Abstract
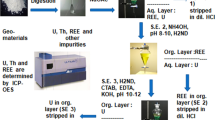
A novel method for simple, rapid and effective separation of thorium for it’s determination in geological samples such as rocks, and minerals has been developed. Thorium forms 1:2 neutral complex with the bi-dentate ligand, 2,3-dihydroxynaphthalene (2,3-H2ND), which is extracted into ethylacetate at pH 5–6. Thorium from the organic phase is quantitatively stripped off using dilute hydrochloric acid, and then determined spectrophotometrically as well as by ICP-AES. The effects of variables affecting the quantitative extraction of thorium have been studied in detail. The method has been validated by analyzing certified reference materials (CRM). RSD of the method varied over the range 1 to 5 %. The method has been successfully applied to rocks and minerals.




Similar content being viewed by others
Reference
He Q, Chang X, Wu Q, Huang X, Hu Z, Zhai Y (2007) Synthesis and applications of surface grafted Th(IV)-imprinted polymers for selective solid phase extraction of Th(IV). Anal Chim Acta 605:192–197
Gopalkrishnan M, Radhakrishnan K, Dhami PS, Kulkarni VT, Joshi MV, Patwardhan AB, Ramanujam A, Mathur JN (1997) Determination of trace impurities in uranium, thorium and plutonium matrices by solvent extraction and ICP-AES. Talanta 44:169–176
Joannon S, Telouk P, Pin C (1997) Determination of U and Th at ultra-trace levels by isotope dilution ICP-MS using a geyser-type ultrasonic nebulizer: application to geological samples. Spectrochim Acta Part B 52:1783–1789
Elyahyaoui A, Zarki R, Chiadli A (2003) A method for the rapid radiochemical analysis of uranium and thorium isotopes in impure carbonates. Appl Radiat Isot 58:119–124
Liu BF, Liu LB, Cheng CK (1998) Separation and determination of thorium, uranium and mixed rare earth elements as their UV/Vis absorbing complexes by capillary zone electrophoresis. Talanta 47(2):291–299
Anasuya DVS, Krishna RV (2013) 2-hydroxy-1-naphthaldehyde-p-hydroxybenzoichydrazone: a new chromogenic reagent for the determination of thorium(IV) and uranium(VI). J Chem, Hindawi Publishing Corporation, volume 2013, Article ID 697379.
Agarawal YK, Dayal U (1985) Spectrophotometric determination of trace level thorium with N-phenylbenzohydroxamic acid. J Radioanal Nucl Chem 90(2):303–308
Kolthaoff IM (1963) Ed. Treatise on Analytical Chemistry, Part II, Analytical Chemistry of elements, vol. 8, Interscience Publishers, John Wiley and Sons, New York
Premadas A (2012) Determination of trace level lanthanides and thorium by ICP-AES in different types of geological, red mud and coal fly ash samples after separation as oxalates using calcium as carrier. Atomic Spectrosc 33(1):14–23
Korkish J (1989) Handbook of Ion exchange resins; their application to Inorganic Analytical Chemistry, vol II. CRC Press Inc., Boca Raton
Raju Ch, Kesava Siva, Subramanian MS (2007) Sequential separation of lanthanides, thorium and uranium using novel solid phase extraction method from high acidic nuclear wastes. J Hazard Mater 145:315–322
Caramella VC, Maggi L, Ganzerli MTV (1999) Separation of traces of thorium from large amounts uranium by adsorption on lanthanum ammonium oxalate. Appl Radiat Isot 51:353–357
Pourreza N, Parham H, Mirzavand P (2009) Flotation-spectrophotometric determination of trace amounts of thorium. J Chinese Chem Soc 56(4):785–788
Tamhina B, Gojmerac A, Herak MJ (1978) Extraction and complex formation of thorium (IV) with 3-hydroxy-2-methyl-1-(4-tolyl)-pyridone. J Inorg Nuclear Chem 40(2):335–338
Roopa Bose, Murthy DSR, Chakrapani G (2005) Extraction of thorium(IV) as perchlorate and chloroacetate complexes with 1-phenyl-2,3-dimethyl-5-pyrazolone (antipyrine). J Radioanal Nucl Chem 265:115–122
Patkar NS, Burungale AS, Patil RJ (2009) Separation and liquid liquid extraction of thorium(IV) as sulphate complex with synergistic mixture of N-n-octylaniline and trioctylamine as an extractant. RASAYAN J Chem 2(4):825–832
Tarafder PK, Murugan P, Kunkal L, Rathore DPS (2002) Extraction of uranium with 2,3 dihydroxynaphthalene and cetyltrimethylammoniumbromide, and its fluorimetric determination in silicate rocks. J Radioanal Nucl Chem 253:135–142
Tarafder PK, Sardana AK (2000) A rapid method for the spectrophotometric determination of iron in silicate rock. Chem Anal (Warshaw) 45:145–166
Tarafder PK, Durani S, Saran R, Ramnaiah GV (1994) Rapid spectrophotometric method (aqueous and extractive) for the determination of titanium in silicate rock. Talanta 41:1345–1351
Tarafder PK, Mondal RK, Kunkal L, Rathore DPS (2004) Liquid-liquid extraction and spectrophotometric determination of Mn(II) in geological samples. Chem Anal (Warshaw) 49:251–260
Tarafder PK, Pradhan SK, Mondal RK, Sircar JK (2013) Liquid-liquid extraction of rare earth elements (total) using 2,3 dihydroxynaphthalene as an extractant and its application to the determination of REEs(total) in rocks, soil and sediments samples by spectrophotometry. J Indian Chem Soc 90(11):1961–1967
Thakur R, Pradhan SK, Jha RR, Tarafder PK (2013) Liquid-liquid extraction of molybdenum and its ultra-trace determination by GF-AAS. Explor Res Atomic Minerals 23:117–123
Onishi H (1989) Photometric determination of trace metals, vol Part IIB, 4th edn. A Wiley-Interscience Publication, John Wiley and Sons, New York, pp 449–485
Acknowledgments
The authors express their sincere thanks and gratitude to Shri P.S.Parihar, Director, AMD for his kind permission to publish the paper. The authors are also thankful to Shri Promod Kumar, Regional Director and Shri G.S.Yadav, Dy. Regional Director, AMD, Eastern Region, Jamshedpur for providing necessary facilities to carry out the work. Also, the authors are thankful to Dr. G. Chakrapani, Head, Chemistry Group, for his constant encouragement to do the work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tarafder, P.K., Pradhan, S.K. & Mondal, R.K. Separation of thorium by facile liquid–liquid extraction, and its rapid spectrophotometric and ICP-AES determination in rocks and minerals. J Radioanal Nucl Chem 309, 1021–1028 (2016). https://doi.org/10.1007/s10967-016-4738-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-016-4738-0