Abstract
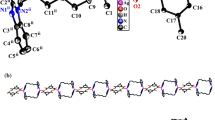
A new dinuclear two-dimensional metal–organic framework (MOF) Ag(I) complex of 4-amino-N-pyridin-2yl-benzenesulfonamide (Ag-sprd) was successfully synthesized and characterized by 1H-NMR, FT-IR, and single-crystal X-ray diffraction (SCXRD) analysis. The Ag(I) complex crystallizes in monoclinic space group P21/n. The crystal structure shows an Ag(I) metal coordinated with the secondary ligand tetrahydropyrrole (THP). The molecular structure of the dinuclear [Ag2(C11H9N3O2S)2.(C4H8N)2]n displays the two-dimensional polymeric chain linked via Ag1 metal atom to amino nitrogen N1(Ag1-N1 = 2.831 Å). The crystal structure of Ag(I) complex reveals an argentophilic interaction with Ag1-Ag1a distance of 2.9625(4) Å, showing distorted pyramidal shape and geometry index τ5 = 0.51 for Ag1. The Ag1a forms distorted tetrahedral coordination behavior and geometry index τ4 = 0.54. The Ag(I) complex exhibited remarkable photo-degradation of 98.49% within 120 min against the methylene blue (MB) in visible light radiation. Furthermore, we explored interconnects and interaction energy in crystal packing, complex’s active and non-active surfaces, the Hirshfeld surfaces (HS), and energy framework analysis. The stability of the molecule was assessed using DFT calculations like MEP, MPA, HOMO–LUMO energy, and global reactivity descriptors parameters. The Kb (binding constant) value was obtained for the binding behavior of the Ag(I) complex towards CT-DNA by UV–Vis. absorption titration, confirming the intercalation mode of binding. The Ag(I) complex shows higher inhibitory activity and lower toxicity tendencies than the sprd ligand against a panel of gram (+ ve) and gram (− ve) organisms, according to data of minimum inhibitory concentration (MIC) and cytotoxicity, respectively. Bioinformatics prospecting of biological activities of a compound was carried out using Molinspirational and ADMET-SAR studies.











Similar content being viewed by others
References
G.M. Brown, The biosynthesis of folic acid. II. Inhibition by sulfonamides. J. Biol. Chem. 237(2), 536–40 (1962)
P. Huovinen et al., Trimethoprim and sulfonamide resistance. Antimicrob. Agents Chemother. 39(2), 279–289 (1995)
I.M. Marzano et al., Crystal structure, antibacterial and cytotoxic activities of a new complex of bismuth(III) with sulfapyridine. Molecules 18(2), 1464–1476 (2013)
M. El-Gaby et al., Sulfonamides: synthesis and the recent applications in medicinal chemistry. Egypt. J. Chem. 63(12), 5289–5327 (2020)
Shukla, P.K., et al., Significance of nitrogen heterocyclic nuclei in the search of pharmacological active compounds. New Perspective in Agricultural Human Health, 2017: p. 100–126.
Ajani, O.O., Synthesis of New N, N-Disubstituted Aryl-and Alkylaryl Sulphonamides and their Antimicrobial Properties. Doctoral dissertation, 2012.
G. Velraj, S. Soundharam, Structure, vibrational, electronic, NBO and NMR analyses of 4-amino-N-[2-pyridinyl] benzene sulfonamide (sulfapyridine) by experimental and theoretical approach. J. Mol. Struct. 1074, 475–486 (2014)
A. Ebrahiminezhad et al., Ancient and novel forms of silver in medicine and biomedicine. J. Adv. Med. Sci. Appl. Technol. 2(1), 122–128 (2016)
B.N. Socha et al., Qualitative and quantitative contributions of intermolecular interactions of dinuclear Ag complexes of sulfathiazole and sulfadiazine: X-ray crystallographic, Hirshfeld surface analysis, DFT studies and biological activities. J. Mol. Struct. 1255, 132426 (2022)
B.N. Socha et al., Role of metal Cu(II) and Ag(I) on molecular structure of 4-amino-N-(2, 6-dimethoxypyrimidin-4-yl) benzenesulfonamide in presence of 3-methyl pyridine: synthesis, spectral, crystallographic and DNA interaction studies. Polyhedron 188, 114696 (2020)
M.D. Alalawy et al., Exploring qualitative and quantitative contributions of intermolecular interactions, DNA-binding and cytotoxic activity of isostructural and isomorphous Cd and Zn complexes. Polyhedron 185, 114595 (2020)
M. Zhang et al., Two pure MOF-photocatalysts readily prepared for the degradation of methylene blue dye under visible light. Dalton Trans. 47(12), 4251–4258 (2018)
R.P. Dubey et al., Cadmium complex of sulfathiazole dihydrate with secondary ligand pyridine: structure, DFT studies, Hirshfeld surface analysis and antimicrobial activity. Indian J. Phys. 95, 33–42 (2021)
SFAC, C., + SHELXS-97-CRYSTAL STRUCTURE SOLUTION-WinGX VERSION. SFAC.
L.J. Farrugia, WinGX suite for small-molecule single-crystal crystallography. J. Appl. Crystallogr. 32(4), 837–838 (1999)
O.V. Dolomanov et al., a complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 42(2), 339–341 (2009)
C.F. Macrae et al., Mercury 4.0: from visualization to analysis, design and prediction. J. Appl. Crystallogr. 53(Pt1), 226–235 (2020)
Frisch, A.J.W., USA, 25p, gaussian 09W Reference. Wallingford, 2009.
Y.O. Bhola et al., Molecular structure, DFT studies, Hirshfeld surface analysis, energy frameworks, and molecular docking studies of novel (E)-1-(4-chlorophenyl)-5-methyl-N′-((3-methyl-5-phenoxy-1-phenyl-1H-pyrazol-4-yl) methylene)-1H-1, 2, 3-triazole-4-carbohydrazide. Mol. Cryst. Liq. Cryst. 692(1), 83–93 (2019)
S.B. Pandya et al., Combined experimental and computational studies on molecular structure of nickel complex of 4-amino-N-(1, 3-thiazol-2-yl) benzenesulfonamide with coordinated pyridine. Inorg. Nano-Metal Chem. 51(9), 1280–1290 (2021)
K. Agarwal et al., Wireless Power transfer strategies for implantable bioelectronics. IEEE Rev. Biomed. Eng. 10, 136–161 (2017)
D.L. Namera et al., Arylidene analogues as selective COX-2 inhibitors: synthesis, characterization, in silico and in vitro studies. J. Biomol. Struct. Dyn. 39(18), 7150–7159 (2021)
A.B. Thakkar et al., Isolation, identification, and characterization of alpha- asarone, from hydromethanolic leaf extract of Acorus calamus L. and its apoptosis-inducing mechanism in A549 cells. J. Biomol. Struct. Dyn. (2023). https://doi.org/10.1080/07391102.2023.2227712
B.N. Socha et al., 1-D MOF [Ag(CHNOS)(CHN)]n: photocatalytic treatment, crystallographic evaluation, ADMET parameters, CT-DNA and anticancer activity. J. Biomol. Struct. Dyn. (2023). https://doi.org/10.1080/07391102.2023.2236732
F. Arjmand, B. Mohani, S. Ahmad, Synthesis, antibacterial, antifungal activity and interaction of CT-DNA with a new benzimidazole derived Cu(II) complex. Eur. J. Med. Chem. 40(11), 1103–1110 (2005)
T.J. Padariya et al., Synthesis, physicochemical, quantitative analysis and crystal structures of two mononuclear diperchlorate cobalt (II) complexes of 1, 10-phenanthroline: antimicrobial, cytotoxicity and DNA/BSA interaction studies. Struct. Chem. 34(4), 1405–1425 (2023)
M.D. Alalawy et al., Role of secondary ligand on Cd complex of sulfamethizole: synthesis, spectroscopic characterization, Hirshfeld surface analysis and DNA interaction. J. Mol. Struct. 1247, 131411 (2022)
M.D. Alalawy et al., Molecular structure investigation, Hirshfeld surface analysis and DNA interaction of cadmium complex of 4-amino-N-(1, 3-thiazol-2-yl) benzenesulfonamide monohydrate in the presence of the secondary ligand β-picoline. Polyhedron 200, 115137 (2021)
P. Thakor et al., Extraction and purification of phytol from : cytotoxic and apoptotic activity. RSC Adv. 6(54), 48336–48345 (2016)
K.P. Chaudhary et al., Divalent zinc complex of succinylsulfathiazole: synthesis, spectral, molecular structure, DNA interaction profile and Hirshfeld surface analysis. Inorg. Chem. Commun. 122, 108318 (2020)
P. Thakor et al., Phytol induces ROS mediated apoptosis by induction of caspase 9 and 3 through activation of TRAIL, FAS and TNF receptors and inhibits tumor progression factor Glucose 6 phosphate dehydrogenase in lung carcinoma cell line (A549). Biomed. Pharmacother. 92, 491–500 (2017)
A.B. Thakkar et al., Induction of apoptosis in lung carcinoma cells (A549) by hydromethanolic extract of Acorus calamus L. Process Biochem. 123, 1–10 (2022)
C.H. Koo, Y.J. Lee, The crystal and molecular structure of sulfapyridine. Arch. Pharm. Res. 2(2), 99–110 (1979)
B.R. Chavda et al., Coordination behavior of dinuclear silver complex of sulfamethoxazole with solvent molecule having static rotational disorder: spectroscopic characterization, crystal structure, Hirshfeld surface and antimicrobial activity. J. Mol. Struct. 1228, 129777 (2021)
K.K. Lalvani et al., Synthesis, crystallographic investigation, DFT studies and Hirshfeld surface analysis of novel bromo hydrazine derivative:(E)-2-(2-bromo-4, 5-dimethoxybenzilidene)-1-(4, 5-dimethoxy-2-methylphenyl)-1-hydroxyhydrazine. Mol. Cryst. Liq. Cryst. 732(1), 87–101 (2022)
B.N. Socha et al., Gas-phase DFT studies, quantum chemical calculation and 3D energy framework analysis of novel copper complex of sulfadimethoxine in the presence of secondary ligand 3-methyl pyridine. Inorg. Nano-Metal Chem. 51(12), 1822–1830 (2021)
K.K. Dodeja et al., Lipophilicity, gas-phase optimized geometry, quantum chemical calculations, Hirshfeld surface analysis, energy frameworks, and molecular docking studies of novel (Z)-2-((3-(4-chlorophenyl)-1-phenyl-1H-pyrazol-4-yl)methylene)-3,4-dihydronaphthalen-1(2H)-one. Inorg. Nano-Metal Chem. (2021). https://doi.org/10.1080/24701556.2021.1983839
M.K. Priya et al., Molecular structure, spectroscopic (FT-IR, FT-Raman, 13C and 1H NMR) analysis, HOMO-LUMO energies, Mulliken, MEP and thermal properties of new chalcone derivative by DFT calculation. Mater. Today Proc. 8, 37–46 (2019)
M. El-Mansy et al., DFT approach for FT-IR spectra and HOMO–LUMO energy gap for N-(p-dimethylaminobenzylidene)-p-nitroaniline (DBN). Spectrochim. Acta Part A: Mol. Biomol. Spectrosc. 111, 217–222 (2013)
S. Sundaram, V.N. Vijayakumar, V. Balasubramanian, Electronic and structure conformational analysis (HOMO-LUMO, MEP, NBO, ELF, LOL, AIM) of hydrogen bond binary liquid crystal mixture: DFT/TD-DFT approach. Comput. Theor. Chem. 1217, 113920 (2022)
R.P. Dubey et al., Quantum chemical calculations, molecular docking, Hirshfeld surface analysis, biological activity, and characterizations (IR and NMR) of cadmium complex of triple sulfa drugs constituent sulfamethazine. Inorg. Nano-Metal Chem. 51(10), 1442–1451 (2020)
I. Salahshoori et al., A deep insight of solubility behavior, mechanical quantum, thermodynamic, and mechanical properties of Pebax-1657 polymer blends with various types of vinyl polymers: a mechanical quantum and molecular dynamics simulation study. J. Mol. Liq. 363, 119793 (2022)
J.B.F. Fankam et al., Study of electronic structure, optoelectronics, linear and nonlinear optical properties and chemical descriptors of dibromodinitrofluorescein isomers in gasphase and solvent media using abinitio and DFT methods. Chin. J. Phys. 66, 461–473 (2020)
L. Feng et al., Molecular dynamics and quantum simulation of different cationic dyes removal from contaminated water using UiO-66 (Zr)-(COOH) 2 metal–organic framework. J. Mol. Liq. 349, 118085 (2022)
P. Geerlings, F. De Proft, W. Langenaeker, Conceptual density functional theory. Chem. Rev. 103(5), 1793–1873 (2003)
J. Haribabu et al., Design and synthesis of heterocyclic azole based bioactive compounds: Molecular structures, quantum simulation, and mechanistic studies through docking as multi-target inhibitors of SARS-CoV-2 and cytotoxicity. J. Mol. Struct. 1250, 131782 (2022)
M.K. Patel et al., Crystal structure elucidation, Hirshfeld surface analysis, lattice energy frame work, and molecular docking studies of copper(II) complex of 1-(3-bromo-5-chloro-2-hydroxyphenyl)ethan-1-one in the presence of solvent pyridine as secondary ligand. Inorg. Nano-Metal Chem. (2022). https://doi.org/10.1080/24701556.2022.2078353
P. Thakor et al., Cytotoxic, apoptosis inducing effects and anti-cancerous drug candidature of jasmonates, in Drug development for cancer and diabetes (Cambridge, Apple Academic Press, 2020), pp.117–127
S.S. Thakkar, F. Shelat, P. Thakor, Magical bullets from an indigenous Indian medicinal plant Tinospora cordifolia: an in silico approach for the antidote of SARS-CoV-2. Egypt J Pet 30(1), 53–66 (2021)
S.S. Thakkar et al., Benzothiazole analogues: synthesis, characterization, MO calculations with PM6 and DFT, studies and antimalarial as DHFR inhibitors and antimicrobial activities. Bioorg. Med. Chem. 25(20), 5396–5406 (2017)
T.J. Padariya et al., Synthesis, physicochemical, quantitative analysis and crystal structures of two mononuclear diperchlorate cobalt(II) complexes of 1,10-phenanthroline: antimicrobial, cytotoxicity and DNA/BSA interaction studies. Struct. Chem. 34(4), 1405–1425 (2023)
R. Mehandi et al., Synthesis, characterization, DFT calculation, antifungal, antioxidant, CT-DNA/pBR322 DNA interaction and molecular docking studies of heterocyclic analogs. J. Mol. Struct. 1245, 131248 (2021)
K.M.P. Kumar et al., Synthesis, structural characterization, CT-DNA interaction study and antithrombotic activity of new ortho-vanillin-based chiral (Se, N, O) donor ligands and their Pd complexes. Inorg. Chim. Acta 528, 120609 (2021)
K.P. Chaudhary et al., Coordination behavior of succinylsulfathiazole–crystal structure of [Cu (SST).(Pyridine) 3. H2O] n, DNA interaction and cytotoxic studies. J. Mol. Struct. 1225, 129262 (2021)
S.B. Pandya et al., DNA interaction, cytotoxicity and molecular structure of cobalt complex of 4-amino-N-(6-chloropyridazin-3-yl) benzene sulfonamide in the presence of secondary ligand pyridine. Appl. Organomet. Chem. 33(12), e5235 (2019)
S.B. Pandya et al., DNA interaction, cytotoxicity and molecular structure of cobalt complex of 4-amino-N-(6-chloropyridazin-3-yl) benzene sulfonamide in the presence of secondary ligand pyridine. Appl. Organomet. Chem. 33(12), e5235 (2019)
Chavda, B.R. and B.N.J.C.-N.I.i.T.A. Socha, Cytotoxicity Study. Cytotoxicity-New Insights into Toxic Assessment, 2021: p. 93.
A. Poojary et al., Minimum inhibitory concentration (MIC) of ceftriaxone for blood culture isolates of nalidixic acid resistant Salmonella (NARS)–A 9 year study. Int. J. Infect. Dis. 101, 55–56 (2020)
P. Gong et al., Enzyme coordination conferring stable monodispersity of diverse metal–organic frameworks for photothermal/starvation therapy. J. Colloid Interface Sci. 642, 612–622 (2023)
P. Gong et al., Fluorescent COFs with a Highly conjugated structure for combined starvation and gas therapy. ACS Appl. Mater. Interfaces 14(41), 46201–46211 (2022)
S.H. Song et al., Donor-acceptor structured photothermal COFs for enhanced starvation therapy. Chem. Eng. J. 442, 135963 (2022)
K. Zhao et al., Size-controllable covalent organic frameworks with high NIR absorption for targeted delivery of glucose oxidase. J. Mol. Liq. 346, 117896 (2022)
A.B. Thakkar et al., Hydromethanolic leaves extract of Dalbergia sissoo Roxb. ex DC. Induces apoptosis in Lung adenocarcinoma cells. Process Biochem. (2023). https://doi.org/10.1016/j.procbio.2023.10.006
G.S. Das et al., Biomass-derived carbon quantum dots for visible-light-induced photocatalysis and label-free detection of Fe(III) and ascorbic acid. Sci. Rep. 9(1), 15084 (2019)
J. Yang et al., High-performance photodetectors and enhanced photocatalysts of two-dimensional TiO 2 nanosheets under UV light excitation. Nanoscale 8(15), 8170–8177 (2016)
Z.J. Chen et al., Waste-derived catalysts for water electrolysis: circular economy-driven sustainable green hydrogen energy. Nano-Micro Lett. 15(1), 4 (2023)
L. Ma et al., A high-energy aqueous Zn‖ NO 2 electrochemical cell: a new strategy for NO 2 fixation and electric power generation. Energy Environ. Sci. 16(3), 1125–1134 (2023)
I. Khan et al., Review on methylene blue: its properties, uses, toxicity and photodegradation. Water 14(2), 242 (2022)
S.B. Pandya et al., Visible light-driven photocatalysts, quantum chemical calculations, ADMET-SAR parameters, and DNA binding studies of nickel complex of sulfadiazine. Sci. Rep. 13(1), 15275 (2023)
Acknowledgements
We are thankful to DST-FIST, New Delhi for funding the single crystal diffractometer facility at the Department of Physics, Sardar Patel University, Vallabh Vidyanagar. Bhavesh N. Socha is thankful to UGC CPEPA-II/2018-19/2442(11) program of P.G. Department of Chemistry, Sardar Patel University, Vallabh Vidyanagar, for the project fellowship. We are thankful for SICART, Vallabh Vidyanagar for NMR, and IR studies. The authors are also thankful to the Department of Chemistry, Sardar Patel University, for DNA binding studies. We are also thankful to the Department of Materials Science for Uv-Vis spectroscopy to analyze the degradation rate.
Author information
Authors and Affiliations
Contributions
BNS: Investigation, Conceptualization, Writing-original draft, Software, Formal analysis, Photocatalyst activity. UHP: Conceptualization, Editing, Supervision, Methodology. SBP: Data curation, Software, Formal analysis. RHP: Supervision., Formal analysis BSB: DNA interactions studies. Parth Thakor: Anticancer activity, Formal analysis ABT: Anticancer activity, Formal analysis
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Socha, B.N., Pandya, S.B., Patel, U.H. et al. Silver-Based 3D-Supramolecular Metal–Organic Framework: Crystallographic Evaluation, Antibiotic, Anticancer, and Visible-Light-Driven Photocatalytic Activity. J Inorg Organomet Polym (2023). https://doi.org/10.1007/s10904-023-02946-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10904-023-02946-9