Abstract
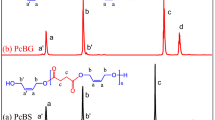
A series of triblock copolyesters were produced by ring-opening polymerization of l-lactide initiated by the dihydroxy-terminated poly(alkylene dodecanedioate)s as macroinitiators. The dihydroxy-terminated poly(alkylene dodecanedioate)s as low melting blocks were prepared by using 1,12-dodecanedioic acid and methyl substituted aliphatic diols. The influence of the different polyesters middle block on microstructure, thermal, crystallization and tensile properties of the triblock copolyesters was investigated. It was found that the poly(alkylene dodecanedioate)s containing methyl substitutions still exhibited crystallization and melting behavior, but the crystallization ability decreased when compared with linear poly(alkylene dodecanedioate). The diffraction peaks of soft and hard segments, measured by WAXD, showed a gentle trend after copolymerization. Through the TGA test, the results showed that thermal stability of the triblock polyesters was improved. Tensile tests exhibited that the elongation at break was improved significantly, with the highest elongation at break reaching 400%, while the tensile strength of triblock copolyesters decreased slightly compared with polylactide. The correlation between the middle block structures and their physical properties was explored and discussed.









Similar content being viewed by others
References
Pretula J, Slomkowski S, Penczek S (2016) Polylactides—methods of synthesis and characterization. Adv Drug Deliv Rev 107:3–16
Nofar M, Sacligil D, Carreau PJ, Kamal MR, Heuzey M-C (2019) Poly(lactic acid) blends: processing, properties and applications. Int J Biol Macromol 125:307–360
Roslaniec Z (2006) Polyester thermoplastic elastomers: synthesis, properties, and some applications. In: Fakirov RZ (ed) Handbook of condensation thermoplastic elastomers. Wiley-VCH, Weinheim
Pitet LM, Hillmyer MA (2009) Combining ring-opening metathesis polymerization and cyclic ester ring-opening polymerization to form ABA triblock copolymers from 1,5-cyclooctadiene and d, l-lactide. Macromolecules 42(11):3674–3680
Konwar DB, Sethy S, Satapathy BK, Jacob J (2017) Effect of poly(l-lactide) chain length on microstructural and thermomechanical properties of poly(l-lactide)-b-poly(butylene carbonate)-b-poly(l-lactide) triblock copolymers. Polymer 123:87–99
Wang W, Lu W, Goodwin A, Wang H, Yin P, Kang N-G et al (2019) Recent advances in thermoplastic elastomers from living polymerizations: macromolecular architectures and supramolecular chemistry. Prog Polym Sci 95:1–31
Liu Q, Jiang L, Shi R, Zhang L (2012) Synthesis, preparation, in vitro degradation, and application of novel degradable bioelastomers—a review. Prog Polym Sci 37(5):715–765
Huang MH, Li S, Vert M (2004) Synthesis and degradation of PLA–PCL–PLA triblock copolymer prepared by successive polymerization of ε-caprolactone and -lactide. Polymer 45(26):8675–8681
Jing Z, Shi X, Zhang G (2017) Synthesis and properties of biodegradable supramolecular polymers based on polylactide-block-poly(δ-valerolactone)-block-polylactide triblock copolymers. Polym Int 66(11):1487–1497
Schneiderman DK, Hillmyer MA (2016) Aliphatic polyester block polymer design. Macromolecules 49(7):2419–2428
Konwar DB, Satapathy BK, Jacob J (2019) Influence of aliphatic polycarbonate middle block on mechanical and microstructural behaviour of triblock copolymers based on poly. Polym Int 68(3):400–409
Zhang Z, Grijpma DW, Feijen J (2004) Triblock copolymers based on 1,3-trimethylene carbonate and lactide as biodegradable thermoplastic elastomers. Macromol Chem Phys 205(7):867–875
Jang J, Park H, Jeong H, Mo E, Kim Y, Yuk JS et al (2019) Thermoset elastomers covalently crosslinked by hard nanodomains of triblock copolymers derived from carvomenthide and lactide: tunable strength and hydrolytic degradability. Polym Chem 10(10):1245–1257
Watts A, Kurokawa N, Hillmyer MA (2017) Strong, resilient, and sustainable aliphatic polyester thermoplastic elastomers. Biomacromolecules 18(6):1845–1854
Wang Z, Yuan L, Tang C (2017) Sustainable elastomers from renewable biomass. Acc Chem Res 50(7):1762–1773
Andronova N, Albertsson A-C (2006) Resilient bioresorbable copolymers based on trimethylene carbonate, l-lactide, and 1,5-dioxepan-2-one. Biomacromolecules 7(5):1489–1495
Zhou Y, Faust R, Richard R, Schwarz M (2005) Syntheses and characterization of poly(cyclohexyl vinyl ether-stat-vinyl alcohol)-b-polyisobutylene-b-poly(cyclohexyl vinyl ether-stat-vinyl alcohol) triblock copolymers and their application as coatings to deliver paclitaxel from coronary stents. Macromolecules 38(20):8183–8191
Hillmyer MA, Tolman WB (2014) Aliphatic polyester block polymers: renewable, degradable, and sustainable. Acc Chem Res 47(8):2390–2396
Kasyapi N, Mehta R, Bhowmick AK (2015) Raman and NMR spectroscopic studies on hydrolytic degradation of d, l-lactide−δ-valerolactone–d, l-lactide copolymer. ACS Sustain Chem Eng 3(7):1381–1393
Widjaja LK, Kong JF, Chattopadhyay S, Lipik VT, Liow SS, Abadie MJM et al (2011) Triblock copolymers of ε-caprolactone, l-lactide, and trimethylene carbonate: biodegradability and elastomeric behavior. J Biomed Mater Res Part A 99A(1):38–46
Lee X, Wahit MU, Adrus N (2016) Biodegradable and temperature-responsive thermoset polyesters with renewable monomers. J Appl Polym Sci 133(40):44007
Jia Z, Wang J, Sun L, Zhu J, Liu X (2018) Fully bio-based polyesters derived from 2,5-furandicarboxylic acid (2,5-FDCA) and dodecanedioic acid (DDCA): from semicrystalline thermoplastic to amorphous elastomer. J Appl Polym Sci 135(14):46076
Zhou C, Wei Z, Yu Y, Shao S, Leng X, Wang Y et al (2019) Biobased long-chain aliphatic polyesters of 1,12-dodecanedioic acid with a variety of diols: odd-even effect and mechanical properties. Mater Today Commun 19:450–458
Save M, Schappacher M, Soum A (2002) Controlled ring-opening polymerization of lactones and lactides initiated by lanthanum isopropoxide, 1. General aspects and kinetics. Macromol Chem Phys 203(5–6):889–899
Zhang Y, Zhu Q, Fei Z, Lin X, Xia B, Wu Q (2019) Stereoselectivity-tailored chemo-enzymatic synthesis of enantiocomplementary poly(ω-substituted-δ-valerolactone) enabled by engineered lipase. Eur Polym J 119:52–60
Marand H, Alizadeh A, Farmer R, Desai R, Velikov V (2000) Influence of structural and topological constraints on the crystallization and melting behavior of polymers. 2. Poly(arylene ether ether ketone). Macromolecules 33(9):3392–3403
Voet VSD, Alberda van Ekenstein GOR, Meereboer NL, Hofman AH, Gt B, Loos K (2014) Double-crystalline PLLA-b-PVDF-b-PLLA triblock copolymers: preparation and crystallization. Polym Chem 5(7):2219–2230
Zhou J, Jiang Z, Wang Z, Zhang J, Li J, Li Y et al (2013) Synthesis and characterization of triblock copolymer PLA-b-PBT-b-PLA and its effect on the crystallization of PLA. RSC Adv 3(40):18464–18473
Kong JF, Lipik V, Abadie MJ, Deen GR, Venkatraman SS (2012) Biodegradable elastomers based on ABA triblocks: influence of end-block crystallinity on elastomeric character. Polym Int 61(1):43–50
Debuissy T, Pollet E, Avérous L (2016) Synthesis of potentially biobased copolyesters based on adipic acid and butanediols: Kinetic study between 1,4- and 2,3-butanediol and their influence on crystallization and thermal properties. Polymer 99:204–213
Pan C, Lu J, Wu B, Wu L, Li B-G (2017) Effect of monomer structure on crystallization and glass transition of flexible copolyesters. J Polym Environ 25:1051–1061
Olsén P, Borke T, Odelius K, Albertsson A-C (2013) ε-Decalactone: a thermoresilient and toughening comonomer to poly(l-lactide). Biomacromolecules 14(8):2883–2890
Persenaire O, Alexandre M, Degée P, Dubois P (2001) Mechanisms and kinetics of thermal degradation of poly(ε-caprolactone). Biomacromolecules 2(1):288–294
Tong J-D, Jerôme R (2000) Dependence of the ultimate tensile strength of thermoplastic elastomers of the triblock type on the molecular weight between chain entanglements of the central block. Macromolecules 33(5):1479–1481
Zhou C, Wei Z, Jin C, Wang Y, Yu Y, Leng X et al (2018) Fully biobased thermoplastic elastomers: synthesis of highly branched linear comb poly(β-myrcene)-graft-poly(l-lactide) copolymers with tunable mechanical properties. Polymer 138:57–64
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 51803019) and the Fundamental Research Funds for the Central Universities (No. DUT19LAB27).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Leng, X., Jin, C., Zhou, C. et al. ABA Triblock Copolyesters Composed of Poly(L-lactide) A Hard Blocks: Long Chain Aliphatic Polyesters as B Soft Midblocks. J Polym Environ 28, 1420–1430 (2020). https://doi.org/10.1007/s10924-020-01694-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-020-01694-y