122. https://doi.org/10.1038/s41598-021-98555-y
Kurtul N, Resim S, Koçarslan S (2018) Giant renal metastasis from prostate cancer mimicking renal cell carcinoma. Turk J Urol 44(4):367–369. https://doi.org/10.5152/tud.2017.39225
Ray Banerjee S, Chen Z, Pullambhatla M, Lisok A, Chen J et al (2016) Preclinical comparative study of (68)Ga-labeled DOTA, NOTA, and HBED-CC chelated radiotracers for targeting PSMA. Bioconjug Chem 27(6):1447–1455. https://doi.org/10.1021/acs.bioconjchem.5b00679
Benešová M, Schäfer M, Bauder-Wüst U, Afshar-Oromieh A, Kratochwil C, Mier W et al (2015) Preclinical evaluation of a tailor-made DOTA-conjugated PSMA inhibitor with optimized linker moiety for imaging and endoradiotherapy of prostate cancer. J Nucl Med 56(6):914–920. https://doi.org/10.2967/jnumed.114.147413
Wüstemann T, Bauder-Wüst U, Schäfer M, Eder M, Benesova M, Leotta K et al (2016) Design of Internalizing PSMA-specific Glu-ureido-based Radiotherapeuticals. Theranostics 6(8):1085–1095. https://doi.org/10.7150/thno.13448
Acknowledgements
The authors would like to thank the National Natural Science Foundation of China for funding this work (U20A20384); the Doctoral Research Initiation Fund of Affiliated Hospital of Southwest Medical University; Nuclear Medicine and Molecular Imaging Key Laboratory of Sichuan Province (HYX19001), the Sichuan Science and Technology Foundation (2021YJ0131 and 2020ZYD101), and Luzhou-Southwest Medical University Cooperative Application Foundation (2020LZXNYDJ50)
Author information
Authors and Affiliations
Contributions
Conceptualization: ZZ. Methodology: ZZ, NL, YL. Validation: YL, LX. Formal analysis: ZZ, YL. Investigation: YL, LX, PC, YW, YF, WZ. Resources: YC, WZ. Data curation: YL, LX. Writing: ZZ. Funding acquisition: YC, ZZ, WZ. Review: ZZ, YL, LX, PC, YW, YF, WZ, YC, NL.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
Institutional Review Board Statement: The Ethics Committee for Southwest Medical University (2021-02-29) approved the study. All methods were carried out following relevant guidelines and regulations, and all methods are reported per ARRIVE guidelines.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, Y., Xia, L., Cai, P. et al. In vitro and in vivo comparative study of 68Ga-labeled DOTA-, NOTA-, and HBEDCC-chelated radiotracers targeting prostate-specific membrane antigen. J Radioanal Nucl Chem 332, 617–628 (2023). https://doi.org/10.1007/s10967-022-08731-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-022-08731-1