Abstract
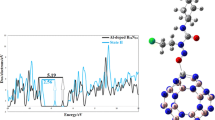
The search for a potential system with desirable electronic properties for sensing and delivering lomustine anticancer drugs led us to the porous B6N6 covalent organic framework (COF). The capability of B6N6 for the recognition and delivery of lomustine investigated using density functional theory calculations at GGA/PBE/DNP computational level. The most stable S1 complex results from the interaction of drug oxygen atoms with the B atom and the B–B bond of the considered COF. The lomustine adsorption causes a remarkable decrease of 83.6% in the band gap (Eg) of the B6N6 moiety in the most stable complexes. The recovery time for this complex at cancer cell temperature are acceptable values of 1.99 × 10–6 and 0.0016 s in the presence and absence of UV irradiation, respectively. The lomustine drug released from the desired carrier in the presence of an external electric field along the z-axis. It is concluded that B6N6 is a potential sensor and drug delivery system for lomustine while treating cancerous tissues.





Similar content being viewed by others
Data Availability
N/A.
Code Availability
N/A.
References
W.H. Mahmoud, F.N. Sayed, G.G. Mohamed, Appl. Organomet. Chem. 30, 959–973 (2016)
F.J.K. Rehmann, L.P. Cuffe, O. Mendoza, D.K. Rai, N. Sweeney, K. Strohfeldt et al., Appl. Organomet. Chem. 19, 293–300 (2005)
H. Sun, P. She, G. Lu, K. Xu, W. Zhang, Z. Liu, J. Mater. Sci. 49, 6845–6854 (2014)
H. Chen, B. Wang, D. Gao, M. Guan, L. Zheng, H. Ouyang, Z. Chai, Y. Zhao, W. Feng, Small 9, 2735–2746 (2013)
G. Ciofani, V. Rafa, J. Yu, Y. Chen, Y. Obata, S. Takeoka, A. Menciassi, A. Cuschieri, Curr. Nanosci. 5, 33–38 (2009)
G. Ciofani, Expert Opin. Drug Deliv. 7, 889–893 (2010)
A.K. Attia, N.F. Abo-Talib, M.H. Tammam, Adv. Pharm. Bull. 7, 151–157 (2017)
Z. Noroozi, R. Rahimi, M. Solimannejad, Comput. Theor. Chem. 1129, 9–15 (2018)
R. Rahimi, M. Solimannejad, Appl. Surf. Sci. 525, 146577 (2020)
R. Rahimi, M. Solimannejad, J. Mol. Liq. 321, 114917 (2021)
R. Rahimi, M. Solimannejad, Z. Ehsanfar, J. Mol. Model. 27, 347 (2021)
S. Yao, Z. Liu, L. Li, Nano-Micro Lett. 13, 1–20 (2021)
S. Bhunia, K.A. Deo, A.K. Gaharwar, Adv. Funct. Mater. 30, 2002046 (2020)
Q. Fang, J. Wang, S. Gu, R.B. Kaspar, Z. Zhuang, J. Zheng, H. Guo, S. Qiu, Y. Yan, J. Am. Chem. Soc. 137, 8352–8355 (2015)
L. Bai, S.Z.F. Phua, W.Q. Lim, A. Jana, Z. Luo, H.P. Tham, L. Zhao, Q. Gao, Y. Zhao, Chem. Commun. 52, 4128–4131 (2016)
N. Liu, W. Huang, X. Zhang, L. Tang, L. Wang, Y. Wang, M. Wu, Appl. Catal. B 221, 119–128 (2018)
Y. Wang, R. Wang, L. Yu, Y. Wang, C. Zhang, X. Zhang, Chem. Eng. J. 401, 126057 (2020)
X. Zhang, K. Yue, R. Rao, J. Chen, Q. Liu, Y. Yang, F. Bi, Y. Wang, J. Xu, N. Liu, Appl. Catal. B 310, 121300 (2022)
X. Zhang, S. Xiang, Q. Du, F. Bi, K. Xie, L. Wang, Mol. Catal. 522, 112226 (2022)
S. Jiang, Z. Zhao, J. Chen, Y. Yang, C. Ding, Y. Yang, Y. Wang, N. Liu, L. Wang, X. Zhang, Surf. Interfaces 30, 101843 (2022)
N. Lin, Y. Gong, R. Wang, Y. Wang, X. Zhang, J. Hazard. Mater. 424, 127637 (2022)
E.C. Anota, A.B. Hernandez, A.E. Morales, M. Castro, J. Mol. Graph. Model. 74, 135–142 (2017)
E.C. Anota, M.S. Villanueva, A.E. Morales, M. Castro, Fuller. Nanotub. Carbon Nanostruct. 26, 93–99 (2018)
J.C. Ordaz, E.C. Anota, M.S. Villanueva, M. Castro, New J. Chem. 41, 8045–8052 (2017)
H. Sajid, M. Asif, K. Ayub, M.A. Gilani, M.S. Akhter, T. Mahmood, Surf. Interfaces 27, 101587 (2021)
Y.Z. Abdullahi, Comput. Theor. Chem. 1197, 113155 (2021)
H. Fisli, N. Bensouilah, N. Dhaoui, M. Abdaoui, J. Incl. Phenom. Macrocycl. Chem. 73, 369–376 (2012)
S. Agarwal, D.K. Jangir, P. Singh, R. Mehrotra, J. Photochem. Photobiol. B 130, 281–286 (2014)
L. Zhuang, J. Gao, Y. Zeng, F. Yu, B. Zhang, M. Li, H. Derendorf, C. Liu, Eur. J. Drug Metab. Pharmacokinet. 36, 61–69 (2011)
L. Dirikolu, T. Chakkath, T. Fan, N.R. Mente, J. Anal. Toxicol. 33, 595–603 (2009)
C.R. Bethune, R.J. Geyer, A.M. Spence, R.J.Y. Ho, Cancer Res. 61, 3669–3674 (2001)
P. Mauricc, O. Glidewel, C. Jacquillat, R. Silver, R. Carey, A. Ten Pas, C. Cornell, R.A. Burningham, N.L. Nissen, J.F. Holland, Cancer 41, 1658–1663 (1978)
R.B. Weiss, B.F. Issell, Cancer Treat. Rev. 9, 313–330 (1982)
B. Delley, J. Chem. Phys. 92, 508–517 (1990)
B. Delley, J. Phys. Chem. 100, 6107–6110 (1996)
B. Delley, J. Chem. Phys. 113, 7756–7764 (2000)
J.R. Frisch, M.J. Trucks, G.W. Schlegel, H.B. Scuseria, G.E. Robb, M.A. Cheeseman, G.A. Scalmani, G. Barone, V. Mennucci, B. Petersson, Gaussian 09, Rev. D.0.1 (Gaussian Inc., Wallingford, 2013)
E. Kim, P.F. Weck, S. Berber, D. Tománek, Phys. Rev. B 78, 113404 (2008)
A. Wang, X. Zhang, M. Zhao, Nanoscale 6, 11157–11162 (2014)
S. Sarikurt, F. Ersan, Marmara Fen Bilimleri Dergisi 30, 383–387 (2018)
T. Suter, V. Brazdova, K. McColl, T.S. Miller, H. Nagashima, E. Salvadori, A. Sella, C.A. Howard, C.W. Kay, F. Cora, J. Phys. Chem. C 122, 25183–25194 (2018)
Y.Z. Abdullahi, T.L. Yoon, A.A. Kassimu, Mater. Chem. Phys. 254, 123470 (2020)
L. Arrue, R. Pino-Rios, Int. J. Quantum Chem. 120, 26403 (2020)
C.A. Celaya, L.F. Hernández-Ayala, F.B. Zamudio, J.A. Vargas, M. Reina, J. Mol. Liq. 329, 115528 (2021)
J. Li, Y. Lu, Q. Ye, M. Cinke, J. Han, M. Meyyappan, Nano Lett. 3, 929–933 (2003)
N.L. Hadipour, A. Ahmadi Peyghan, H. Soleymanabadi, J. Phys. Chem. C 119, 6398–6404 (2015)
P.T. Wong, S.K. Choi, Chem. Rev. 115, 3388–3432 (2015)
P. Swietach, R.D. Vaughan-Jones, A.L. Harris, A. Hulikova, Philos. Trans. R. Soc. B 369, 20130099 (2014)
E.S. Fatemi, M. Solimannejad, Phys. Chem. Res. 9, 623–636 (2021)
P.R. Chandran, N. Sandhyarani, RSC Adv. 4, 44922–44929 (2014)
J. Ge, E. Neofytou, T.J. Cahill III., R.E. Beygui, R.N. Zare, ACS Nano 6, 227–233 (2012)
R. Rahimi, M. Solimannejad, J. Mol. Liq. 354, 118855 (2022)
Funding
There is no funds for performing current research.
Author information
Authors and Affiliations
Contributions
MH: Software, Investigation, Writing original draft. MS: Supervision, Conceptualization, Validation, Reviewing and Editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Heidari, M., Solimannejad, M. The Porous B6N6 Boron Nitride Covalent Organic Framework as a Potential Platform for Sensing and Delivering Lomustine Anticancer Drug: A First-Principles Study. J Inorg Organomet Polym 32, 4216–4222 (2022). https://doi.org/10.1007/s10904-022-02407-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-022-02407-9