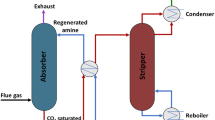
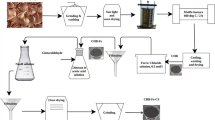
Abstract
The adsorption/regeneration kinetics of CO2 capture by amine-functionalized SBA-16 was presented to understand the potential mechanism of adsorption of CO2 by adsorption column. Three adsorption kinetic models were used to simulate the isothermal CO2 adsorption data. Based on the value of the correlation coefficient R 2, the fractional order model is considered to be the most suitable model for the CO2 adsorption performance at different temperatures and CO2 concentrations. The results of kinetic analysis show that the adsorption process follows the pseudo second order reaction, and the higher concentration of CO2 or operating temperature will promote the diffusion of CO2 into the adsorbent. Steam stripping and VTSA regeneration were carried out to study the behavior of adsorbent regeneration. Results show that both regeneration methods can achieve rapid regeneration of the adsorbent, and temperature is a major factor in the regeneration of adsorbent due to the chemical bond between CO2 and adsorbents. The Avrami fractional-order models simulates the experimental data well. The absolute value of the activation energy obtained from the Arrhenius equation for CO2 desorption was 59.05 kJ/mol.







Similar content being viewed by others
References
M.E. Boot-Handford, J.C. Abanades, E.J. Anthony, M.J. Blunt, S. Brandani, N. Mac Dowell, J.R. Fernández, M.-C. Ferrari, R. Gross, J.P. Hallett, R.S. Haszeldine, P. Heptonstall, A. Lyngfelt, Z. Makuch, E. Mangano, R.T.J. Porter, M. Pourkashanian, G.T. Rochelle, N. Shah, J.G. Yao, P.S. Fennell, Energy Environ. Sci. 7, 130 (2014)
T.-H. Bae, M.R. Hudson, J.a.. Mason, W.L. Queen, J.J. Dutton, K. Sumida, K.J. Micklash, S.S. Kaye, C.M. Brown, J.R. Long, Energy Environ. Sci. 6, 128 (2013)
D.M. D’Alessandro, B. Smit, J.R. Long, Angew. Chem. Int. Ed. 49, 6058 (2010)
Q. Wang, J. Luo, Z. Zhong, A. Borgna, Energy Environ. Sci. 4, 42 (2011)
A. Zukal, I. Dominguez, J. Mayerová, J. Čejka, Langmuir 25, 10314 (2009)
S. Chen, Y. Fu, Y. Huang, Z. Tao, J. Porous Mater. 23, 713 (2016)
B. Ma, L. Zhuang, S. Chen, J. Porous Mater. 23, 529 (2016)
J. Li, M. Hitch, Powder Technol. 291, 408 (2016)
R. Kishor, A.K. Ghoshal, Ind. Eng. Chem. Res. 56, (2017)
M. Ranjbar, M.A. Taher, A. Sam, J. Porous Mater. 23, 375 (2016)
K. Sumida, D.L. Rogow, J.A. Mason, T.M. Mcdonald, E.D. Bloch, Z.R. Herm, T.H. Bae, J.R. Long, Chem. Rev. 112, 724 (2012)
Y. Jing, L. Wei, Y. Wang, Y. Yu, Microporous Mesoporous Mater. 183, 124 (2014)
C. Chen, Y.R. Lee, W.S. Ahn, J. Nanosci. Nanotechnol. 16, 4291 (2016)
A.E. Creamer, B. Gao, Environ. Sci. Technol. 50, 7276 (2016)
Y.C. Chiang, R.S. Juang, J. Taiwan Inst. Chem. Eng. 71, (2016)
Y. Liu, Y. Chen, L. Tian, R. Hu, J. Porous Mater. 24, 583 (2017)
M.K.R. Reddy, Z.P. Xu, G.Q. Lu, J.C.D. Da Costa, Ind. Eng. Chem. Res. 45, 7504 (2006)
D.V. Quang, A. Dindi, A.V. Rayer, N. El Hadri, A. Abdulkadir, M.R.M. Abu-Zahra, Greenh. Gases Sci. Technol. 5, 91 (2015)
D.V. Quang, N. El Hadri, M.R.M. Abu-zahra, Eur. Sci. J. 9, 82 (2013)
G. Evano, Angew. Chem. Int. Ed. 49, 6025 (2010)
E.E. Ünveren, B.Ö. Monkul, Ş. Sarıoğlan, N. Karademir, E. Alper, Petroleum 3, 37 (2016)
P. Bollini, S.A. Didas, C.W. Jones, J. Mater. Chem. 21, 15100 (2011)
R. Serna-Guerrero, A. Sayari, Chem. Eng. J. 161, 182 (2010)
A. Samanta, A. Zhao, G.K.H. Shimizu, P. Sarkar, R. Gupta, Ind. Eng. Chem. Res. 51, 1438 (2012)
Y. Liu, X. Lin, X. Wu, M. Liu, R. Shi, X. Yu, Powder Technol. 318, 186 (2017)
W. Li, S. Choi, J. Drese, M. Hornbostel, G. Krishnan, P. Eisenberger, C. Jones, ChemSusChem 3, 899 (2010)
Y. Liu, Q. Ye, M. Shen, J. Shi, J. Chen, H. Pan, Y. Shi, Environ. Sci. Technol. 45, 5710 (2011)
L. Ecn, D.A. Fsc, V. Efs, A.R. Cestari, J. Colloid Interface Sci. 263, 542 (2003)
I. Kinefuchi, H. Yamaguchi, Y. Sakiyama, S. Takagi, Y. Matsumoto, J. Chem. Phys. 128, 164712 (2008)
Q. Liu, J. Shi, S. Zheng, M. Tao, H. Yi, S. Yao, Ind. Eng. Chem. Res. 53, 11677 (2014)
S. Sircar, J.R. Hufton, Adsorption 6, 137 (2000)
A. Heydari-Gorji, A. Sayari, Chem. Eng. J. 173, 72 (2011)
X. Xu, C. Song, J.M. Andresen, B.G. Miller, A.W. Scaroni, Energy Fuels 16, 1463 (2002)
X. Xu, C. Song, B.G. Miller, A.W. Scaroni, Ind. Eng. Chem. Res. 44, 8113 (2005)
J.C. Hicks, J.H. Drese, D.J. Fauth, M.L. Gray, G. Qi, C.W. Jones, J. Am. Chem. Soc. 130, 2902 (2008)
M. Avrami, J. Chem. Phys. 7, 1103 (1939)
Z. Sun, M. Fan, M. Argyle, Energy Fuels 25, 2988 (2011)
Acknowledgements
We gratefully acknowledge the financial support of the Science Foundation of Fujian Province (Grant Nos. 2015J01033, 2017J01472, 2017 J01568), the Scientific Research Foundation of the Fujian University of Technology (Grant No. GY-Z11045), and the Science Foundation of Fuzhou (Grant No. GY-Z17059).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wu, X., Liu, M., Shi, R. et al. CO2 adsorption/regeneration kinetics and regeneration properties of amine functionalized SBA-16. J Porous Mater 25, 1219–1227 (2018). https://doi.org/10.1007/s10934-017-0532-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10934-017-0532-9