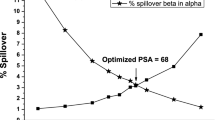
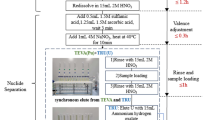
Abstract
A new method has been developed to measure Iron-55 (55Fe) in urine samples, which could be used for dose assessment of 55Fe in an accidental exposure. 55Fe was effectively precipitated from 1 litre of urine by bringing the sample to 2M NaOH. Potential interferences for 55Fe (e.g., Cr, Co, Ni, Mn, Zn and Mo) were removed by anion exchange chromatography. The Fe recovery was measured by UV–Vis absorption spectroscopy and 55Fe was determined by liquid scintillation counting. The method was validated using spiked samples.







Similar content being viewed by others
References
Holden NE (2013) CRC handbook of chemistry and physics. http://www.hbcpnetbase.com/. Accessed June 2014
Evans JC, Lepel EL, Sanders RW, Wilkerson CL, Silker W, Thomas CW, Abel KH, Robertson DR (1984) Long-lived activation products in reactor materials. Pacific Northwest Laboratory, Richland
Brechignac F, Deville-Cavelin G (2002) Fiche radionucléide 59Fe et environnement. IRSN, Fontenay-aux-Roses
Warwick PE, Croudace IW (2006) Anal Chim Acta 567:277–285
Hou X, Østergaard LF, Nielsen SP (2004) Anal Chim Acta 535:297–307
König W, Schupfner R, Schüttelkopf H (1995) J Radioanal Nucl Chem 193:119–125
Nielsen JM (1960) The radiochemistry of iron. N.R.C, Washington
Collier CG, Stradling GN, Foster PP, Hodgson A (1994) Radiat Prot Dosim 53:173–177
Bukhave K, Sorensen AD, Hansen MA (2001) J Trace Elem Med Biol 15:56–58
ICRP Publication 69 (1995) Age-dependent doses to members of the public from intake of radionuclides: part 3 ingestion dose coefficients. Pergamon, Oxford
ICRP Publication 23 (1975) Report on the task group on reference man. International commission on radiological protection. Pergamon, Oxford
Jäggi M, Rüthi M, Eikenberg J (2008) Method for 55Fe and 63Ni determination by LSC in radioactive waste. In: Proceedings of the LSC conference, Davos
Grahek Ž (2006) Rožmari Mačefat M. J Radioanal Nucl Chem 267:131–137
Dai X, Kramer-Tremblay S (2014) Anal Chem 86(11):5441–5447
Taddei MHT, Macacini JF, Vicente R, Marumo JT, Sakata SK, Terremoto LAA (2013) J Radioanal Nucl Chem 295:2267–2272
Mikelic L, Orescanin V, Lulic S (2007) Nucl Instrum Methods Phys Res Sect B 263:95–98
Geckeis H, Hentschel D, Jensen D, Görtzen A, Kerner N (1997) J Anal Chem 357:864–869
Iron-55 in Water (2001) Analytical Procedures. Eichrom Tecnologies, LLC, Lisle
Gascó C, Navarrob N, Alvarezb A, Herasa C, Suañeza A (2008) The difficulties of measuring 55Fe and 63Ni in environmental samples. Department of Radiation Protection, CIEMAT, Madrid
Vajda N, Molnár S, Osváth S (2005) Extraction chromatography and liquid scintillation counting for the analysis of long-lived radionuclides. In: Proceedings of the LSC conference, Davos
Priya S, Gopalakrishnan RK, Goswami A (2014) J Radioanal Nucl Chem. doi:10.1007/s10967-014-3190-2
Razdolescua AC, Cassette PH, Sahagia M (2008) Appl Radiat Isot 66:750–755
El-kot AM (1990) Isot Environ Health Stud 26(10):499–502
Cregan SP, Leon JW, Linauskas SH (1993) Bioassays techniques for 55Fe in urine samples, AECL internal report
Putnam DF (1971) Composition and concentrative properties of human urine. N.A.S.A, Washington
Currie LA (1968) Anal Chem 40:586–593
Guerin N, Dai X (2014) Anal Chem 86:6026–6031
Kolthoff IM, Sandell EB (1945) Textbook of quantitative inorganic analysis. The Macmillan Company, New York
Silva AMN, Kong XL, Parkin MC, Cammack R, Hider RC (2009) Dalton Trans 40:8616–8625
Rue EL, Bruland KW (1995) Mar Chem 50:117–138
Schweitzer GK, Pesterfield LL (2010) The aqueous chemistry of the elements. Oxford University Press, New York
Sato K, Hyodo M, Takagi J, Aoki M, Noyori R (2000) Tetrahedron Lett 9:1439–1442
Jain AK, Kumar M, Kumar D (2014) Pharm Chem 6:187–193
Pol UN, Yamgar RS, Dodwad SS (1997) Asian J Chem 9:58–62
Mathew A, Panda JK (2012) Int Res J Pure Appl Chem 2:196–210
Theis Thomas L, Singer Philip C (1974) Environ Sci Technol 8:569–573
Greenwood NN, Earnshaw A (2003) Chemistry of the elements, 2nd edn. Elsevier Butterworth-Heinemann, Oxford
Kraus KA, Nelson F (1995) Distribution coefficients of the elements on strong anion exchange resins in HCl. In: Proceedings of 1st international conference on peaceful uses atomic energy,7,118
Klueh RL (2004) Elevated-temperature ferritic and martensitic steels and their application to future nuclear reactors. Oak Ridge National Laboratory, Oak Ridge
Lee BS, Kim MC, Yoon JH, Hong JH (2010) Int J Press Vessel Pip 87:74–80
Kim KY, Kim HY, Rho GH, Sohn DS (2011) J Nucl Sci Technol 1:24–27
Performance criteria for radiobioassay N13.30 (1996) ANSI/HPS, New York
Mughabghab SF (2003) Thermal neutron capture cross sections resonance integrals and G-factors. I.A.E.A., Vienna
Acknowledgments
This research was funded by the AECL Research and Development program. The authors wish to thank the Dosimetry Services Laboratory staff of Chalk River Laboratories for their assistance with this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guérin, N., Dai, X. Determination of 55Fe in urine by liquid scintillation counting. J Radioanal Nucl Chem 304, 1059–1069 (2015). https://doi.org/10.1007/s10967-015-4012-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-015-4012-x