Abstract
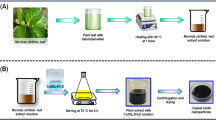
Nowadays, environmentally friendly, economical and efficient production of nanoparticles is increasingly important. The most appropriate nanoparticle synthesis that can be used in this process is green synthesis performed with biological materials (bacteria, fungi or plant). Metallic nanoparticles can be used in many applications and several studies have been conducted to use these particles as antimicrobial and anticancer agents. However, there are limited studies on the green synthesis and biocompatibility of nanocrystalline copper oxide. In this study extracts of Carica papaya leaves were used for the production of copper oxide (CuO) nanoparticles (NPs). UV-Vis spectral analysis showed copper nanoparticles exhibit an absorption peak at around 250 nm. The CuO NPs were analysed with Fourier transform infrared spectroscopy (FT-IR), Scanning electron microscope (SEM), X-ray diffraction (XRD) spectrum and Dynamic light scattering (DLS). The SEM and XRD analysis exposed CuO NPs are spherical in shape with an average particle size of 77 nm. Further, as-formed CuO NPs exhibit significant antioxidant and anticancer activity.










Similar content being viewed by others

References
D.E. Fouad, C. Zhang, H. El-Didamony, L. Yingnan, T.D. Mekuria, A.H. Shah, Results Phys 12, 1253–1261 (2019)
D. Kalpana, J.H. Han, W.S. Park, S.M. Lee, R. Wahab, Y.S. Lee, Arab. J. Chem. 12(7), 1722–1732 (2019)
H. Fu, X. Yang, X. Jiang, A. Yu, Langmuir. 29(23), 7134–7142 (2013)
S.J. Kwon, A.J. Bard, J. Am, Chem. Soc. 134(26), 10777–10779 (2012)
N. Roy, A. Gaur, A. Jain, S. Bhattacharya, V. Rani, Environ. Toxicol. Pharmacol. 36(3), 807–812 (2013)
D.A. Tomalia, S.N. Khanna, Chem. Rev. 116(4), 2705–2774 (2016)
J.A. Darr, J. Zhang, N.M. Makwana, X. Weng, Chem. Rev. 117(17), 11125–11238 (2017)
V. Castagnola, J. Cookman, J.M. De Araújo, E. Polo, Q. Cai, C.P. Silveira, Y.Y. Krpetić, L. Boselli, K.A. Dawson, Nanoscale Horizons 2(4), 187–198 (2017)
W.K. Shin, J. Cho, A.G. Kannan, Y.S. Lee, D.W. Kim, Sci. Rep. 6, 26332 (2016)
I. Khan, K. Saeed, I. Khan, Arab. J. Chem. 12(7), 908–931 (2019)
T. Imaoka, K. Yamamoto, Bull. Chem. Soc. Jpn. 92(5), 941–948 (2019)
A. Loureiro, N.G. Azoia, A.C. Gomes, A. Cavaco-Paulo, Curr. Pharm. Des. 22(10), 1371–1390 (2016)
E. Martis, R. Badve, M. Degwekar, Chronicles Young Sci. 3(1), 68–73 (2012)
A.P. Nikalje, Med. Chem. (Los. Angeles) 5(2), 081–089 (2015)
A. Tadjarodi, R. Roshani, Curr. Chem. Lett. 3(4), 215–220 (2014)
M.H. Habibi, R. Kamrani, R. Mokhtari, Microchim. Acta 171(1-2), 91–95 (2010)
N. Ramgir, N. Datta, M. Kaur, S. Kailasaganapathi, A.K. Debnath, D.K. Aswal, S.K. Gupta, Colloids Surfaces A Physicochem. Eng. Asp. 439, 101–116 (2013)
A.M. Md Jani, D. Losic, N.H. Voelcker, Prog. Mater. Sci. 58(5), 636–704 (2013)
A.E. Shalan, M.M. Rashad, Y. Yu, M. Lira-Cantú, M.S.A. Abdel-Mottaleb, Electrochim. Acta 89, 469–478 (2013)
C. De Montferrand, L. Hu, I. Milosevic, V. Russier, D. Bonnin, L. Motte, A. Brioude, Y. Lalatonne, Acta Biomater. 9(4), 6150–6157 (2013)
S.J. Ahmadi, M. Outokesh, M. Hosseinpour, T. Mousavand, Particuology. 9(5), 480–485 (2011)
V.K.H.R.M. Rajgurunagar, P.K.H.R. Mahavid, V.D. Kulkarni, P.S. Kulkarni, Int. J. Chem. Stud. 1(3), 1–4 (2013)
S. Gunalan, R. Sivaraj, R. Venckatesh, Spectrochim. Acta - Part A Mol. Biomol. Spectrosc. 97, 1140–1144 (2012)
V. Parashar, R. Parashar, B. Sharma, A.C. Pandey, Dig. J. Nanomater. Biostructures. 4(1), 45–50 (2009)
J.L. Gardea-Torresdey, J.G. Parsons, E. Gomez, J. Peralta-Videa, H.E. Troiani, P. Santiago, M.J. Yacaman, Nano Lett. 2(4), 397–401 (2002)
P. Rajiv, S. Rajeshwari, R. Venckatesh, Spectrochim. Acta - Part A Mol. Biomol. Spectrosc. 112, 384–387 (2013)
S. Pandey, A. Mewada, M. Thakur, R. Shah, G. Oza, M. Sharon, Mater. Sci. Eng. C 33(7), 3716–3722 (2013)
P. Rauwel, S. Küünal, S. Ferdov, E. Rauwel, Adv. Mater. Sci. Eng. 2015, 1–9 (2015) (Article ID 682749)
G. Aravind, D. Bhowmik, S. Duraivel, G. Harish, J. Med, Plants Stud. 1(1), 7–15 (2013)
S.L.C.A. Dharmarathna, S. Wickramasinghe, R.N. Waduge, R.P.V.J. Rajapakse, S.A.M. Kularatne, Asian Pac. J. Trop. Biomed. 3(9), 720–724 (2013)
N. Ahmad, H. Fazal, M. Ayaz, B.H. Abbasi, I. Mohammad, L. Fazal, Asian Pac. J. Trop. Biomed. 5(4), 330–333 (2011)
Z.R. Addai, A. Abdullah, S.A. Mutalib, K.H. Musa, E.M.A. Douqan, Adv. J. Food Sci. Technol. 5(7), 859–865 (2013)
A.M. Maisarah, B. Nurul Amira, R. Asmah, O. Fauziah, Int. Food Res. J. 20(3), 1043–1048 (2013)
A. Özkan, H. Gübbük, E. Güneş, A. Erdoǧan, Turkish J. Biol. 35(5), 619–625 (2011)
R. Re, N. Pellegrini, A. Proteggente, A. Pannala, M. Yang, C. Rice-Evans, Free Radic. Biol. Med. 26(9-10), 1231–1237 (1999)
L. Barros, M.J. Ferreira, B. Queirós, I.C.F.R. Ferreira, P. Baptista, Food Chem. 103(2), 413–419 (2007)
C. Perez, M. Pauli, P. Bazerque, Acta Biol Med Exp. 15, 113–115 (1990)
F.R. Cockerill, M.A. Wikler, J. Alder, M.N. Dudley, G.M. Eliopoulos, M.J. Ferraro, D.J. Hardy, D.W. Hecht, J.A. Hindler, J.B. Patel, M. Powell, J.M. Swenson, R.B. Thomson, M.M. Traczewski, J.D. Turnbrighe, M.P. Weinstein, B.L. Zimmer, Methods for dilution antimicrobial susceptibility tests for Bacteria that grow aerobically. Ninth Edition. 18, 32 (2012)
D.A. Scudiero, R.H. Shoemaker, K.D. Paull, A. Monks, S. Tierney, T.H. Nofziger, M.J. Currens, D. Seniff, M.R. Boyd, Cancer Res. 48(17), 4827–4833 (1988)
S. Sukumar, A. Rudrasenan, D. Padmanabhan Nambiar, ACS Omega. 5(2), 1040–1051 (2020)
H. Alishah, S. Pourseyedi, S.Y. Ebrahimipour, S.E. Mahani, N. Rafiei, Rend. Lincei. 28(1), 65–71 (2017)
R. Sankar, P. Manikandan, V. Malarvizhi, T. Fathima, K.S. Shivashangari, V. Ravikumar, Spectrochim. Acta - Part A Mol. Biomol. Spectrosc. 121, 746–750 (2014)
N. Kumar, L.S.B. Upadhyay, Appl. Surf. Sci. 385, 225–233 (2016)
H. Wang, X. Qiao, J. Chen, S. Ding, Colloids Surfaces A Physicochem. Eng. Asp. 299(1-3), 22–28 (2005)
S. Singh, N. Kumar, M. Kumar, A.A. Jyoti, B. Mizaikoff, Chem. Eng. J. 313, 283–292 (2017)
K.S. Tan, K.Y. Cheong, J. Nanopart. Res. 15(4), 1537 (2013)
A. Azam, A.S. Ahmed, M. Oves, M.S. Khan, A. Memic, Int. J. Nanomedicine 7, 6003 (2012)
M. Nasrollahzadeh, S. Mohammad Sajadi, A. Rostami-Vartooni, M. Khalaj, J. Mol, Catal. A Chem. 396, 31–39 (2015)
P. Yugandhar, N. Savithramma, Appl. Nanosci. 6(2), 223–233 (2016)
G. Ren, D. Hu, E.W.C. Cheng, M.A. Vargas-Reus, P. Reip, R.P. Allaker, Int. J. Antimicrob. Agents 33(6), 587–590 (2009)
M.V.D.Z. Park, A.M. Neigh, J.P. Vermeulen, L.J.J. de la Fonteyne, H.W. Verharen, J.J. Briedé, H. van Loveren, W.H. de Jong, Biomaterials. 32(36), 9810–9817 (2011)
R. Sivaraj, P.K.S.M. Rahman, P. Rajiv, S. Narendhran, R. Venckatesh, Spectrochim. Acta - Part A Mol. Biomol. Spectrosc. 129, 255–258 (2014)
P.C. Nagajyothi, P. Muthuraman, T.V.M. Sreekanth, D.H. Kim, J. Shim, Arab. J. Chem. 10(2), 215–225 (2017)
V.D. Mendhulkar, Y. Anu, Asian J. Pharm. Clin. Res. 10, 82–88 (2017)
Acknowledgements
The authors acknowledge Shoolini University, Solan, for providing infrastructure support to conduct the research work. Solan, India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dulta, K., Ağçeli, G.K., Chauhan, P. et al. Biogenic Production and Characterization of CuO Nanoparticles by Carica papaya Leaves and Its Biocompatibility Applications. J Inorg Organomet Polym 31, 1846–1857 (2021). https://doi.org/10.1007/s10904-020-01837-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-020-01837-7