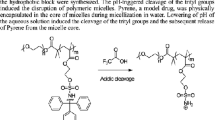
Abstract
A thiolactone-containing maleimide Tla-Mal is synthesized and used for the copolymerization with N-iso-propylacrylamide (NIPAAm) via RAFT polymerization to prepare thermo-responsive diblock copolymer PNIPAAm-b-P(Tla-Mal). Thiolactone groups on the side chains can be opened by primary amine, releasing thiol groups which can be used for further thiol-Michael addition reaction. Double postmodification of PNIPAAm-b-P(Tla-Mal) is revealed using mPEG11-NH2 as nucleophile for the thiolactone ring-opening reaction and light-responsive o-nitrobenzyl acrylate for the subsequent thiol-Michael addition reaction. Because of the introduction of light-responsive o-nitrobenzyl ester group, the double modified polymer shows light- and thermo-responsiveness. The responsiveness of PNIPAAm-b-P(Tla-Mal) and its double modified polymer PNIPAAm-b-P(Tla-Mal)DM is demonstrated. It provides an easy strategy for the design of tailor-made stimuli-responsive materials using thiolactone-containing maleimide.












Similar content being viewed by others
References
Ramasamy T, Ruttala HB, Gupta B, Poudel BK, Choi HG, Yong CS, Kim JO (2017) J Control Release 258:226–253
Bauri K, Nandi M, De P (2018) Polym Chem 9:1257–1287
Ekkelenkamp AE, Elzes MR, Engbersen JFJ, Paulusse JMJJ (2018) Mater Chem B 6:210–235
Lu CL, Urban MW (2018) Prog Polym Sci 78:24–46
Delplace V, Nicolas J (2015) Nat Chem 7:771–784
Seidi F, Jenjob R, Crespy D (2018) Chem Rev 118:3965–4036
Concellón A, Blasco E, Martínez-Felipe A, Martínez JC, Šics I, Ezquerra TA, Nogales A, Piñol M, Oriol L (2016) Macromolecules 49:7825–7836
Liu XC, Hu D, Jiang ZW, Zhuang JM, Xu YS, Guo XH, Thayumanavan S (2016) Macromolecules 49:6186–6192
Dutta K, Hu D, Zhao B, Ribbe AE, Zhuang JM, Thayumanavan S (2016) J Am Chem Soc 139:5676–5679
Wang K, Chen SL, Zhang WQ (2017) Macromolecules 50:4686–4698
Xia Y, Zeng Y, Hu D, Shen H, Deng J, Lu Y, Xia X, Xu W (2017) J Polym Sci Part A: Polym Chem 55:1773–1783
Hu D, Li Y, Niu Y, Li L, He J, Liu X, Xia X, Lu Y, Xiong Y, Xu W (2014) RSC Adv 4:47929–47936
Zhai S, Hu X, Hu Y, Wu B, Xing D (2017) Biomaterials 121:41–54
Wei H, Zhuo RX, Zhang XZ (2013) Prog Polym Sci 38:503–535
Krappitz T, Brauer D, Theato P (2016) Polym Chem 7:4525–4530
Huang X, Huang X, Yu A, Wang C, Dai Z, Xu Z (2011) Macromol Chem Phys 212:272–277
Lipshutz BH (2013) Bošković Z, Crowe CS, Davis VK, Whittemore HC, Vosburg DA, Wenzel AG. J Chem Edu 90:1514–1517
Stolz RM, Northrop BH (2013) J Org Chem 78:8105–8116
He L, Szopinski D, Wu Y, Luinstra GA, Theato P (2015) ACS Macro Lett 4:673–678
Huynh VT, Chen G, de Souza P, Stenzel MH (2011) Biomacromol 12:1738–1751
Dong Y, Saeed AO, Hassan W, Keigher C, Zheng Y, Tai H, Pandit A, Wang W (2012) Macromol Rapid Commun 33:120–126
Zhuang J, Jiwpanich S, Deepak VD, Thayumanavan S (2012) ACS Macro Lett 1:175–179
Das A, Theato P (2015) Chem Rev 116:1434–1495
Kakuchi R, Theato P (2015) Adv Polym Sci 269:133–162
Lin S, Shang J, Theato P (2018) ACS Macro Lett 7:431–436
Espeel P, Goethals F, Stamenović MM, Petton L, Du Prez FE (2012) Polym Chem 3:1007–1015
Reinicke S, Espeel P, Stamenović MM, Du Prez FE (2013) ACS Macro Lett 2:539–543
Stamenovic MM, Espeel P, Baba E, Yamamoto T, Tezuka Y, Du Prez FE (2013) Polym Chem 4:184–193
Chen Y, Espeel P, Reinicke S, Du Prez FE, Stenzel MH (2014) Macromol Rapid Commun 35:1128–1134
Ferris C, Casas M, Lucero MJ, de Paz MV, Jimenez-Castellanos MR (2014) Carbohydr Polym 111:125–132
Yan J, Wang R, Pan D, Yang R, Xu Y, Wang L, Yang M (2016) Polym Chem 7:6241–6249
Su Y, Tian L, Yu M, Gao Q, Wang D, Xi Y, Yang P, Lei B, Ma PX, Li P (2017) Polym Chem 8:3788–3800
Espeel P, Carrette LLG, Bury K, Capenberghs S, Martins JC, Du Prez FE, Madder A (2013) Angew Chem 125:13503–13506
Yu L, Wang L, Hu Z, You Y, Wu D, Hong C (2015) Polym Chem 6:1527–1532
Rudolph T, Espeel P, Du Prez FE, Schacher FH (2015) Polym Chem 6:4240–4251
Halperin A, Kröger M, Winnik FM (2015) Angew Chem Int Ed 54:15342–15367
Yu B, Chan JW, Hoyle CE, Lowe AB (2009) J Polym Sci Part A: Polym Chem 47:3544–3557
Postma A, Davis TP, Evans RA, Li G, Moad G, O’Shea MS (2006) Macromolecules 39:5293–5306
Keddie DJ, Moad G, Rizzardo E, Thang SH (2012) Macromolecules 45:5321–5342
Kwak Y, Nicolay R, Matyjaszewski K (2009) Macromolecules 42:3738–3742
Skey J, O’Reilly RK (2008) Chem Commun 4183–4185
Liu W, Greytak AB, Lee J, Wong CR, Park J, Marshall LF, Jiang W, Curtin PN, Ting AY, Nocera DG, Fukumura D, Jain RK, Bawendi MG (2010) J Am Chem Soc 132:472–483
Jiang J, Tong X, Morris D, Zhao Y (2006) Macromolecules 39:4633–4640
Xia Y, He H, Liu X, Hu D, Yin L, Lu Y, Xu W (2016) Polym Chem 7:6330–6339
Shen H, Xia Y, Qin Z, Wu J, Zhang L, Lu Y, Xia X, Xu W (2017) J Polym Sci Part A: Polym Chem 55:2770–2780
Park CY, Lim J, Yun MY, Kim C (2008) Angew Chem 120:3001–3005
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declare that she has no competing fnancial interests that could have appeared to infuence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lu, Y. Thermo-responsive diblock copolymer with pendant thiolactone group and its double postmodification. J Polym Res 29, 468 (2022). https://doi.org/10.1007/s10965-022-03298-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-022-03298-7