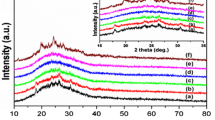
Abstract
Nanocomposites of polymer/V2O5 based on aniline and p-benzoquinine monomers have been synthesized by in situ polymerization method. The PANI/V2O5 and poly(ANI-pBQ)/V2O5 with different amounts of V2O5 (0.5 g and 1.5 g) were characterized by XRD, FT-IR, UV–Vis, SEM and TGA. This results confirmed the distribution of V2O5 in polymers matrix and confirms that poly(ANI-pBQ)/V2O5 form demonstrates a good electrical conductivity and higher crystalline nature than that of PANI/V2O5 samples. FT-IR spectrum has confirmed successful synthesis of polymer/v2O5, optical and electrical properties were discussed. Also, the results of the electrochemical test showed a clear and good electroactivity for samples.







Similar content being viewed by others
References
J.D. Sudha, A. Pich, V.L. Reena, S. Sivakala, H.J.P. Adler, Water-dispersible multifunctional polyaniline-laponite-keggin iron nanocomposites through a template approach. J. Mater. Chem. 21, 16642–16650 (2011)
E. Ozkazanc, H. Ozkazanc, Multifunctional polyaniline/chloroplatinic acid composite material: characterization and potential applications. Polm. Eng. Sci. 59(1), 66–73 (2019)
V.L. Reena, C. Pavithran, V. Verma, J.D. Sudha, Nanostructured multifunctional electromagnetic materials from the guest-host inorganic-organic hybrid ternary system of a polyaniline-clay-polyhydroxy iron composite: preparation and properties. J. Phys. Chem. B. 114(8), 2578–2585 (2010)
N. Asim, S. Radiman, M.A.B. Yarmo, Preparation and characterization of core-shell polyaniline/V2O5 nanocomposite via microemulsion method. Mater. Lett. 62, 1044–1047 (2008)
V.S. de Souza, H.O. da Frota, E.A. Sanches, Polyaniline-CuO hybrid nanocomposite with enhanced electrical conductivity. J. Mol. Struct. 1153, 20–27 (2018)
P. Prabhu, S.M. Iqbal, A. Balaji, B. Karthikeyan, Experimental investigation of mechanical and machining parameters of hybrid nanoclay glass fiber-reinforced polyester composites. Adv. Compos. Hybrid Mater. 2, 93 (2019)
M. Liras, M. Barawi, V.A.P. O’Shea, Hybrid materials based on conjugated polymers and inorganic semiconductors as photocatalysts: from environmental to energy applications. Chem. Soc. Rev. 48, 5454–5487 (2019)
M. Hasan, M.O. Ansari, M.H. Cho, M. Lee, Ammonia sensing and DC electrical conductivity studies of p-toluene sulfonic acid doped cetyltrimethylammonium bromide assisted V2O5@polyaniline composite nanofibers. J. Ind. Eng. Chem. 22, 147–152 (2015)
Z. Luo, Z. Wu, X. Xu, M. Du, T. Wang, Y. Jiang, Impact of substrate temperature on the microstructure, electrical and optical properties of sputtered nanoparticle V2O5 thin films. Vacuum 85, 145–150 (2010)
S. Beke, A review of the growth of V2O5 films from 1885 to 2010. Thin Solid Films 519, 1761–1771 (2011)
B.H. Kim, A. Kim, S.Y. Oh, S.S. Bae, Y.J. Yun, H.Y. Yu, Energy gap modulation in V2O5 nanowires by gas adsorption. Appl. Phys. Lett. 93, 233101–233103 (2008)
E.A. Arbab, G.T. Mola, V2O5 thin film deposition for application in organic solar cells. Appl. Phys. A 122, 405–412 (2016)
A. Mane, M. Suryawanshi, J. Kim, A. Moholka, Fast response of sprayed vanadium pentoxide (V2O5) nanorods towards nitrogen dioxide (NO2) gas detection. Appl. Surf. Sci. 403, 540–550 (2017)
B. Fisher, J. Genossar, L. Patlagan, K. Chashka, G. Reisner, Electric-field-induced semiconductor–semiconductor transition in V2O5. Appl. Phys. A 120, 435–442 (2015)
N. Abd-Alghafour, N.M. Ahmed, Z. Hassan, M.A. Almessiere, M. Bououdina, N.H. Al-Hardan, High sensitivity extended gate effect transistor based on V2O5 nanorods. J. Mater. Sci.: Mater. Electron. 28, 1364–1369 (2017)
A. Talled, C.G. Granqvist, Electrochromic vanadium-pentoxide-based films: structural, electrochemical, and optical properties. J. Appl. Phys. 77, 4655–4666 (1995)
T. Zhai, H. Liu, H. Li, X. Fang, M. Liao, L. Li, H. Zhou, Y. Koide, Y. Bando, D. Golberg, Centimeter-Long V2O5 nanowires: from synthesis to field-emission, electrochemical, electrical transport, and photoconductive properties. Adv. Mater. 22, 2547–2552 (2010)
K.I. Park, H.M. Song, Y. Kim, S.I. Mho, W.I. Cho, I.H. Yeo, Electrochemical preparation and characterization of V2O5/polyaniline composite film cathodes for Li battery. Electrochim. Acta 55, 8023–8029 (2010)
I. Boyano, M. Bengoechea, I. de Meatza, O. Miguel, I. Cantero, E. Ochoteco, H. Grande, M.L. Cantu, P.G. Romero, Influence of acids in the Ppy/V2O5 hybrid synthesis and performance as a cathode material. J. Power Sources 174, 1206–1211 (2007)
D. Li, J. Huang, R.B. Kaner, Polyaniline nanofibers: a unique polymer nanostructure for versatile applications. Acc. Chem. Res. 20, 135–145 (2009)
S. Bhadra, D. Khastgir, N.K. Singha, J.H. Lee, Progress in preparation, processing and applications of polyaniline. Progress Polym. Sci. 34, 783–810 (2009)
G. Wang, R. Vivek, J.Y. Wang, Polyaniline nanoparticles: synthesis, dispersion and biomedical applications. Mini-Rev. Organ. Chem. 14, 56–64 (2017)
S. Liu, L. Liu, F. Meng, Y. Li, F. Wang, Protective performance of polyaniline-sulfosalicylic acid/epoxy coating for 5083 aluminum. Materials 13, 292 (2018)
M. Silakhori, M.S. Naghavi, H.S.C. Metselaar, T.M.I. Mahlia, H. Fauzi, M. Mehrali, Accelerated thermal cycling test of microencapsulated paraffin wax/polyaniline made by simple preparation method for solar thermal energy storage. Materials 6, 1608–1620 (2013)
L. Zu, X. Cui, Y. Jiang, Z. Hu, H. Lian, Y. Liu, Y. Jin, Y. Li, X. Wang, Preparation and electrochemical characterization of mesoporous polyaniline-silica nanocomposites as an electrode material for pseudocapacitors. Materials 8, 1369–1383 (2015)
G.A. Planes, J.L. Rodríguez, M.C. Miras, G. García, E. Pastor, C.A. Barbero, Spectroscopic evidence for intermediate species formed during aniline polymerization and polyaniline degradation. Phys. Chem. Chem. Phys. 12, 10584–10593 (2010)
M. Bláha, F. Marek, Z. Morávková, J. Svoboda, J. Brus, J. Dybal, J. Prokeš, M. Varga, J. Stejskal, Role of p-benzoquinone in the synthesis of a conducting polymer, polyaniline. ACS Omega 4, 7128–7139 (2019)
S. Daikh, F.Z. Zeggai, A. Bellil, A. Benyoucef, Chemical polymerization, characterization and electrochemical studies of PANI/ZnO doped with hydrochloric acid and/or zinc chloride: differences between the synthesized nanocomposites. J. Phys. Chem. Solids 121, 78–84 (2018)
A. Belalia, A. Zehhaf, A. Benyoucef, Preparation of hybrid material based of PANI with SiO2 and its adsorption of phenol from aqueous solution. Polym. Sci. Ser. B 60, 816–824 (2018)
Z. Guo, N. Liao, M. Zhang, A. Feng, Enhanced gas sensing performance of polyaniline incorporated with graphene: a first-principles study. Phys. Lett. A 383, 2751–2754 (2019)
A. Bekhoukh, A. Zehhaf, A. Benyoucef, S. Bousalem, M. Belbachir, Nanoparticules mass effect of ZnO on the properties of poly(4-chloroaniline)/zinc oxide nanocomposites. J. Inorg. Organometall. Polym. Mater. 27, 13–20 (2017)
F.Z. Kouidri, R. Berenguer, A. Benyoucef, E. Morallon, Tailoring the properties of polyanilines/SiC nanocomposites by engineering monomer and chain substituents. J. Mol. Struct. 1188, 121–128 (2019)
S. Kundua, B. Satpati, T. Kar, S.K. Pradhan, Microstructure characterization of hydrothermally synthesized PANI/V2O5·nH2O heterojunction photocatalyst for visible light induced photodegradation of organic pollutants and non-absorbing colorless molecules. J. Hazard. Mater. 339, 161–173 (2017)
C.H.B. Silva, D.C. Ferreira, R.A. Ando, M.L.A. Temperini, Aniline-1,4-benzoquinone as a model system for the characterization of products from aniline oligomerization in low acidic media. Chem. Phys. Lett. 551, 130–133 (2012)
J. Stejskal, M. Trchová, Z. Morávková, P. Bober, M. Bláha, J. Pfleger, P. Magdziarz, J. Prokeš, M. Havlicek, N.S. Sariciftci, A. Sperlich, V. Dyakonov, Z. Zujovic, Conducting materials prepared by the oxidation of p-phenylenediamine with p-benzoquinone. J. Solid State Electrochem. 19, 2653–2664 (2015)
C. Han, Y. Ye, G. Wang, W. Hong, C. Feng, Selective electro-oxidation of phenol to benzoquinone/hydroquinone on polyaniline enhances capacitance and cycling stability of polyaniline electrodes. Chem. Eng. J. 347, 648–659 (2018)
Y. Zhang, J. Liu, Y. Zhang, J. Liu, Y. Duan, Facile synthesis of hierarchical nanocomposites of aligned polyaniline nanorods on reduced graphene oxide nanosheets for microwave absorbing materials. RSC Adv. 7, 54031–54038 (2017)
J. Yan, T. Wei, B. Shao, Z. Fan, W. Qian, M. Zhang, F. Wei, Preparation of a graphene nanosheet/polyaniline composite with high specific capacitance. Carbon 48, 487–493 (2010)
H. Borchert, E.V. Shevchenko, A. Robert, I. Mekis, A. Kornowski, G. Grübel, H. Weller, Determination of nanocrystal sizes: a comparison of TEM, SAXS and XRD studies of highly monodisperse CoPt3 particles. Langmuir 21, 1931–1936 (2005)
P. Scherrer, Nachrichten von der Gesellschaft der Wissenschaften zu Göttingen. Mathematisch-Physikalische Klasse 2, 98 (1918)
R. Paulraj, P. Shankar, G.K. Mani, L. Nallathambi, J.B.B. Rayappan, Fabrication of PANI–ZnO nanocomposite thin film for room temperature methanol sensor. J. Mater. Sci.: Mater. Electron. 28, 10799–10805 (2017)
M. Shi, Y. Zhang, M. Bai, B. Li, Facile fabrication of polyaniline with coral-like nanostructure as electrode material for supercapacitors. Synth. Met. 233, 74–78 (2017)
A.T. Chidembo, K.I. Ozoemena, B.O. Agboola, V. Gupta, G.G. Wildgoosed, R.G. Compton, Nickel(II) tetra-aminophthalocyanine modified MWCNTs as potential nanocomposite materials for the development of supercapacitors. Energy Environ. Sci. 3, 228–236 (2010)
Acknowledgements
The authors thank the D.G.R.S.D.T of Algeria. They also thank the Prof. Emilia Morallon of Alicante University (Spain) for cooperation support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ouis, D., Zeggai, F.Z., Belmokhtar, A. et al. Role of p-Benzoquinone on Chemically Synthesized Nanocomposites by Polyaniline with V2O5 Nanoparticle. J Inorg Organomet Polym 30, 3502–3510 (2020). https://doi.org/10.1007/s10904-020-01508-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-020-01508-7