Abstract
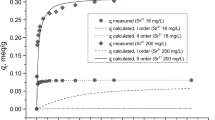
Kinetics of Cs+ uptake by natural and synthetic mordenites was studied. The experimental data is best fitted to the pseudo-second-order rate model and the parameters k2 and qe are calculated. The application of intra-particle diffusion and liquid film diffusion models show multilinear graphical dependence, indicating that both mechanisms influence the run of uptake. Rietveld XRD structural analyses were used for identification of the positions preferably occupied by Cs+ cations during the ion-exchange as a function of contact time. The obtained results show that the predominance of Na+ ions in the initial samples makes the exchanged sites more accessible by Cs+ ions.











Similar content being viewed by others
References
Misaelides P (2011) Application of natural zeolites in environmental remediation: a short review. Microporous Mesoporous Mater 144:15–18
Yuna Z (2016) Review of the natural, modified, and synthetic zeolites for heavy metals removal from wastewater. Environ Eng Sci 33:443–454
Rajec P, Macášek F, Feder M, Misaelides P, Šamajová E (1998) Sorption of caesium and strontium on clinoptilolite- and mordenite-containing sedimentary rocks. J Radioanal Nucl Chem 229:49–55
Cortés-Martínez R, Olguín MT, Solache-Ríos M (2010) Cesium sorption by clinoptilolite-rich tuffs in batch and fixed-bed systems. Desalination 258:164–170
Borai EH, Harjula R, Paajanen A (2009) Efficient removal of cesium from low-level radioactive liquid waste using natural and impregnated zeolite minerals. J Hazard Mater 172:416–422
Munthali MW, Johan E, Aono H, Matsue N (2015) Cs+ and Sr2+ adsorption selectivity of zeolites in relation to radioactive decontamination. J Asian Ceram Soc 3:245–250
de Gennaro B, Colella A, Aprea P, Colella C (2003) Evaluation of an intermediate-silica sedimentary chabazite as exchanger for potentially radioactive cations. Microporous Mesoporous Mater 61:159–165
Baek W, Ha S, Hong S, Kim S, Kim Y (2018) Cation exchange of cesium and cation selectivity of natural zeolites: chabazite, stilbite, and heulandite. Microporous Mesoporous Mater 264:159–166
Johan E, Yamada T, Munthali MW, Kabwadza-Corner P, Aono H, Matsue N (2015) Natural zeolites as potential materials for decontamination of radioactive cesium. Procedia Environ Sci 28:52–56
Meier WM, Olson DH, Baerlocher C (1996) Atlas of zeolite structure types. Zeolites 17:1–230
Meier WM (1961) The crystal structure of mordenite (ptilolite). Z Kristallogr 115:439–450
Alberti A, Davoli P, Vezzalini G (1986) The crystal structure refinement of a natural mordenite. Z Kristallogr – Cryst Mater 175:249–256
Armbruster T, Gunter ME (2001) Crystal structures of natural zeolites. In: Bish DL, Ming DW (eds) Reviews in mineralogy and geochemistry, 45, natural zeolites: occurrence, properties, use. Mineralogical Society of America, Washington DC, pp 1–67
Simoncic P, Armbruster T (2004) Peculiarity and defect structure of the natural and synthetic zeolite mordenite: a single-crystal X-ray study. Am Mineral 89:421–431
Meier WM (1978) Constituent sheets in the zeolite frameworks of the mordenite group. In: Sand LB, Mumpton FA (eds) Natural zeolites, occurrence, properties, use. Pergamon Press, Oxford, pp 99–103
Ohnuki T, Kozai N (2013) Adsorption behavior of radioactive cesium by non-mica minerals. J Nucl Sci Technol 50:369–375
Chmielewska E, Lesný J (2012) Selective ion exchange onto Slovakian natural zeolites in aqueous solutions. J Radioanal Nucl Chem 293:535–543
Rahman RA, Ibrahium HA, Hung YT (2011) Liquid radioactive wastes treatment: a review. Water 3:551–565
El-Kamash AM (2008) Evaluation of zeolite a for the sorptive removal of Cs+ and Sr2+ ions from aqueous solutions using batch and fixed bed column operations. J Hazard Mater 151:432–445
Aleksiev B, Djurova E (1976) Mordenite zeolites from the North-Eastern Rhodopes. Compt Rend Acad Bulg Sci 29:865–867
Lihareva N, Tzvetanova Y, Petrov O, Dimova L (2013) Sorption of silver cations by natural and Na-exchanged mordenite. Sep Sci Technol 48:617–625
Lagergren S (1898) Zur theory der sogenannten adsorption gelöster stoffe. Kungliga Svenska Vetenkapsakademiens Handingar 24:1–39
Ho YS, McKay G (1999) Pseudo-second order model for sorption processes. Process Biochem 34:451–465
Gupta GS, Prasad G, Panday KK, Singh VN (1988) Removal of chrome dye from aqueous solutions by fly ash. Water Air Soil Poll 37:13–24
Weber WJ Jr, Morris JC (1963) Kinetic of adsorption on carbon from solution. J Sanit Eng Div 89:31–59
Boyd GE, Adamson AW, Myers LS (1947) The exchange adsorption of ions from aqueous solutions by organic zeolites. II. Kinetics. J Am Chem Soc 69:2836–2848
Reichenberg D (1953) Properties of ion-exchange resins in relation to their structure. III. Kinetics of exchange. J Am Chem Soc 75:589–597
Rietveld HM (1969) A profile refinement method for nuclear and magnetic structures. J Appl Crystallogr 2:65–71
Topas (2009) V 4.2: general profile and structure analysis software for powder diffraction. Bruker AXS, Karlsruhe, Germany
Schlenker JL, Pluth JJ, Smith JV (1978) Positions of cations and molecules in zeolites with the mordenite-type framework: V dehydrated Rb-mordenite. Mater Res Bull 13:77–82
Passaglia E, Artioli G, Gualtieri A, Carnevali R (1995) Diagenetic mordenite from Ponza, Italy. Eur J Mineral 7:429–438
Kragović M, Sekulić Ž, Stojanović M, Petrović M, Dondur V, Damjanović L, Jović A (2014) Kinetics of Pb(II) ions removal from aqueous solution using the natural and Fe(III)-modified zeolite. In: Daković A, Trgo M, Langella A (eds) Proceedings of the 9-th international conference of the occurrence, properties and utilization of natural zeolites. Zeolite 2014, Belgrade, pp 109–110
Wang S, Li H, Xu L (2006) Application of zeolite MCM-22 for basic dye removal from wastewater. J Colloid Interface Sci 295:71–78
Albadarin AB, Mangwandi C, Ala’a H, Walker GM, Allen SJ, Ahmad MN (2012) Kinetic and thermodynamics of chromium ions adsorption onto low-cost dolomite adsorbent. Chem Eng J 179:193–202
Toor M, Jin B (2012) Adsorption characteristics, isotherm, kinetics, and diffusion of modified natural bentonite for removing diazo dye. Chem Eng J 187:79–88
Chen H, Wang A (2007) Kinetic and isothermal studies of lead ion adsorption onto palygorskite clay. J Colloid Interface Sci 307:309–316
Quattrini F, Galceran J, David CA, Puy J, Alberti G, Rey-Castro C (2017) Dynamics of trace metal sorption by an ion-exchange chelating resin described by a mixed intraparticle/film diffusion transport model. The Cd/Chelex case. Chem Eng J 317:810–820
Walker GM, Weatherley LR (1999) Kinetics of acid dye adsorption on GAC. Water Res 33:1895–1899
Jovanovic M, Grbavcic Z, Rajic N, Obradovic B (2014) Removal of Cu (II) from aqueous solutions by using fluidized zeolite A beads: Hydrodynamic and sorption studies. Chem Eng Sci 117:85–92
Song SG (1999) Crystal defects of mordenite structures. J Mater Res 14:2616–2620
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dimowa, L., Lihareva, N., Tzvetanova, Y. et al. Kinetics and Rietveld XRD structural study of Cs+ sorption on natural and synthetic mordenites in the first 12 h. J Radioanal Nucl Chem 331, 5741–5752 (2022). https://doi.org/10.1007/s10967-022-08648-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-022-08648-9