Abstract
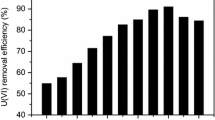
Ceratophyllum demersum was used to remove U(VI) from aqueous solution. Effects of various adsorption parameters such as pH, shaking time, initial U(VI) concentration, and temperature were investigated. The optimum pH for U(VI) removal was 5.0. Pseudo-first-order kinetic model and Langmuir isotherm model could best describe the adsorption process. The maximum monolayer adsorption capacity was 140.45 mg/g. Thermodynamic calculations suggested that the U(VI) adsorption process was spontaneous and exothermic. The amino, hydroxyl, and carbonyl groups on the algal surface could contribute to U(VI) adsorption. The present study suggested that C. demersum biomass could be an efficient biosorbent to remove U(VI) contaminants.







Similar content being viewed by others
References
Zhang S, Shu XW, Zhou Y, Huang L, Hua DB (2014) Highly efficient removal of uranium(VI) from aqueous solutions using poly(acrylic acid)-functionalized microspheres. Chem Eng J 253:55–62
Mellah A, Chegrouche S, Barkat M (2007) The precipitation of ammonium uranyl carbonate (AUC): thermodynamic and kinetic investigations. Hydrometallurgy 85(2–4):163–171
Prasadaao TR, Metilda P, Gladis JM (2006) Preconcentration techniques for uranium(VI) and thorium(IV) prior to analytical determination-an overview. Talanta 68(4):1047–1064
Newsome L, Morris K, Trivedi D, Atherton N, Lloyd JR (2014) Microbial reduction of uranium(VI) in sediments of different lithologies collected from Sellafield. Appl Geochem 51:55–64
Ambashta RD, Sillanpaa MET (2011) Membrane purification in radioactive waste management: a short review. J Environ Radioact 105:76–84
Kumar JR, Kim JS, Lee JY, Yoon HS (2011) A brief review on solvent extraction of uranium from acidic solutions. Sep Purif Rev 40(2):77–125
Stucker V, Ranville J, Newman M, Peacock A, Cho J, Hatfield K (2011) Evaluation and application of anion exchange resins to measure groundwater uranium flux at a former uranium mill site. Water Res 45(16):4846–4876
Bhat SV, Melo JS, Chaugule BB, D’Souza SF (2011) Biosorption of uranium(VI) from aqueous solutions by Ca-pretreated Cystoseira indica alga: Breakthrough curves studies and modeling. J Hazard Mater 189(1–2):141–149
Khani MH (2011) Statistical analysis and isotherm study of uranium biosorption by Padina sp. algae biomass. Environ Sci Pollut Res 18(5):790–799
Lee KY, Kim KW, Baek YJ, Chung DY, Lee EH, Lee SY, Moon JK (2014) Biosorption of uranium(VI) from aqueous solution by biomass of brown algae Laminaria japonica. Water Sci Technol 70(1):136–143
Bampaiti A, Yusan S, Aytas S, Pavlidou E, Noli F (2016) Investigation of uranium biosorption from aqueous solutions by Dictyopteris polypodioides brown algae. J Radioanal Nucl Chem 307(2):1335–1343
Markich SJ (2013) Water hardness reduces the accumulation and toxicity of uranium in a freshwater macrophyte (Ceratophyllum demersum). Sci Total Environ 443:582–589
Zhang ZB, Yu XF, Cao XH, Hua R, Li M, Liu YH (2014) Adsorption of U(VI) from aqueous solution by sulfonated ordered mesoporous carbon. J Radioanal Nucl Chem 301(3):821–830
Xie SB, Zhang C, Zhou XH, Yang J, Zhang XJ, Wang JS (2009) Removal of uranium(VI) from aqueous solution by adsorption of hematite. J Environ Radioact 100(2):162–166
Song J, Kong H, Jang J (2011) Adsorption of heavy metal ions from aqueous solution by polyrhodanine-encapsulated magnetic nanoparticles. J Colloid Interface Sci 359(2):505–511
Tan LC, Liu Q, Jing XY, Liu JY, Song DL, Hu SX, Liu LH, Wang J (2015) Removal of uranium(VI) ions from aqueous solution by magnetic cobalt ferrite/multiwalled carbon nanotubes composites. Chem Eng J 273:307–315
Solgy M, Taghizadeh M, Ghoddocynejad D (2015) Adsorption of uranium(VI) from sulphate solutions using Amberlite IRA-402 resin: Equilibrium, kinetics and thermodynamics study. Ann Nucl Energy 75:132–138
Zhang XF, Jiao CS, Wang J, Liu Q, Li RM, Yang PP, Zhang ML (2012) Removal of uranium(VI) from aqueous solutions by magnetic Schiff base: kinetic and thermodynamic investigation. Chem Eng J 198–199:412–419
Yang SK, Tan N, Yan XM, Chen F, Lin YC (2013) Adsorption of thorium(IV) from aqueous solution by non-living biomass of mangrove endophytic fungus Fusarium sp. #ZZF51. J Radioanal Nucl Chem 298(2):827–833
Ai L, Luo XG, Lin XY, Zhang SZ (2013) Biosorption behaviors of uranium(VI) from aqueous solution by sunflower straw and insights of binding mechanism. J Radioanal Nucl Chem 298(3):1823–1834
Bai J, Fan FL, Wu XL, Tian W, Zhao L, Yin XJ, Fan FY, Li Z, Tian LL, Wang Y, Qin Z, Guo JS (2013) Equilibrium, kinetic and thermodynamic studies of uranium biosorption by calcium alginate beads. J Environ Radioact 126:226–231
Ortaboy S, Atun G (2014) Kinetics and equilibrium modeling of uranium(VI) sorption by bituminous shale from aqueous solution. Ann Nucl Energy 73:345–354
Keshtkar AR, Mohammadi M, Moosavian MA (2015) Equilibrium biosorption studies of wastewater U(VI), Cu(II) and Ni(II) by the brown alga Cystoseira indica in single, binary and ternary metal systems. J Radioanal Nucl Chem 303(1):363–376
Ding DX, Xin X, Li L, Hu N, Li GY, Wang YD, Fu PK (2014) Removal and recovery of U(VI) from low concentration radioactive wastewater by ethylenediamine modified biomass of Aspergillus niger. Water Air Soil Pollut 225:2206
Li XY, Li FB, Jin Y, Jiang CJ (2015) The uptake of uranium by tea wastes investigated by batch, spectroscopic and modeling techniques. J Mol Liq 209:413–418
Tan LC, Wang J, Liu Q, Sun YB, Zhang HS, Wang YL, Jing XY, Liu JY, Song DL (2015) Facile preparation of oxine functionalized magnetic Fe3O4 particles for enhanced uranium(VI) adsorption. Colloids Surf, A 466:85–91
Singhal RK, Basu H, Pimple MV, Manisha V, Basan MKT, Reddy AVR (2013) Spectroscopic determination of U(VI) species sorbed by the Chlorella (Chlorella pyrenoidosa) fresh water algae. J Radioanal Nucl Chem 298(1):587–592
Gunther A, Raff J, Geipel G, Bernhard G (2008) Spectroscopic investigations of U(VI) species sorbed by the green algae Chlorella vulgaris. Biometals 21(3):333–341
Nancharaiah YV, Joshi HM, Mohan TVK, Venugopalan VP, Narasimhan SV (2006) Aerobic granular biomass: a novel biomaterial for efficient uranium removal. Curr Sci 91(4):503–509
Acknowledgements
This work is supported in part by Grants from Key Project from National Natural Science Foundation of China (41430106), National Natural Science Foundation of China (41573080, U1402234, 41711530030, 41711530150), Public welfare project of Chinese Ministry of Environmental Protection (201509049), and International key project of Ministry of Science and Technology of China(S2016G2135).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yi, Zj., Yao, J., Zhu, Mj. et al. Biosorption characteristics of Ceratophyllum demersum biomass for removal of uranium(VI) from an aqueous solution. J Radioanal Nucl Chem 313, 19–27 (2017). https://doi.org/10.1007/s10967-017-5269-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-017-5269-z