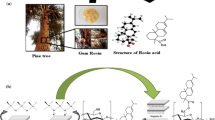
Abstract
In this work, potato starch was hydrolyzed by acid (HCl) to form nanoparticles (SNPs) that were esterified with acid Rosin using a green heterogeneous catalyst based on Algerian montmorillonite clay known as “Magnhite”. It has a similar activity to the classical Brönsted (H2SO4) and Lewis (Al2O3) acids, which implies that it can replace homogeneous acid catalysts. Particular emphasis was devoted to investigate the influence of temperature, reaction time, and amount of catalyst on the degree of substitution (DS) on the reaction conditions to achieve a high DS. Fourier Transform Infrared analysis confirms the esterification of SNPs by the appearance of new bands at 1724 cm−1 assigned to ester group. X-ray diffraction and scanning electron microscopy indicate that the esterification reaction reduces the crystallinity while the morphology changes from SNPs nanoparticles to a completely destroyed form. Besides, the thermal stability of esterified SNPs has been slightly reduced compared to unmodified starch. The volumetric assay was used to evaluate the DS of the products and the effects of the rosin/glucose anhydrous acid unit in the molar ratio. It is found that DS increases with increasing the molar ratio and reaches its maximum value of 0.135 at a molar ratio of 4:1. Furthermore, antimicrobial activity study of SNPs esterified with rosin acid (DS between 0 and 0.141), revealed that the esterified SNPs were effective against all the tested bacterial strains. Moreover, the DS is directly proportional to the zone of inhibition. This research showcases the importance of the newly designed SNPs-Rosin formulation in the biomedical and food industries.











Similar content being viewed by others
References
F. Zhu, Encapsulation and delivery of food ingredients using starch based systems. Food Chem. (2017). https://doi.org/10.1016/j.foodchem.2017.02.101
R.C. Eerlingen, J.A. Delcour, Formation, analysis, structure and properties of type III enzyme resistant starch. J. Cereal Sci. 22, 129–138 (1995)
Y. Zhu, C. Romain, C.K. Williams, Sustainable polymers from renewable resources. Nature (2016). https://doi.org/10.1038/nature21001
A.O. Ashogbon, E. Akintayo, A. Ekiti, Recent trend in the physical and chemical modification of starches from different botanical sources: a review. Strach (2014). https://doi.org/10.1002/star.201300106
L. Yu, K. Dean, L. Li, Polymer blends and composites from renewable resources. Prog. Polym. Sci. 31, 576–602 (2006). https://doi.org/10.1016/j.progpolymsci.2006.03.002
A. Bule, Starch granules: structure and biosynthesis. Int. J. Biol. Macromol. 23, 85–112 (1998)
A.O. Ashogbon, E.T. Akintayo, Recent trend in the physical and chemical modification of starches from different botanical sources: a review. Starch 66, 41–57 (2014). https://doi.org/10.1002/star.201300106
A.N.E.H. Sid, B. Kouini, A. Hazourli, R. Djafar, N. Gherraf, M. Bououdina, The synergistic effect of algerian Na-bentonite/potato starch/grass powder on the enhancement of aged water-based drilling fluids. Arab. J. Sci. Eng. (2022). https://doi.org/10.1007/s13369-021-06519-1
N. Masina et al., A review of the chemical modification techniques of starch. Carbohydr. Polym. (2016). https://doi.org/10.1016/j.carbpol.2016.09.094
D.L. Topping, P.M. Clifton, Short-chain fatty acids and human colonic function: roles of resistant starch and nonstarch polysaccharides. Physiol. Rev. 81(3), 1031–1064 (2018)
R. Hoover, Composition, molecular structure, and physicochemical properties of tuber and root starches: a review. Carbohydr. Polym. (2001). https://doi.org/10.1016/S0144-8617(00)00260-5
S. Pe, The molecular structures of starch components and their contribution to the architecture of starch granules: a comprehensive review. Strach 62, 389–420 (2010). https://doi.org/10.1002/star.201000013
A. Rodrigues, M. Emeje, Recent applications of starch derivatives in nanodrug delivery. Carbohydr. Polym. 87(2), 987–994 (2012). https://doi.org/10.1016/j.carbpol.2011.09.044
B. Bhushan, Introduction to nanotechnology (Springer Handbooks, Berlin, 2017), pp.1–19
Y. Fan, F. Picchioni, Modification of starch: a review on the application of ‘green’ solvents and controlled functionalization. Carbohydr. Polym. 241, 116350 (2020). https://doi.org/10.1016/j.carbpol.2020.116350
Q. Chen et al., Recent progress in chemical modification of starch and its applications. RSC Adv. 5(83), 67459–67474 (2015). https://doi.org/10.1039/c5ra10849g
A.S. Babu, R. Parimalavalli, K. Jagannadham, J.S. Rao, Chemical and structural properties of sweet potato starch treated with organic and inorganic acid. J. Food Sci. Technol. 52(9), 5745–5753 (2015). https://doi.org/10.1007/s13197-014-1650-x
S. Maiti, S.S. Ray, A.K. Kundu, Rosin: a renewable resource for polymers and polymer chemicals. Progress Polym Sci 14(3), 297–338 (1989)
P.M. Division, M.S. Centre, Rosin: a renewable resource for polymers and polymer chemicals. Prog. Polym. Sci. 14, 297–338 (1989)
A. Zaoui, V. Mahendra, G. Mitchell, Z. Cherifi, A. Harrane, Design, synthesis and thermo—chemical properties of rosin vinyl imidazolium based compounds as potential advanced biocompatible materials. Waste Biomass Valoriz. (2019). https://doi.org/10.1007/s12649-019-00691-0
H. Derdar et al., Green nanocomposites from rosin-limonene copolymer and algerian clay. Polymers (Basel) 12, 2020 (1971)
P.A. Wilbon, F. Chu, C. Tang, N.E. NatuKochkina, Y. Khokhlova, Synthesis of silver nanoparticles in DMSO solutions of starch: a comparative investigation of native and soluble starches. Prog. Renew. Polym. (2015). https://doi.org/10.17586/2220-8054-2015-6
Z. Xu, W. Lou, G. Zhao, M. Zhang, J. Hao, X. Wang, Pentaerythritol rosin ester as an environmentally friendly multifunctional additive in vegetable oil-based lubricant. Tribol. Int. (2019). https://doi.org/10.1016/j.triboint.2019.02.038
A.M. Atta, I.F. Nassar, H.M. Bedawy, Unsaturated polyester resins based on rosin maleic anhydride adduct as corrosion protections of steel. React. Func. Polym. 67, 617–626 (2007). https://doi.org/10.1016/j.reactfunctpolym.2007.04.001
S. Karakus, M. Ilgar, I.M. Kahyaoglu, A. Kilislioglu, Influence of ultrasound irradiation on the intrinsic viscosity of guar gum–PEG/rosin glycerol ester nanoparticles. Int. J. Biol. Macromol. (2019). https://doi.org/10.1016/j.ijbiomac.2019.08.254
M. Akeb, M. Belbachir, A. Harrane, Polymerization of β-pinene by using natural montmorillonite clay as a green catalyst. Green Mater. 6(2), 58–64 (2018). https://doi.org/10.1680/jgrma.17.00040
S. Peng, X. Fan, J. Zhang, F. Wang, A highly efficient heterogeneous catalyst of Ru/MMT: preparation, characterization, and evaluation of catalytic effect. Appl. Catal. B. 140–141, 115–124 (2013). https://doi.org/10.1016/j.apcatb.2013.03.029
L. De Chimie, D. De Chimie, F. Sciences, Preparation of Poly(oxybutyleneoxymaleoyl) catalyzed by a proton exchanged montmorillonite clay. Molecules 9, 968–977 (2004)
S. Lycourghiotis, D. Makarouni, E. Kordouli, K. Bourikas, C. Kordulis, V. Dourtoglou, Transformation of limonene into high added value products over acid activated natural montmorillonite. Catal. Today 355, 757–767 (2020). https://doi.org/10.1016/j.cattod.2019.04.036
Y. Zhang, S. Li, Y. Xu, X. Shi, M. Zhang, Y. Huang, Engineering of hollow polymeric nanosphere-supported imidazolium-based ionic liquids with enhanced antimicrobial. Nano Res. 15(6), 5556–5568 (2022)
C. Vanlalveni, S. Lallianrawna, A. Biswas, M. Selvaraj, B. Changmai, S.L. Rokhum, Green synthesis of silver nanoparticles using plant extracts and their antimicrobial activities: a review of recent literature. RSC Adv. 11, 2804–2837 (2021). https://doi.org/10.1039/d0ra09941d
M.Z.I. Nizami, B.D.L. Campéon, A. Satoh, Y. Nishina, Graphene oxide-based multi-component antimicrobial hydrogels. Bull. Chem. Soc. Jpn. 95(5), 713–720 (2022). https://doi.org/10.1246/bcsj.20220017
J.P. Hill, R.G. Shrestha, J. Song, Q. Ji, K. Ariga, L.K. Shrestha, Monitoring the release of silver from a supramolecular fullerene C60-AgNO3 Nanomaterial. Bull. Chem. Soc. Jpn. 94(4), 1347–1354 (2021). https://doi.org/10.1246/bcsj.20210028
A.S. Abreu, M. Oliveira, A. Sá, R.M. Rodrigues, M.A. Cerqueira, A.A. Vicente, A.V. Machado, Antimicrobial nanostructured starch based films for packaging. Carbohydr. Polym. 129, 127–134 (2015). https://doi.org/10.1016/j.carbpol.2015.04.021
M.B. Vásconez, S.K. Flores, C.A. Campos, J. Alvarado, L.N. Gerschenson, Antimicrobial activity and physical properties of chitosan—tapioca starch based edible films and coatings. Food Res. Int. 42(7), 762–769 (2009). https://doi.org/10.1016/j.foodres.2009.02.026
R. Syafiq, S.M. Sapuan, M.Y.M. Zuhri, R.A. Ilyas, A. Nazrin, S.F.K. Sherwani, Antimicrobial activities of starch-based biopolymers and biocomposites incorporated with plant essential oils: a review. Polymers (2020). https://doi.org/10.3390/polym12102403
Y. Zhao, J. Yang, X. Ding, X. Ding, S. Duan, F. Xu, Polycaprolactone/polysaccharide functional composites for low- temperature fused deposition modelling. Bioact. Mater. 5(2), 185–191 (2020). https://doi.org/10.1016/j.bioactmat.2020.02.006
K. Saravanakumar et al., Synthesis, characterization, and cytotoxicity of starch-encapsulated biogenic silver nanoparticle and its improved anti-bacterial activity. Int. J. Biol. Macromol. 182, 1409–1418 (2021). https://doi.org/10.1016/j.ijbiomac.2021.05.036
Z. Draoua, A. Harrane, M. Belbachir, Amphiphilic biodegradable poly(ε-caprolactone)-poly (ethylene glycol)—poly(ε-caprolactone) triblock copolymer synthesis by maghnite-H+ as a green catalyst. J. Macromol. Sci. A 52(2), 130–137 (2015). https://doi.org/10.1080/10601325.2015.980763
D. Le, H. Angellier-Coussy, Preparation and application of starch nanoparticles for nanocomposites: a review. React. Func. Polym. (2014). https://doi.org/10.1016/j.reactfunctpolym.2014.09.020
C.S.W. Adhesive, Preparation and properties of normal temperature cured starch-based wood adhesive. BioResources 11(2), 4839–4849 (2016)
R. Lin, H. Li, H. Long, J. Su, W. Huang, Structure and characteristics of lipase-catalyzed rosin acid starch. Food Hydrocoll 43, 1–8 (2014). https://doi.org/10.1016/j.foodhyd.2014.06.008
K. Wang, M. Nicholaou, Suppression of antimicrobial resistance in MRSA using CRISPR-dCas9. Amer Soc Clinic Lab Sci 30(4), 207–213 (2017)
F. Haddouchi, H.A. Lazouni, P. Naturels, D. De Biologie, F. Sciences, U.A. Belkaid, Etude physicochimique et microbiologique de l’ huile essentielle de Thymus fontanesii Boiss & Reut Résumé. Afrique Sci. 05(2), 246–259 (2009)
F. Han, C. Gao, M. Liu, F. Huang, B. Zhang, Synthesis, optimization and characterization of acetylated corn starch with the high degree of substitution. Int. J. Biol. Macromol. 59, 372–376 (2013). https://doi.org/10.1016/j.ijbiomac.2013.04.080
P. Li, X. He, Y. Zuo, X. Li, Y. Wu, Synthesis and characterization of lactic acid esterified starch by an in-situ solid phase method. Int. J. Biol. Macromol. 156, 1316–1322 (2020). https://doi.org/10.1016/j.ijbiomac.2019.11.171
A. Agi et al., Ultrasound-assisted weak-acid hydrolysis of crystalline starch nanoparticles for chemically enhanced oil recovery. Int. J. Biol. Macromol. (2019). https://doi.org/10.1016/j.ijbiomac.2019.10.099
A.O. Odiongenyi, N.B. Essien, R.A. Ukpe, Corn starch as a substitute for commercial food starch: FT-IR and rheological characterization. J. Sci. Eng. Res. 3(6), 494–501 (2016)
S. Na et al., Effect of Rosin modification on the visual characteristics of round bamboo culm. Polymers 13(20), 3500 (2021)
K. Zhang et al., Synthesis of long-chain fatty acid starch esters in aqueous medium and its characterization. Eur. Polym. J. 119, 136–147 (2019). https://doi.org/10.1016/j.eurpolymj.2019.07.021
Y. Xu et al., Preparation and characterization of organic-soluble acetylated starch nanocrystals. Carbohydr. Polym. 80(4), 1078–1084 (2010). https://doi.org/10.1016/j.carbpol.2010.01.027
S. Susanti, D. Al Karoma, D. Mulyani, M. Masruri, Physical properties and characterization of cassava peel waste modified by esterification. J. Pure Appl. Chem. Res. 6(3), 255–260 (2017). https://doi.org/10.21776/ub.jpacr.2017.006.03.346
J. Xin, Y. Wang, T. Liu, K. Lin, L. Chang, C. Xia, Biosysthesis of corn starch palmitate by lipase novozym 435. Int. J. Mol. Sci. (2012). https://doi.org/10.3390/ijms13067226
R. Lin, H. Li, H. Long, J. Su, W. Huang, Synthesis of rosin acid starch catalyzed by lipase. BioMed Res. Int. (2014). https://doi.org/10.1155/2014/647068
M. Amin, B. Amine, M. Belalia, A. Mostefai, N. Leila, B.M. Bououdina, Green synthesis of starch nanoparticles (SNPs) by esterification with rosin acid catalyzed by maghnite-H + (Algerian montmorillonite) with enhanced antioxidant activity. Arab. J. Sci. Eng. (2022). https://doi.org/10.1007/s13369-022-07033-8
H. Sun, T. Peng, B. Liu, H. Xian, Effects of montmorillonite on phase transition and size of TiO2 nanoparticles in TiO2/montmorillonite nanocomposites. Appl. Clay Sci. 114, 440–446 (2015). https://doi.org/10.1016/j.clay.2015.06.026
U. Holzwarth, N. Gibson, The Scherrer equation versus the ‘Debye-Scherrer equation.’ Nat. Publ. Gr. 6(9), 534 (2011). https://doi.org/10.1038/nnano.2011.145
C. Costa, P.H. Campelo, S.M. De Souza, Rietveld-based quantitative phase analysis of B-type starch crystals subjected to ultrasound and hydrolysis processes. J. Appl. Polym. Sci. (2020). https://doi.org/10.1002/app.49529
K. Dome, E. Podgorbunskikh, A. Bychkov, O. Lomovsky, Changes in the crystallinity degree of starch having different types of crystal structure after mechanical pretreatment. Polymers (2020). https://doi.org/10.3390/polym12030641
T. Desalegn, I.J.V. Garcia, J. Titman, P. Licence, Y. Chebude, Synthesis of starch vernolate in 1-butyl-3-methylimidazolium chloride ionic liquid. Starch (2015). https://doi.org/10.1002/star.201400114
S. Jiang, L. Dai, Y. Qin, L. Xiong, Q. Sun, Preparation and characterization of octenyl succinic anhydride modified taro starch nanoparticles. PLoS ONE (2016). https://doi.org/10.1371/journal.pone.0150043
F. Li et al., Bifunctional reinforcement of green biopolymer packaging nanocomposites with natural cellulose nanocrystals-rosin hybrids. ACS Appl. Bio. Mater. (2020). https://doi.org/10.1021/acsabm.9b01100
S. Ketkaew, P. Kasemsiri, S. Hiziroglu, Effect of oregano essential oil content on properties of green biocomposites based on cassava starch and sugarcane bagasse for bioactive packaging. J. Polym. Environ. (2017). https://doi.org/10.1007/s10924-017-0957-x
M.V. Limaye, V. Gupta, S.B. Singh, G.R. Paik, P. Singh, Antimicrobial activity of composite consisting of cellulose nanofibers and silver nanoparticles. ChemistrySelect 4(41), 12164–12169 (2019). https://doi.org/10.1002/slct.201901572
C. Pan, J. Qian, C. Zhao, H. Yang, X. Zhao, H. Guo, Study on the relationship between crosslinking degree and properties of TPP crosslinked chitosan nanoparticles. Carbohydr. Polym. (2020). https://doi.org/10.1016/j.carbpol.2020.116349
Acknowledgements
The authors are grateful for the substantial financial support provided by the General—Direction of Scientific Research and Technology Development (DGRSDT, MESRS, Algeria).
Author information
Authors and Affiliations
Contributions
A did the lab practice and wrote the manuscript. BEF performed and interpreted the physicochemical analyzes. C did an antibacterial activity in the lab. D corrected English and interpreted the DRX.
Corresponding author
Ethics declarations
Conflict of interest
The authors have not disclosed any competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bezzekhami, M.A., Belalia, M., Hamed, D. et al. Nanoarchitectonics of Starch Nanoparticles Rosin Catalyzed by Algerian Natural Montmorillonite (Maghnite-H+) for Enhanced Antimicrobial Activity. J Inorg Organomet Polym 33, 193–206 (2023). https://doi.org/10.1007/s10904-022-02490-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-022-02490-y