Abstract
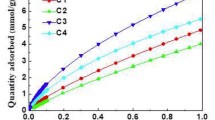
It is known that the use of the Dubinin–Radushkevich method in micro-mesoporous samples does not give adequate values of micropore volumes, unlike when the samples contain only microporous. Based on that, in this work, we propose an easy method to calculate a reliable micropore volume (VμP) of micro-mesoporous (nanopores) samples, separating the microporous region from the experimental isotherm. For this, the original isotherm is modified, estimating the thickness of the adsorbed layer (t) as a function of relative pressure and changing the external surface area (Sext) to obtain a Type I adsorption isotherm in the microporous region; then, the DR method can be applied to the modified isotherm. This proposal, named the DR_t method, allows the calculation of a reliable VμP of any nanoporous material using different adsorbates. Using this method, we analyzed adsorbents of distinct nature (i.e., carbons and silicas) with different adsorbates as N2 and O2 at 77 K, Ar at 87 K, and CO2 at 273 K. We used this method to calculate VμP in different samples and compare them with those obtained with the traditional DR method, highlighting that unlike the latter the DR_t method showed similar and consistent results with the different adsorbates. Therefore, the values of micropore volume calculated using the DR_t method demonstrate consistency across various adsorbates, not only for N2 but especially for CO2, which is suggested to analyze narrow micropore volumes.






Similar content being viewed by others
Data availability
Not applicable.
References
M. Thommes, K. Kaneko, A.V. Neimark, J.P. Olivier, F. Rodriguez-Reinoso, J. Rouquerol, K.S.W. Sing, Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 87, 1051–1069 (2015)
K.Y. Montiel-Centeno, D. Barrera, J. Villarroel-Rocha, M.S. Moreno, K. Sapag, Hierarchical nanostructured carbons as CO2 adsorbents. Adsorption 25, 1287–1297 (2019)
M. Sevilla, J.B. Parra, A.B. Fuertes, Assessment of the role of micropore size and N-doping in CO2 capture by porous carbons. Appl. Mater. Interfaces 5, 6360–6368 (2013)
M.E. Casco, M. Martínez-Escandell, J. Silvestre-Albero, F. Rodríguez-Reinoso, Effect of the porous structure in carbon materials for CO2 capture at atmospheric and high-pressure. Carbon 67, 230–235 (2014)
G. Srinivas, V. Krungleviciute, Z.-X. Guo, T. Yildirim, Exceptional CO2 capture in a hierarchically porous carbon with simultaneous high surface area and pore volume. Energy Environ. Sci. 7, 335–342 (2014)
G. Sethia, A. Sayari, Comprehensive study of ultra-microporous nitrogen-doped activated carbon for CO2 capture. Carbon 93, 68–80 (2015)
J. Serafin, U. Narkiewicz, A.W. Morawski, R.J. Wróbel, B. Michalkiewicz, Highly microporous activated carbons from biomass for CO2 capture and effective micropores at different conditions. J. CO2 Util. 18, 73–79 (2017)
A.A. García Blanco, J.C. Alexandre de Oliveira, R. López, J.C. Moreno-Piraján, L. Giraldo, G. Zgrablich, K. Sapag, A study of the pore size distribution for activated carbon monoliths and their relationship with the storage of methane and hydrogen. Colloids Surf. A Physicochem. Eng. Asp. 357, 74–83 (2010)
J. Serafin, B. Dziejarski, O.F. Cruz Junior, J. Sreńscek-Nazzal, Design of highly microporous activated carbons based on walnut shell biomass for H2 and CO2 storage. Carbon 201, 633–647 (2023)
J.S. Mirzaei, A. Ahmadpour, A. Shahsavand, A.N. Pour, L. Lotfi Katooli, A.G. Asil, B. Pouladi, A. Arami-Niya, Experimental and simulation study of the effect of surface functional groups decoration on CH4 and H2 storage capacity of microporous carbons. Appl. Surf. Sci. 533, 147487 (2020)
L. Zubizarreta, E.I. Gomez, A. Arenillas, C.O. Ania, J.B. Parra, J.J. Pis, H2 storage in carbon materials. Adsorption 14, 557–566 (2008)
L. Zubizarreta, A. Arenillas, J.J. Pis, Carbon materials for H2 storage. Adsorption 34, 4575–4581 (2009)
S. Stelitano, G. Conte, A. Policicchio, A. Aloise, G. Desiderio, R.G. Agostino, Low pressure methane storage in pinecone-derived activated carbons. Energy Fuels 32, 10891–10897 (2018)
E. Mahmoud, Evolution of the design of CH4 adsorbents. Surfaces 3, 433–466 (2020)
F. Béguin, V. Presser, A. Balducci, E. Frackowiak, Carbons and electrolytes for advanced supercapacitors. Adv. Mater. 26, 2219–2251 (2014)
P. Ratajczak, M.E. Suss, F. Kaasik, F. Béguin, Carbon electrodes for capacitive technologies. Energy Stor. Mater. 16, 126–145 (2019)
D. Barrera, M. Florent, M. Kulko, T.J. Bandosz, Ultramicropore-influenced mechanism of oxygen electroreduction on metal-free carbon catalysts. J. Matter. Chem. A. 7, 27110–27123 (2019)
M.J. Remy, G. Poncelet, A new approach to the determination of the external surface and micropore volume of zeolites from the nitrogen adsorption isotherm at 77 K. J. Phys. Chem. 99, 773–779 (1995)
E. Falabella Sousa-Aguiar, A. Liebsch, B.C. Chaves, A.F. Costa, Influence of the external surface area of small crystallite zeolites on the micropore volume determination. Microp. Mesop. Mater. 25, 185–192 (1998)
S. Brouwer, J.C. Groen, L.A.A. Peffer, The impact of mesoporosity on microporosity assessment by CO2 adsorption. Stud. Surf. Sci. Catal. 160, 145–152 (2007)
J. Villarroel-Rocha, D. Barrera, A.A. García Blanco, M.E. Roca Jalil, K. Sapag, Importance of the αS-plot method in the characterization of nanoporous materials. Adsorp. Sci. Technol. 31, 165–183 (2013)
M.M. Dubinin, O. Kadlec, New ways in determination of the parameters of porous structure of microporous carbonaceous adsorbents. Carbon 13, 263–265 (1975)
M.M. Dubinin, The potential theory of adsorption of gases and vapors for adsorbents with energetically nonuniform surfaces. Chem. Rev. 60, 235–241 (1960)
J.M. Martín-Martínez, Physical adsorption of gases and vapors by coals, University of Alicante, Doctoral thesis, 1990 http://hdl.handle.net/10045/4291
J. Villarroel-Rocha, Adsorción de diferentes gases y vapores en la caracterización textural y estructural de materiales nanoporosos (Adsorption of different gases and vapors in the textural and structural characterization of nanoporous materials), Universidad Nacional de San Luis, Doctoral thesis, 2015 http://biblioteca.unsl.edu.ar/baea/search2.html?bool=Jhonny%20and%20Villaroel%20and%20Rocha&tipo=&biblio=B1
J. Choma, J. Jagiello, M. Jaroniec, Assessing the contribution of micropores and mesopores from nitrogen adsorption on nanoporous carbons: application to pore size analysis. Carbon 183, 150–157 (2021)
M. Jaroniec, M. Kruk, J.P. Olivier, Standard nitrogen adsorption data for characterization of nanoporous silicas. Langmuir 15, 5410–5413 (1999)
M. Kruk, M. Jaroniec, Accurate method for calculating mesopore size distributions from argon adsorption data at 87 K developed using model MCM-41 materials. Chem. Mater. 12, 222–230 (2000)
M. Kruk, M. Jaroniec, Determination of mesopore size distributions from argon adsorption data at 77 K. J. Phys. Chem. B 106, 4732–4739 (2002)
M. Kruk, M. Jaroniec, K.P. Gadkaree, Nitrogen adsorption studies of novel synthetic active carbons. J. Colloid Interf. Sci. 192, 250–256 (1997)
J. Jagiello, C. Ania, J.B. Parra, C. Cook, Dual gas analysis of microporous carbons using 2D-NLDFT heterogeneous surface model and combined adsorption data of N2 and CO2. Carbon 91, 330–337 (2015)
J. Jagiello, J. Kenvin, Consistency of carbon nanopore characteristics derived from adsorption of simple gases and 2D-NLDFT models. advantages of using adsorption isotherms of oxygen (O2) at 77 K. J. Colloid Interf. Sci. 542, 151–158 (2019)
K. Nakai, M. Yoshida, J. Sonoda, Y. Nakada, M. Hakuman, H. Naono, High resolution N2 adsorption isotherms by graphitized carbon black and nongraphitized carbon black – αS-Curves, adsorption enthalpies and entropies. J. Colloid Interf. Sci. 351, 507–514 (2010)
L. Gardner, M. Kruk, M. Jaroniec, Reference data for argon adsorption on graphitized and nongraphitized carbon blacks. J. Phys. Chem. B 105, 12516–12523 (2001)
M.J.G. De Araújo, J. Villarroel-Rocha, V.C. De Souza, K. Sapag, S.B.C. Pergher, Carbon foams from sucrose employing different metallic nitrates as blowing agents: application in CO2 capture. J. Anal. Appl. Pyrolysis 141, 104627 (2019)
J. Villarroel-Rocha, D. Barrera, J.J. Arroyo-Gómez, K. Sapag, Insights of adsorption isotherms with different gases at 77 K and their use to assess the BET area of nanoporous silica materials. Adsorption 27, 1081–1093 (2021)
J.J. Garrido, J.M. Martín-Martínez, M. Molina-Sabio, F. Rodríguez-Reinoso, R. Torregosa, Adsorption of hydrocarbons on CO2-reacted activated carbons. Carbon 24, 469–475 (1986)
Acknowledgements
The authors wish to thank UNSL, ANPCyT and CONICET for the financial support of this work. The authors also thank Dr. Jacek Jagiello from Micromeritics Instrument Corporation for providing the reference isotherm data of CABOT BP280 for CO2 and O2 at 273 and 77 K, respectively.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
J.V.R and K.S.: conceptualization; J.V.R: methodology; J.V.R, J.J.A.G., and D.B.: formal analysis; K.S.: resources; J.V.R, J.J.A.G., and D.B.: data curation; J.V.R, J.J.A.G., and D.B.: writing original draft; J.V.R, J.J.A.G., D.B., and K.S.: writing‒review & editing. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Declarations
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Villarroel-Rocha, J., Arroyo-Gómez, J.J., Barrera, D. et al. A new and reliable method to obtain micropore volume in nanoporous solids by gas adsorption based on Dubinin works and the thickness of the adsorbed layer. J Porous Mater (2024). https://doi.org/10.1007/s10934-024-01573-0
Published:
DOI: https://doi.org/10.1007/s10934-024-01573-0