Abstract
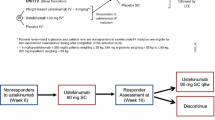
Exposure–response modeling is important to optimize dose and dosing regimen in clinical drug development. The joint modeling of multiple endpoints is made possible in part by recent progress in latent variable indirect response (IDR) modeling for ordered categorical endpoints. This manuscript presents the results of joint modeling of continuous and ordered categorical endpoints in the latent variable IDR modeling framework through the sharing of model parameters, with an application to the exposure–response modeling of sirukumab. Sirukumab is a human anti- interleukin-6 (IL-6) monoclonal antibody that binds soluble human IL-6 thus blocking IL-6 signaling, which plays a major role in the pathophysiology of rheumatoid arthritis (RA). A phase 2 clinical trial was conducted in patients with active RA despite methotrexate therapy, who received subcutaneous (SC) administration of either placebo or sirukumab of 25, 50 or 100 mg every 4 weeks (q4w) or 100 mg every 2 weeks (q2w). Major efficacy endpoints were the 20, 50, and 70% improvement in the American College of Rheumatology (ACR20, ACR50, and ACR70) disease severity criteria, and the 28-joint disease activity score using C-reactive protein (DAS28). The ACR endpoints were treated as ordered categorical and DAS28 as continuous. The results showed that, compared with the common approach of separately modeling the endpoints, the joint model could describe the observed data better with fewer parameters through the sharing of random effects, and thus more precisely characterize the dose–response relationship. The implications on future dose and dosing regimen optimization are discussed in contrast with those from landmark analysis.







Similar content being viewed by others
References
Hu C (2014) Exposure-response modeling of clinical end points using latent variable indirect response models. CPT 3:e117. https://doi.org/10.1038/psp.2014.15
Hu C, Zhou H, Sharma A (2017) Landmark and longitudinal exposure-response analyses in drug development. J Pharmacokinet Pharmacodyn. https://doi.org/10.1007/s10928-017-9534-0
Sharma A, Jusko WJ (1996) Characterization of four basic models of indirect pharmacodynamic responses. J Pharmacokinet Biopharm 24(6):611–635
Felson DT, Anderson JJ, Boers M et al (1995) American College of Rheumatology. Preliminary definition of improvement in rheumatoid arthritis. Arthritis Rheum 38(6):727–735
Hutmacher MM, Krishnaswami S, Kowalski KG (2008) Exposure-response modeling using latent variables for the efficacy of a JAK3 inhibitor administered to rheumatoid arthritis patients. J Pharmacokinet Pharmacodyn 35:139–157
Hu C, Xu Z, Rahman MU, Davis HM, Zhou H (2010) A latent variable approach for modeling categorical endpoints among patients with rheumatoid arthritis treated with golimumab plus methotrexate. J Pharmacokinet Pharmacodyn 37(4):309–321
Hu C, Xu Z, Mendelsohn A, Zhou H (2013) Latent variable indirect response modeling of categorical endpoints representing change from baseline. J Pharmacokinet Pharmacodyn 40(1):81–91
Hu C, Szapary PO, Yeilding N, Zhou H (2011) Informative dropout modeling of longitudinal ordered categorical data and model validation: application to exposure-response modeling of physician’s global assessment score for ustekinumab in patients with psoriasis. J Pharmacokinet Pharmacodyn 38(2):237–260
Hu C, Yeilding N, Davis HM, Zhou H (2011) Bounded outcome score modeling: application to treating psoriasis with ustekinumab. J Pharmacokinet Pharmacodyn 38(4):497–517
Hu C, Zhou H (2016) Improvement in latent variable indirect response joint modeling of a continuous and a categorical clinical endpoint in rheumatoid arthritis. J Pharmacokinet Pharmacodyn 43(1):45–54
Hu C, Randazzo B, Sharma A, Zhou H (2017) Improvement in latent variable indirect response modeling of multiple categorical clinical endpoints: application to modeling of guselkumab treatment effects in psoriatic patients. J Pharmacokinet Pharmacodyn. https://doi.org/10.1007/s10928-017-9531-3
Smolen JS, Weinblatt ME, Sheng S, Zhuang Y, Hsu B (2014) Sirukumab, a human anti-interleukin-6 monoclonal antibody: a randomised, 2-part (proof-of-concept and dose-finding), phase II study in patients with active rheumatoid arthritis despite methotrexate therapy. Ann Rheum Dis 73(9):1616–1625. https://doi.org/10.1136/annrheumdis-2013-205137
Hu C, Zhang J, Zhou H (2011) Confirmatory analysis for phase III population pharmacokinetics. Pharm Stat 10(7):812–822
Hu C, Zhou H (2008) An improved approach for confirmatory phase III population pharmacokinetic analysis. J Clin Pharmacol 48(7):812–822. https://doi.org/10.1177/0091270008318670
Zhang L, Beal SL, Sheiner LB (2003) Simultaneous vs. sequential analysis for population PK/PD data I: best-case performance. J Pharmacokinet Pharmacodyn 30(6):387–404
Hu C, Szapary PO, Mendelsohn AM, Zhou H (2014) Latent variable indirect response joint modeling of a continuous and a categorical clinical endpoint. J Pharmacokinet Pharmacodyn 41(4):335–349. https://doi.org/10.1007/s10928-014-9366-0
Ma L, Zhao L, Xu Y, Yim S, Doddapaneni S, Sahajwalla CG, Wang Y, Ji P (2014) Clinical endpoint sensitivity in rheumatoid arthritis: modeling and simulation. J Pharmacokinet Pharmacodyn 41(5):537–543. https://doi.org/10.1007/s10928-014-9385-x
Woo S, Pawaskar D, Jusko WJ (2009) Methods of utilizing baseline values for indirect response models. J Pharmacokinet Pharmacodyn 36:381–405
Beal SL, Sheiner LB, Boeckmann A, Bauer RJ (2009) NONMEM user’s guides (1989-2009). Icon Development Solutions, Ellicott City
Zhuang Y, Xu Z, de Vries DE, Wang Q, Shishido A, Comisar C, Ford JA, Keen M, Achira M, Tsukamoto Y, Petty KJ, Davis HM, Zhou H (2013) Pharmacokinetics and safety of sirukumab following a single subcutaneous administration to healthy Japanese and Caucasian subjects. Int J Clin Pharmacol Ther 51(3):187–199. https://doi.org/10.5414/CP201785
Savic RM, Karlsson MO (2009) Importance of shrinkage in empirical bayes estimates for diagnostics: problems and solutions. AAPS J 11(3):558–569. https://doi.org/10.1208/s12248-009-9133-0
Li L, Zhang Y, Ma L, Ji P, Yim S, Chowdhury BA, Doddapaneni S, Liu J, Wang Y, Sahajwalla C (2017) Exposure-response modeling and power analysis of components of ACR response criteria in rheumatoid arthritis (Part 2: continuous model). J Clin Pharmacol 57(9):1107–1125. https://doi.org/10.1002/jcph.967
Hutmacher MM, French JL, Krishnaswami S, Menon S (2011) Estimating transformations for repeated measures modeling of continuous bounded outcome data. Stat Med 30(9):935–949. https://doi.org/10.1002/sim.4155
Smithson M, Verkuilen J (2006) A better lemon squeezer? Maximumlikelihood regression with beta-distributed dependent variables. Psychol Methods 11(1):54–71
Li L, Zhang Y, Ma L, Ji P, Yim S, Chowdhury B, Doddapaneni S, Liu J, Wang Y, Sahajwalla C (2017) Exposure-response modeling and power analysis of components of ACR response criteria in rheumatoid arthritis (Part 1: binary model). J Clin Pharmacol 57(9):1097–1106. https://doi.org/10.1002/jcph.891
Overgaard RV, Ingwersen SH, Tornoe CW (2015) Establishing good practices for exposure-response analysis of clinical endpoints in drug development. CPT 4(10):565–575. https://doi.org/10.1002/psp4.12015
Funding
This research was funded by Janssen Research and Development, LLC.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hu, C., Xu, Y., Zhuang, Y. et al. Joint longitudinal model development: application to exposure–response modeling of ACR and DAS scores in rheumatoid arthritis patients treated with sirukumab. J Pharmacokinet Pharmacodyn 45, 679–691 (2018). https://doi.org/10.1007/s10928-018-9598-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10928-018-9598-5