Abstract
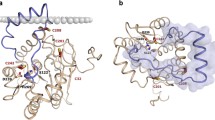
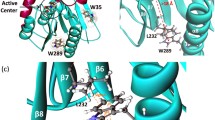
Human monoacylglycerol lipase (MGL) catalyzes the hydrolysis of 2-arachidonoylglycerol to arachidonic and glycerol, which plays a pivotal role in the normal biological processes of brain. Co-crystal structure of the MGL in complex with its inhibitor, compound 1, shows that the helix α4 undergoes large-scale conformational changes in response to the compound 1 binding compared to the apo MGL. However, the detailed conformational transition pathway of the helix α4 in the inhibitor binding process of MGL has remained unclear. Here, conventional molecular dynamics (MD) and nudged elastic band (NEB) simulations were performed to explore the conformational transition pathway of the helix α4. Conventional MD simulations unveiled that the compound 1 induced the closed conformation of the active site of MGL, reduced the conformational flexibility of the helix α4, and elicited the large-scale conformational rearrangement of the helix α4, leading to the complete folding of the helix α4. Moreover, NEB simulations revealed that the conformational transition pathway of helix α4 underwent an almost 180° counter-clockwise rotation of the helix α4. Our computational results advance the structural and mechanistic understanding of the inhibitory mechanism.







Similar content being viewed by others
Abbreviations
- 2-AG:
-
2-Arachidonoylglycerol
- CNS:
-
Central nervous system
- DCCM:
-
Dynamical cross-correlation matrices
- DSSP:
-
Defined secondary structure of proteins
- MD:
-
Molecular dynamics
- MGL:
-
Monoacylglycerol lipase
- NEB:
-
Nudged elastic band
- RMSD:
-
Root-mean-square deviation
- RMSF:
-
Root-mean-square fluctuation
References
Matsuda LA, Lolait SJ, Brownstein MJ, Young AC, Bonner TI (1990) Structure of a cannabinoid receptor and functional expression of the cloned cDNA. Nature 346:561–564
Munro S, Thomas KL, Abu-Shaar M (1993) Molecular characterization of a peripheral receptor for cannabinoids. Nature 365:61–65
Karlsson M, Contreras JA, Hellman U, Tornqvist H, Holm C (1997) cDNA cloning, tissue distribution, and identification of the catalytic traid of monoglyceride. Evolutionary relationship to esterases, lysophospholipases, and haloperoxidases. J Biol Chem 272:27218–27223
Dinh TP, Carpenter D, Leslie FM, Freund TF, Katona I, Sensi SL, Kathuria S, Piomelli D (2002) Brain monoglyceride lipase participating in endocannabinoid inactivation. Proc Natl Acad Sci USA 99:10819–10824
Saario SM, Laitinen JT (2007) Monoglyceride lipase as an enzyme hydrolyzing 2-arachidonoylglycerol. Chem Biodivers 4:1903–1913
Chon SH, Zhou YX, Dixon JL, Storch J (2007) Intestinal monoacylglycerol metabolism: developmental and nutritional regulation of monoacylglycerol lipase and monoacylglycerol acyltransferase. J Biol Chem 282:33346–33357
Dinh TP, Freund TF, Piomelli D (2002) A role for monoglyceride lipase in 2-arachidonoylglycerol inactivation. Chem Phys Lipids 121:149–158
Gonsiorek W, Lunn C, Fan X, Narula S, Lundell D, Hipkin RW (2000) Endocannabinoid 2-arachidonyl glycerol is a full agonist through human type 2 cannabinoid receptor: antagonism by anandamide. Mol Pharmacol 57:1045–1050
Maresz K, Carrier EJ, Ponomarev ED, Hillard CJ, Dittel BN (2005) Modulation of the cannabinoid CB2 receptor in microglial cells in response to inflammatory stimuli. J Neurochem 95:437–445
Vandevoorde S, Jonsson KO, Labar G, Persson E, Lambert DM, Fowler CJ (2007) Lack of selectivity of URB602 for 2-oleoylglycerol compared to anandamide hydrolysis in vitro. Br J Pharmacol 150:186–191
Long JZ, Li W, Booker L, Burston JJ, Kinsey SG, Schlosburg JE, Pavón FJ, Serrano AM, Selley DE, Parsons LH, Lichtman AH, Cravatt BF (2009) Selective blockade of 2-arachidonoylglycerol hydrolysis produces cannabinoid behavioral effects. Nat Chem Biol 5:37–44
Saario SM, Salo OM, Nevalainen T, Poso A, Laitinen JT, Jarvinen T, Niemi R (2005) Characterization of the sulfhydryl-sensitive site in the enzyme responsible for hydrolysis of 2-arachidonoyl-glycerol in rat cerebellar membranes. Chem Biol 12:649–656
Bertrand T, Augé F, Houtmann J, Rak A, Vallée F, Mikol V, Berne PF, Michot N, Cheuret D, Hoornaert C, Mathieu M (2010) Structural basis for human monoglyceride lipase inhibition. J Biol Chem 396:663–673
Schalk-Hihi C, Schubert C, Alexander R, Bayoumy S, Clemente JC, Deckman I, DesJarlais RL, Dzordzorme KC, Flores CM, Grasberger B, Kranz JK, Lewandowski F, Liu L, Ma H, Maguire D, Macielag MJ, McDonnell ME, Haarlander TM, Miller R, Millgan C, Reynolds C, Kuo LC (2011) Crystal structure of a soluble form of human monoglyceride lipase in complex with an inhibitor at 1.35 Å resolution. Protein Sci 20:670–683
Labar G, Bauvois C, Borel F, Ferrer J-L, Wouters J, Lambert DM (2010) Crystal structure of the human monoacylglycerol lipase, a key actor in endocannabinoid signaling. ChemBioChem 11:218–227
Case DA, Darden TA, Cheatham TE III, Simmerling CL, Wang J, Duke RE, Luo R, Walker RC, Zhang W, Merz KM, Roberts BP, Wang B, Hayik S, Roitberg A, Seabra G, Kolossvary I, Wong KF, Paesani F, Vanicek J, Liu J, Wu X, Brozell SR, Steinbrecher T, Gohlke H, Cai Q, Ye X, Wang J, Hsieh MJ, Cui G, Roe DR, Mathews DH, Seetin MG, Sagui C, Babin V, Luchko T, Gusarov S, Kovalenko A, Kollman PA (2010) AMBER 11. University of California, San Francisco
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Meuuucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA, Peralta Jr. JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers JJHE, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09. Gaussian Inc., Wallingford
Duan Y, Wu C, Chowdhury S, Lee MC, Xiong G, Zhang W, Yang R, Cieplak P, Luo R, Lee T (2003) A point-charge force field for molecular mechanics simulations of proteins. J Comput Chem 24:1999–2012
Jorgensen WL, Chandrasekhar J, Madura JD, Impey RW, Klein ML (1983) Comparison of single potential function for simulating liquid water. J Chem Phys 79:926–935
Lu S-Y, Huang Z-M, Huang W-K, Liu X-Y, Chen Y–Y, Shi T, Zhang J (2013) How calcium inhibits the magnesium-dependent kinase GSK3β: a molecular simulation study. Proteins 81:740–753
Lu S, Huang W, Li X, Huang Z, Liu X, Chen Y, Shi T, Zhang J (2012) Insights into the role of magnesium in myo-inositol monophosphatase: metal mechanism, substrate binding, and lithium therapy. J Chem Inf Model 52:2398–2409
Lu S-Y, Jiang Y-J, Zou J-W, Wu T-X (2011) Molecular modeling and molecular dynamics studies of the GSK3β/ATP/substrate complex: understanding the unique P + 4 primed phosphorylation specificity for GSK3β substrates. J Chem Inf Model 51:1025–1036
Darden T, York D, Pedersen L (1993) Particle mesh Ewald: an N log(N) method for Ewald sums in large systems. J Chem Phys 98:10089–10092
Ryckaert JP, Ciccotti G, Berendsen HJC (1977) Numerical integration of the cartesian equations of motion of a system with constraints: molecular dynamics of n-alkanes. J Comput Phys 23:327–341
Wu X, Brooks BR (2003) Self-guided Langevin dynamics simulation method. Chem Phys Lett 381:512–518
Papaleo E, Lindorff-Larsen K, De Gioia L (2012) Paths of long-range communication in the E2 enzymes of family 3: a molecular dynamics investigation. Phys Chem Chem Phys 14:12515–12525
Henkelman G, Jónsson H (2000) Improved tangent estimate in the nudged elastic band method for finding minimum energy paths and saddle points. J Chem Phys 113:9978–9985
Bergonzo C, Campbell AJ, Walker RC, Simmerling C (2009) A partial nudged elastic band implementation for use with large or explicitly solvated systems. Int J Quant Chem 109:3781–3790
Kabsch W, Sander C (1983) Dictionary of protein secondary structure: pattern recognition of hydrogen-bonded and geometrical features. Biopolymers 22:2577–2637
Jalkute CB, Barage SH, Dhanavade MJ, Sonawane KD (2013) Molecular dynamics simulation and molecular docking studies of angiotensin concerting enzyme with inhibitor lisinopril and amyloid beta peptide. Protein J 32:356–364
Ezat AA, EI-Bialy NS, Mostafa HIA, Ibrahim MA (2014) Molecular docking investigation of the binding interactions of macrocyclic inhibitors with HCV NS3 protease and its mutants (R155K, D168A and A156V). Protein J 33:32–47
Lu S-Y, Jiang Y-J, Zou J-W, Wu T-X (2012) Effect of double mutations K214/A-E215/Q of FRATide on GSK3β: insights from molecular dynamics simulation and normal mode analysis. Amino Acids 43:267–277
Rengachari S, Bezerra GA, Riegler-Berket L, Gruber CC, Sturm C, Tashler U, Boeszoermenyi A, Dreveny I, Zimmermann R, Gruber K, Oberer M (2012) The structure of monoacylglycerol lipase from Bacillus sp. H257 reveals unexpected conservation of the cap architecture between baterial and human enzymes. Biochim Biophys Acta 1821:1012–1021
Saladino G, Gervasio FL (2012) New insights in protein kinase conformational dynamics. Curr Top Med Chem 12:1889–1895
Md S, Rauf A, Endou A, Takaba H, Miyamoto A (2013) Effect of Y220C mutation on p53 and its rescue mechanism: a computer chemistry approach. Protein J 32:68–74
Srikumar PS, Rohini K, Rajesh PK (2014) Molecular dynamics simulations and principal component analysis on human laforin mutation W32G and W32G/K87A. Protein J 33:289–295
Lu S, Jiang Y, Lv J, Zou J, Wu T (2011) Mechanism of kinase inactivation and nonbinding of FRATide to GSK3β due to K85M mutation: molecular dynamics simulation and normal mode analysis. Biopolymers 95:669–681
Huang M, Lu S, Shi T, Zhao Y, Chen Y, Li X, Liu X, Huang Z, Zhang J (2013) Conformational transitional pathway in the activation process of allosteric glucokinase. PLoS ONE 8:e55857
Réblová K, Střelcová Z, Kulhánek P, Beššeová I, Mathews DH, Nostrand KV, Yildirim I, Turner DH, Sponer J (2010) An RNA molecular switch: intrinsic flexibility of 23S rRNA helices 40 and 68 5’-UAA/5’-GAN internal loops studied by molecular dynamics methods. J Chem Theory Comput 6:910–929
Acknowledgments
This study was supported by the National Key Basic Research Program of China (973 Program, No. 2011CBA00800), the Open Funding Project of National Key Laboratory of Biochemical Engineering, and the Key Agriculture Support Project of Jiangsu Province (No. BE2013400).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, H., Tian, R., Ni, Z. et al. Conformational Transition Pathway in the Inhibitor Binding Process of Human Monoacylglycerol Lipase. Protein J 33, 503–511 (2014). https://doi.org/10.1007/s10930-014-9572-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-014-9572-z