Abstract
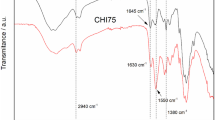
The present study utilized chitosan obtained from crab shell and transition metal salts as precursors to synthesize chitosan-metal coordination biopolymers of Mn(II), Fe(III), Co(II) and Ni(II) [i.e. Chit-Mn(II), Chit-Fe(III), Chit-Co(II) and Chit-NI(II) respectively]. The synthesized coordination biopolymers have been characterized using different instrumental techniques such as spectroscopic (UV–visible, FT-IR, XRD, EDS, and ICP-OES), thermal analysis (TGA and DTA), surface analysis (SEM), and hydrogen-temperature programmed reduction (H2-TPR) analysis. Spectroscopic studies confirmed the successful incorporation of the metals into the biopolymer matrix. Thermal analysis and H2-TPR revealed the reducibility of the Chit-Fe(III) at 120 ℃. While Chit-Fe(III) and Chit-Ni(II) were inactive, Chit-Co(II) and Chit-Mn(II) were found to be active towards vinyl acetate polymerization in the presence of aqueous Na2SO3. Furthermore, the polyvinyl acetate (PVAc) produced from Chit-Co(II) compared perfectly with a commercial PVAc and was in higher yield than PVAc produced from Chit-Mn(II). The polymerization has been shown to proceed via surface-initiated atom transfer radical polymerization (SI-ATRP), and the viscosity average molecular weight of PVAc produced has been measured as 25, 078. The density functional theory approach has been used to ascertain the coordination orientation of the Chit-Co(II) and explain its high efficiency towards vinyl acetate polymerization. The catalyst reusability test revealed an insignificant loss of activity for the Chit-Co(II) after seven cycles of polymerization. Kinetics and thermodynamic studies show that the vinyl acetate polymerization is an endothermic process and proceed at ambient temperature. The result of this work also suggests an investigation of chitosan-metal coordination biopolymer via low-ppm ATRP approach for possible biomedical application.












Similar content being viewed by others
References
Baig RBN, Nadagouda MN, Varma RS (2014) Ruthenium on Chitosan: A Recyclable Heterogeneous Catalyst for Aqueous Hydration of Nitriles to Amides. Green Chem 16(4):2122
Lamblin M, Nassar-Hardy L, Hierso J, Fouquet E, Felpin F (2010) Recyclable Heterogeneous Palladium Catalysts in Pure Water: Sustainable Developments in Suzuki, Heck, Sonogashira and Tsuji-Trost Reactions. Adv Synth Catal 352(1):33–79
Chtchigrovsky M, Primo A, Gonzalez P, Molvinger K, Robitzer M, Quignard F, Taran F (2009) Functionalized chitosan as a green, recyclable, biopolymer-supported catalyst for the [3+2] Huisgen cycloaddition. Angewandte Chemie (International ed. in English) 48(32):5916–5920
Ma J, Sahai Y (2013) Chitosan biopolymer for fuel cell applications. Carbohyd Polym 92(2):955–975
Pillai CKS, Paul W, Sharma CP (2009) Chitin and chitosan polymers: Chemistry, solubility and fiber formation. Prog Polym Sci 34(7):641–678
Zargar V, Asghari M, Dashti A (2015) A Review on Chitin and Chitosan Polymers: Structure, Chemistry, Solubility, Derivatives, and Applications. ChemBioEng reviews 2(3):204–226
Sun J, Wang J, Cheng W, Zhang J, Li X, Zhang S, She Y (2012) Chitosan Functionalized Ionic Liquid as a Recyclable Biopolymer-Supported Catalyst for Cycloaddition of CO2. Green Chem 14(3):654
Varma RS (2012) Greener approach to nanomaterials and their sustainable applications. Curr Opin Chem Eng 1(2):123–128
Lee M, Chen B, Den W (2015) Chitosan as a Natural Polymer for Heterogeneous Catalysts Support: A Short Review on Its Applications. Appl Sci 5(4):1272–1283
Baig RBN, Varma RS (2013) Copper on chitosan: a recyclable heterogeneous catalyst for azide–alkyne cycloaddition reactions in water. Green Chem 15(7):1839
Inaki Y, Otsuru M, Takemoto K (1978) Vinyl Polymerization by Metal Complexes. XXXI. Initiation by Chitosan-Copper(II) Complex. Journal of Macromolecular Science: Part A - Chemistry 12(7):953–970
Otsu T, Yoshida M, Tazaki T (2003) A model for Living Radical Polymerization. Die Makromolekulare Chemie, Rapid Commun 3:133–140
Boyer C, Corrigan NA, Jung K, Nguyen D, Nguyen T, Adnan NNM, Oliver S, Shanmugam S, Yeow J (2016) Copper-Mediated Living Radical Polymerization (Atom Transfer Radical Polymerization and Copper(0) Mediated Polymerization): From Fundamentals to Bioapplications. Chem Rev 116(4):1803–1949
Matyjaszewski K, Sumerlin BS, Tsarevsky NV, Chiefari J (2016) and American Chemical Society, Division of Polymer Chemistry Staff, Controlled Radical Polymerization: Mechanisms. American Chemical Society, Washington, DC
Tang W, Matyjaszewski K (2007) Effects of Initiator Structure on Activation Rate Constants in ATRP. Macromolecules 40(6):1858–1863
Wang Y, Kwak Y, Buback J, Buback M, Matyjaszewski K (2012) Determination of ATRP Equilibrium Constants under Polymerization Conditions. ACS Macro Lett 1(12):1367–1370
Matyjaszewski K, Tsarevsky NV (2014) Macromolecular Engineering by Atom Transfer Radical Polymerization. J Am Chem Soc 136(18):6513–6533
Morick J, Buback M, Matyjaszewski K (2012) Effect of Pressure on Activation-Deactivation Equilibrium Constants for ATRP of Methyl Methacrylate. Macromol Chem Phys 213(21):2287–2292
Zhang Y, Wang Y, Peng C, Zhong M, Zhu W, Konkolewicz D, Matyjaszewski K (2012) Copper-Mediated CRP of Methyl Acrylate in the Presence of Metallic Copper: Effect of Ligand Structure on Reaction Kinetics. Macromolecules 45(1):78–86
Li M, Matyjaszewski K (2003) Reverse Atom Transfer Radical Polymerization in Miniemulsion. Macromolecules 36(16):6028–6035
Li M, Jahed NM, Min K, Matyjaszewski K (2004) Preparation of Linear and Star-Shaped Block Copolymers by ATRP Using Simultaneous Reverse and Normal Initiation Process in Bulk and Miniemulsion. Macromolecules 37(7):2434–2441
Li M, Min K, Matyjaszewski K (2004) ATRP in Waterborne Miniemulsion via a Simultaneous Reverse and Normal Initiation Process. Macromolecules 37(6):2106–2112
Min K, Gao H, Matyjaszewski K (2005) Preparation of Homopolymers and Block Copolymers in Miniemulsion by ATRP Using Activators Generated by Electron Transfer (AGET). J Am Chem Soc 127(11):3825–3830
Yamamura Y, Matyjaszewski K (2007) Methylaluminoxane as a Reducing Agent for Activators Generated by Electron Transfer ATRP. J Macromol Sci Part A 44(9):1035–1039
Jakubowski W, Matyjaszewski K (2005) Activator Generated by Electron Transfer for Atom Transfer Radical Polymerization. Macromolecules 38(10):4139–4146
Kwak Y, Magenau AJD, Matyjaszewski K (2011) ARGET ATRP of Methyl Acrylate with Inexpensive Ligands and ppm Concentrations of Catalyst. Macromolecules 44(4):811–819
Simakova A, Averick SE, Konkolewicz D, Matyjaszewski K (2012) Aqueous ARGET ATRP. Macromolecules 45(16):6371–6379
Dong H, Matyjaszewski K (2008) ARGET ATRP of 2-(Dimethylamino)ethyl Methacrylate as an Intrinsic Reducing Agent. Macromolecules 41(19):6868–6870
Konkolewicz D, Magenau AJD, Averick SE, Simakova A, He H, Matyjaszewski K (2012) ICAR ATRP with ppm Cu Catalyst in Water. Macromolecules 45(11):4461–4468
D'hooge DR, Konkolewicz D, Reyniers M, Marin GB, Matyjaszewski K (2012) Kinetic Modeling of ICAR ATRP. Macromol Theory Simul 21(1):52–69
Williams VA, Ribelli TG, Chmielarz P, Park S, Matyjaszewski K (2015) A Silver Bullet: Elemental Silver as an Efficient Reducing Agent for Atom Transfer Radical Polymerization of Acrylates. J Am Chem Soc 137(4):1428–1431
Konkolewicz D, Krys P, Góis JR, Mendonça PV, Zhong M, Wang Y, Gennaro A, Isse AA, Fantin M, Matyjaszewski K (2014) Aqueous RDRP in the Presence of Cu0: The Exceptional Activity of CuI Confirms the SARA ATRP Mechanism. Macromolecules 47(2):560–570
Abreu CMR, Serra AC, Popov AV, Matyjaszewski K, Guliashvili T, Coelho JFJ (2013) Ambient temperature rapid SARA ATRP of acrylates and methacrylates in alcohol–water solutions mediated by a mixed sulfite/Cu(ii)Br 2 catalytic system. Polym Chem 4(23):5629
Zhang Q, Wilson P, Li Z, McHale R, Godfrey J, Anastasaki A, Waldron C, Haddleton DM (2013) Aqueous Copper-Mediated Living Polymerization: Exploiting Rapid Disproportionation of CuBr with Me6TREN. J Am Chem Soc 135(19):7355–7363
Waldron C, Anastasaki A, McHale R, Wilson P, Li Z, Smith T, Haddleton DM (2014) Copper-mediated living radical polymerization (SET-LRP) of lipophilic monomers from multi-functional initiators: reducing star–star coupling at high molecular weights and high monomer conversions. Polym Chem 5(3):892–898
Samanta SR, Cai R, Percec V (2015) A rational approach to activated polyacrylates and polymethacrylates by using a combination of model reactions and SET-LRP of hexafluoroisopropyl acrylate and methacrylate. Polym Chem 6(17):3259–3327
Samanta SR, Sun H, Anastasaki A, Haddleton DM, Percec V (2014) Self-activation and activation of Cu(0) wire for SET-LRP mediated by fluorinated alcohols. Polym Chem 5(1):89–95
Samanta SR, Cai R, Percec V (2014) SET-LRP of semifluorinated acrylates and methacrylates. Polym Chem 5(18):5479–5491
Waldron C, Zhang Q, Li Z, Nikolaou V, Nurumbetov G, Godfrey J, McHale R, Yilmaz G, Randev RK, Girault M, McEwan K, Haddleton DM, Droesbeke M, Haddleton AJ, Wilson P, Simula A, Collins J, Lloyd DJ, Burns JA, Summers C, Houben C, Anastasaki A, Li M, Becer CR, Kiviaho JK, Risangud N (2014) Absolut “copper catalyzation perfected”; robust living polymerization of NIPAM: Guinness is good for SET-LRP. Polym Chem 5(1):57–61
Xue L, Lyu Z, Shi X, Tang Z, Chen G, Chen H (2014) Fast and Green Synthesis of a Smart Glyco-surface via Aqueous Single Electron Transfer-Living Radical Polymerization. Macromol Chem Phys 215(15):1491–1497
Lepoittevin B, Elzein T, Dragoe D, Bejjani A, Lemée F, Levillain J, Bazin P, Roger P, Dez I (2019) Hydrophobization of chitosan films by surface grafting with fluorinated polymer brushes. Carbohyd Polym 205:437–446
Bhattacharjee M, Pramanik NB, Singha NK, Haloi DJ (2020) Recent advances in RDRP-modified chitosan: a review of its synthesis, properties and applications. Polym Chem 11(42):6718–6738
Zhou J, Zhou X, Dong Z, Yang H, Shi Z, Li R (2013) Pd immobilized on magnetic chitosan as a heterogeneous catalyst for acetalization and hydrogenation reactions. Appl Surf Sci 279:360–366
Nurudeen SA, O. Sanyaolu Saliu A. Amolegbe Abdulahi O. Sobola Olujinmi M. Folarin, (2012) Poly[β- (1→4) -2-amino-2-deoxy-D-glucopyranose] based zero valent nickel nanocomposite for efficient reduction of nitrate in water. J Environ Sci 24(9):1702–1708
Sun L, Zhang L, Liang C, Yuan Z, Zhang Y, Xu W, Zhang J, Chen Y (2011) Chitosan modified Fe0 nanowires in porous anodic alumina and their application for the removal of hexavalent chromium from water. J Mater Chem 21(16):5877
Yi S, Lee D, Lee Y, Sin E (2007) Chitosan-supported palladium(0) catalyst for microwave-prompted Suzuki cross-coupling reaction in water. Tetrahedron Lett 48(38):6771–6775
Alexandra AP, Mansur HS, Ramanery FP, Oliveira LC, Souza PP (2014) “Green” colloidal ZnS quantum dots/chitosan nano-photocatalysts for advanced oxidation processes: Study of the photodegradation of organic dye pollutants. Appl Catal B 158–159:269–279
Khoza P, Nyokong T (2015) “Photocatalytic behaviour of zinc tetraamino phthalocyanine-silver nanoparticles immobilized on chitosan beads,” . J Mol Catal Chem 399:25–32
Huai-min G, Xian-su C (2004) Study of cobalt(II)-chitosan coordination polymer and its catalytic activity and selectivity for vinyl monomer polymerization. Polym Adv Technol 15(1–2):89–92
Adewuyi S, Bisiriyu I, Akinremi C, Amolegbe S (2017) Synthesis, Spectroscopic, Surface and Catalytic Reactivity of Chitosan Supported Co(II) and Its Zerovalentcobalt Nanobiocomposite. J Inorg Organomet Polym Mater 27(1):114–121
Adewuyi S, Bisiriyu IO, Akinremi CA (2015) Zinc(II) metal ion complexes of Chitosan: toward heterogeneous active catalyst for the polymerization of vinyl acetate. IFE J Sci 17(3):749–754
Su B, Feng G (2010) Influence of the metal centers of 2,6-bis(imino)pyridyl transition metal complexes on ethylene polymerization/ oligomerization catalytic activities. Polym Int 59:1058–1063
Thien DT, An NT, Hoa NT (2015) Preparation of Fully Deacetylated Chitosan for Adsorption of Hg(II) Ion from Aqueous Solution. Chem Sci J 6(2):95
Czechowska-Biskup R, Jarosińska D, Rokita B, Ulański P, Rosiak JM (2012) Determination of degree of deacetylation of chitosan - comparision of methods. Progress on Chemistry 12:5–20
Wu T, Zivanovic S (2008) Determination of the degree of acetylation (DA) of chitin and chitosan by an improved first derivative UV method. Carbohyd Polym 73(2):248–253
Nikolaienko TY, Bulavin LA, Hovorun DM (2014) Comp. Theor. Chem. J Comput Chem 1050:15–22
Nikolaienko TY, Bulavin LA (2019) Localized orbitals for optimal decomposition of molecular properties. Int J Quantum Chem 119(3):e25798
Bisiriyu IO, Meijboom R (2020) Adsorption of Cu(II) ions from aqueous solution using pyridine-2,6-dicarboxylic acid crosslinked chitosan as a green biopolymer adsorbent. Int J Biol Macromol 165:2484–2493
Hussaini G (2020) Yakasai Jamila Bashir, Waziri Ibrahim, Bisiriyu Ibraheem O, “Transition Metal Complexes of Schiff Base Ligand Derived from Trimethprim with Cyclohexanone: Synthesis, Characterization, Antimicrobial and Computational Studies,.” Acta Scientific Pharmaceutical Sciences 4(5):36–45
Semsarzadeh MA, Sabzevari A (2018) Highly effective organometallic-mediated radical polymerization of vinyl acetate using alumina-supported Co(acac)2catalyst: A case study of adsorption and polymerization. J Appl Polym Sci 135(13):46057
Sharma R, Bisen DP, Shukla U, Sharma BG (2012) X-ray diffraction: a powerful method of characterizing nanomaterials. Recent Res Sci Technol 4(8):77–79
Gökalp F (2021) An Investigation into the Usage of Monosaccharides with GLUT1 and GLUT3 as Prognostic Indicators for Cancer. Nutr Cancer, vol. ahead-of-print, no. ahead-of-print, pp 1–5
Gokalp F (2018) The inhibition effect of garlic-derived compounds on human immunodeficiency virus type 1 and saquinavir. J Biochem Mol Toxicol 32(11):e22215
Gökalp F (2018) An investigation of the olive phenols activity as a natural medicine. Yàowu ship in fenxi 26(2):657–661
Acknowledgements
The financial assistance of The World Academy of Sciences (TWAS) in collaboration with the National Research Foundation (NRF) and the Department of Science and Technology (DST) of the Republic of South Africa towards this research is hereby acknowledged. Opinions expressed and conclusions arrived at are those of the authors and are not necessarily to be attributed to the DST-NRF-TWAS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
There are no conflicts to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bisiriyu, I.O., Meijboom, R. Chitosan-transition metal coordination biopolymer: a promising heterogeneous catalyst for radical ion polymerization of vinyl acetate at ambient temperature. J Polym Res 28, 363 (2021). https://doi.org/10.1007/s10965-021-02683-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-021-02683-y