Abstract
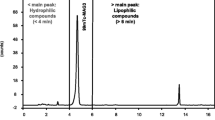
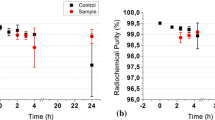
99mTc-ethylene dicysteine diethyl ester (99mTc-ECD) is an established radiopharmaceutical used for brain perfusion imaging; however it does not have any recognized biological quality control to ascertain its in vivo efficacy. The present paper describes an in vitro enzymatic method that can be adapted as a regular biological quality control for ECD kit evaluation before its release for patient end use. The method involves reaction of 99mTc-ECD (< 74 MBq; 0.1 mg ECD) with pig liver esterase enzyme (≥ 20 mg/mL) yielding metabolites in quantitative yield post 1 h incubation. Methodology developed is precise for routine quality control analyses of ECD kits.






Similar content being viewed by others
References
Brodie KK, Hermann K, Schanker LS (1960) Importance of dissociation constant and lipid solubility in influencing the passage of drugs into the cerebrospinal fluid. J Pharmacol Exp Ther 130:20–25
Dischino DD, Welch MJ, Kilbourn MR, Raichle ME (1983) Relationship between lipophilicity and brain extraction of C-11-labelled radiopharmaceuticals. J Nucl Med 24:1030–1038
Levin VA (1980) Relationship of octanol/water partition coefficient and molecular weight to rat brain capillary permeability. J Med Chem 23:683–684
Oldendorf WH (1974) Lipid solubility and drug penetration of the blood brain barrier. Proc Soc Exp Biol Med 147:813–816
Burns HD, Dannals RF, Dannals TE, Kramer AV, Marzilli LG (1981) Synthesis of tetradentate aminothiol ligands and their technetium complexes. J Label Compd Radiopharm 18:54–55
Cheesman EH, Blanchette MA, Calabrese JC, Ganey MV, Maheu LJ, Morgan RA, Walovitch RC, Watson AD, Williams SJ, Miller SJ (1989) Technetium-99m complexes of ester derivatized diamine-dithiol ligands for imaging brain perfusion. J Label Compd Radiopharm 26:421–423
Davison A, Jones AG, Orvig C, Sohn M (1981) A new class of oxotechnetium (5+) chelate complexes containing a TcON2S2 core. Inorg Chem 20:1629–1632
Kung HF, Molnar M, Billings J, Wicks R, Blau M (1984) Synthesis and biodistribution of neutral lipid-soluble Tc-99m complexes that cross the blood-brain barrier. J Nucl Med 25:326–332
Walovitch RC, Hill TC, Garrity ST, Cheesman EH, Burgess BA, O’Leary BH, Watson AD, Ganey MV, Morgan RA, Williams SJ (1989) Characterization of technetium-99m-L, L-ECD for brain perfusion imaging: part 1. Pharmacology of technetium-99m ECD in nonhuman primates. J Nucl Med 30:1892–1901
Leveille J, Demonceau G, De Roo M, Rigo P, Taillefer R, Morgan RA, Kupranick D, Walovitch RC (1989) Characterization of technetium-99m-L, L-ECD for brain perfusion imaging: part 2. Biodistribution and brain imaging in humans. J Nucl Med 30:1902–1910
Vallabhajosula S, Zimmerman RE, Picard M, Stritzke P, Mena I, Hellman RS, Tikofsky RS, Stabin MG, Morgan RA, Goldsmith SJ (1989) 99mTc-ECD: a new brain imaging agent: in vivo kinetics and biodistribution in normal human subjects. J Nucl Med 30:599–604
Walovitch RC, Franceschi M, Piccard M, Cheesman EH, Hall KM, Makuch J, Watson MW, Zimmerman RE, Watson AD, Ganey MV, Williams SJ, Holman BL (1991) Metabolism of 99mTc-L, L-ethyl cysteinate dimer in healthy volunteers. Neuropharmacology 30:283–292
Vanbilloen HP, Cleynhens BJ, Verbruggen AM (1998) Importance of the two ester functions for the brain retention of 99mTc-labelled ethylene dicysteine diethyl ester (99mTc-ECD). Nucl Med Biol 25:569–575
Inoue Y, Momose T, Ohtake T, Nishikawa J, Sasaki Y, Waritani T, Inoue M (1997) Metabolism of technetium-99m-L, L-ethyl cysteinate dimer in rat and cynomolgus monkey tissue. J Nucl Med 38:1733–1737
http://www.britatom.gov.in/docs/rph_ria_cat/rph_cold/rph_tck42.pdf. Accessed 15 June 2018
Tsopelas C, Smyth D (2005) Characterization and quality control analysis of 99mTc-bicisate. J Nucl Med Technol 33:89–93
Mikiciuk-Olasik E, Bilichowski I (2000) Determination of L, L-ethylene dicysteine di-ethylester stability by RP HPLC. Chem Anal (Warsaw) 45:809–813
Verduyckt T, Kieffer D, Huyghe D, Cleynhens B, Verbeke K, Verbruggen A, Bormans G (2003) Identity confirmation of Tc-MAG3, Tc-Sestamibi and Tc-ECD using radio-LCMS. J Pharm Biomed Anal 32:669–678
Chao MY, Liu KT, Hsia YC, Liao MH, Shen LH (2011) Direct determination of ECD in ECD kit: a solid sample quantitation method for active pharmaceutical ingredient in drug product. DOI, J Biomed Biotechnol. https://doi.org/10.1155/2011/196238
Wahl O, Cleynhens J, Verbruggen AM, Holzgrabe U (2018) Impurity profiling of N,N′-ethylenebis-l-cysteine diethyl ester (Bicisate). J Pharm Biomed Anal 150:132–136
Mang’era KO, Bellande E, Pasqualini R, Verbruggen A (1996) Comparison of reversed phase and reversed phase ion pair high performance liquid chromatography for analysis of TcO and TcN complexes of L, L-ethylene dicysteine di-ethylester and its acid analogues. J Pharm Biomed Anal 14:1241–1244
Wahl O, Holzgrabe U (2014) Impurity profiling of carbocisteine by HPLC-CAD, qNMR and UV/vis spectroscopy. J Pharm Biomed Anal 95:1–10
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Das, S., Mathur, A., Nair, P. et al. In vitro enzymatic technique for biological quality control of 99mTc-ECD. J Radioanal Nucl Chem 318, 1287–1295 (2018). https://doi.org/10.1007/s10967-018-6055-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-018-6055-2