Abstract
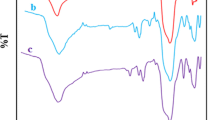
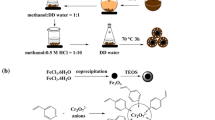
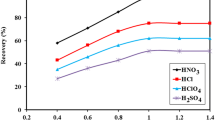
To improve the adsorption capacity of Cd(II) ions, Cd(II) ions were imprinted on the surface of aminoethyl chitosan (AECS), which was coated on Fe3O4@SiO2 nanoparticles. A novel magnetic Cd(II) ion-imprinted polymer (Cd(II)-IIP) was synthesized, characterized, and applied to the selective separation of Cd(II) ions from aqueous solution. The adsorption–desorption properties and selectivity of Cd(II)-IIP and a non-imprinted polymer (Cd(II)-NIP) were investigated. The optimum pH and equilibrium binding time were established at pH 6.0 and 60 min, respectively. Kinetics studies demonstrated that the adsorption process proceeded according to a pseudo-first or second order model, while the adsorption isotherms agreed with the Langmuir model. The maximum adsorption capacities of Cd(II)-IIP and Cd(II)-NIP toward Cd(II) ions, as calculated by the Langmuir equation, at pH 6.0 and 25 °C were 26.1 and 6.7 mg/g, respectively. The imprinted polymer showed higher selectivity toward Cd(II) ions compared to the non-imprinted polymer. The relative selectivity factor (βr) values of Cd(II)/Cu(II), Cd(II)/Cr(II), and Cd(II)/Pb(II) were 3.315, 3.875, and 2.061, respectively. In addition, Cd(II) ions adsorbed on the Cd(II)-IIP adsorbent could be easily released using 0.1 M HNO3, thus showing good material stability and reusability. The adsorption capacity of Cd(II)-IIP was retained at 74% after undergoing six adsorption–desorption cycles.












Similar content being viewed by others
References
F. Fu, Q. Wang, Removal of heavy metal ions from wastewaters: a review. J. Environ. Manag. 92(3), 407–418 (2011)
S.A. Abo-El-Enein, M.A. Eissa, A.A. Diafullah, M.A. Rizk, F.M. Mohamed, Removal of some heavy metals ions from wastewater by copolymer of iron and aluminum impregnated with active silica derived from rice husk ash. J. Hazard. Mater. 172(2–3), 574–579 (2009)
W.S. Wan Ngah, M.A. Hanafiah, Removal of heavy metal ions from wastewater by chemically modified plant wastes as adsorbents: a review. Bioresour. Technol. 99(10), 3935–3948 (2008)
S. Squadrone, M. Prearo, P. Brizio, S. Gavinelli, M. Pellegrino, T. Scanzio, S. Guarise, A. Benedetto, M.C. Abete, Heavy metals distribution in muscle, liver, kidney and gill of European catfish (Silurus glanis) from Italian Rivers. Chemosphere 90(2), 358–365 (2013)
A. Bernard, Renal and Neurological Effects Heavy Metals in the Environment. Encycl Environ Health (2011). https://doi.org/10.1016/B978-0-444-52272-6.00615-2
M. Vigeh, K. Yokoyama, A.A. Shinohara, M. Afshinrokh, Increase in blood heavy metals in early pregnancy induce hypertension in pregnant women. Toxicol. Lett. 205, 197–197 (2011)
M. Kumar, B.P. Tripathi, V.K. Shahi, Crosslinked chitosan/polyvinyl alcohol blend beads for removal and recovery of Cd(II) from wastewater. J. Hazard. Mater. 172(2–3), 1041–1048 (2009)
Y. Xi, Y. Luo, J. Luo, X. Luo, Removal of cadmium(II) from wastewater using novel cadmium ion-imprinted polymers. J. Chem. Eng. Data 60(11), 3253–3261 (2015)
L.P. Lingamdinne, Y.Y. Chang, J.K. Yang, J. Singh, E.H. Choi, M. Shiratani, J.R. Koduru, P. Attri, Biogenic reductive preparation of magnetic inverse spinel iron oxide nanoparticles for the adsorption removal of heavy metals. Chem. Eng. J. 307, 74–84 (2017)
J.K. Bediako, W. Wei, S. Kim, Y.S. Yun, Removal of heavy metals from aqueous phases using chemically modified waste Lyocell fiber. J. Hazard. Mater. 299, 550–561 (2015)
M.I. Shariful, S.B. Sharif, J.J.L. Lee, U. Habiba, B.C. Ang, M.A. Amalina, Adsorption of divalent heavy metal ion by mesoporous-high surface area chitosan/poly (ethylene oxide) nanofibrous membrane. Carbohydr. Polym. 157, 57–64 (2017)
C. Ren, X. Ding, W. Li, H. Wu, H. Yang, Highly efficient adsorption of heavy metals onto novel magnetic porous composites modified with amino groups. J. Chem. Eng. Data 62, 1865–1875 (2017)
D. Mohan, C.U. Pittman Jr., Activated carbons and low cost adsorbents for remediation of tri- and hexavalent chromium from water. J. Hazard. Mater. 137(2), 762–811 (2006)
G. Crini, Recent developments in polysaccharide-based materials used as adsorbents in wastewater treatment. Prog. Polym. Sci. 30(1), 38–70 (2005)
A. Kucinska, A. Cyganiuk, J.P. Lukaszewicz, A microporous and high surface area active carbon obtained by the heat-treatment of chitosan. Carbon 50(8), 3098–3101 (2012)
X. Sun, Q. Li, L. Yang, H. Liu, Removal of chromium(VI) from wastewater using weakly and strongly basic magnetic adsorbents: adsorption/desorption property and mechanism comparative studies. RSC Adv. 6, 18471–18482 (2016)
K. El-Tahlawy, R. Venditti, J. Pawlak, Effect of alkyl ketene dimer reacted starch on the properties of starch microcellular foam using a solvent exchange technique. Carbohydr. Polym. 73, 133–142 (2008)
A. Petrella, V. Petruzzelli, E. Ranieri, V. Catalucci, D. Petruzzelli, Sorption of Pb(II), Cd(II) and Ni(II) from single- and multimetal solutions by recycled waste porous glass. Chem. Eng. Commun. 203(7), 940–947 (2016)
P. Rodríguezestupiñán, A. Erto, L. Giraldo, J.C. Morenopiraján, Adsorption of Cd (II) on modified granular activated carbons: isotherm and column study. Molecules 22(12), 2280 (2017)
A. Petrella, D. Spasiano, P. Acquafredda, N. De Vietro, E. Ranieri, P. Cosma, V. Rizzi, V. Petruzzelli, D. Petruzzelli, Heavy metals retention (Pb(II), Cd (II), Ni (II)) from single and multimetal solutions by natural biosorbents from the olive oil milling operations. Process. Saf. Environ. 114, 79–90 (2018)
M. Hua, S. Zhang, B. Pan, W. Zhang, L. Lv, Q. Zhang, Heavy metal removal from water/wastewater by nanosized metal oxides: a review. J. Hazard. Mater. 211–212, 317–331 (2012)
M.A. El-Sayed, Some interesting properties of metals confined in time and nanometer space of different shapes. Acc. Chem. Res. 32, 257–264 (2001)
Y. Cao, L. Wen, F. Svec, T. Tan, Y. Lv, Magnetic AuNP@Fe3O4, nanoparticles as reusable carriers for reversible enzyme immobilization. Chem. Eng. J. 286, 272–281 (2016)
A.S. de Dios, M.E. Diaz-Garcia, Multifunctional nanoparticles: analytical prospects. Anal. Chim. Acta 666(1–2), 1–22 (2010)
K. Zargoosh, H. Abedini, A. Abdolmaleki, M.R. Moldavians, Effective removal of heavy metal ions from industrial wastes using thiosalicylhydrazide-modified magnetic nanoparticles. Ind. Eng. Chem. Res. 52, 14944–14954 (2013)
Y.C. Lin, H.P. Wang, F. Gohar, M.H. Ullah, X. Zhang, D.F. Xie, H. Fang, J. Huang, J.X. Yang, Preparation and copper ions adsorption properties of thiosemicarbazide chitosan from squid pens. Int. J. Biol. Macromol. 95, 476–483 (2017)
Y. Ren, X. Wei, M. Zhang, Adsorption character for removal Cu(II) by magnetic Cu(II) ion imprinted composite adsorbent. J. Hazard. Mater. 158(1), 14–22 (2008)
S. Cen, W. Li, S. Xu, Z. Wang, Y. Tang, H. Wang, C. Wei, Application of magnetic Cd2+ ion-imprinted mesoporous organosilica nanocomposites for mineral wastewater treatment. RSC Adv. 7, 7996–8003 (2017)
J. Huang, H. Xie, S. Hu, T. Xie, J. Gong, C. Jiang, Q. Ge, Y. Wu, S. Liu, Y. Cui, J. Mao, L. Mei, Preparation, characterization, and biochemical activities of N-(2-carboxyethyl)chitosan from squid pens. J. Agric. Food Chem. 63, 2464–2471 (2015)
J.Y. Je, S.K. Kim, Antimicrobial action of novel chitin derivative. Biochim. Biophys. Acta 1760, 104–109 (2006)
X.Q. Liu, J.M. Xing, Y.P. Guan, G.B. Shan, H.Z. Liu, Synthesis of amino-silane modified superparamagnetic silica supports and their use for protein immobilization. Colloids Surf. A 238, 127–131 (2004)
W. Stöber, A. Fink, E. Bohn, Controlled growth of monodisperse silica spheres in the micron size range. J. Colloid Interface Sci. 26, 62–69 (1968)
A. Hirai, H. Odani, A. Nakajima, Determination of degree of deacetylation of chitosan by 1H NMR spectroscopy. Polym. Bull. 26, 87–94 (1991)
A.A. Ehab, A.T. Dina, Y.N. Mostafa, A tunable template-assisted hydrothermal synthesis of hydroxysodalite zeolite nanoparticles using various aliphatic organic acids for the removal of zinc(II) ions from aqueous media. J. Inorg. Organomet. Polym Mater. 29(1), 229–249 (2019)
B. Liu, X. Lv, X. Meng, G. Yu, D. Wang, Removal of Pb(II) from aqueous solution using dithiocarbamate modified chitosan beads with Pb(II) as imprinted ions. Chem. Eng. J. 220, 412–419 (2013)
E.J. Dunn, X. Zhang, D. Sun, M.F.A. Goosen, Synthesis of N-(aminoalkyl) chitosan for microcapsules. J. Appl. Polym. Sci. 50, 353–365 (2010)
Z. Xu, Y. Feng, X. Liu, M. Guan, C. Zhao, H. Zhang, Synthesis and characterization of Fe3O4@SiO2@poly-l-alanine, peptide brush-magnetic microspheres through NCA chemistry for drug delivery and enrichment of BSA. Colloids Surf. B 81, 503–507 (2010)
L. Wang, Y. Sun, J. Wang, A. Yu, H. Zhang, D. Dong, Preparation of surface plasmon resonance biosensor based on magnetic core/shell Fe3O4/SiO2 and Fe3O4/Ag/SiO2 nanoparticles. Colloids Surf. B 84, 484–490 (2011)
D. Shao, K. Xu, X. Song, J. Hu, W. Yang, C. Wang, Effective adsorption and separation of lysozyme with PAA-modified Fe3O4@silica core/shell microspheres. J. Colloid Interface Sci. 336, 526–532 (2009)
I.F. Nata, G.W. Salim, C.K. Lee, Facile preparation of magnetic carbonaceous nanoparticles for Pb2+ ions removal. J. Hazard. Mater. 183(1–3), 853–858 (2010)
Y. Zhang, X. Liu, J. Nie, L. Yu, Y. Zhong, C. Huang, Improve the catalytic activity of α-Fe2O3, particles in decomposition of ammonium perchlorate by coating amorphous carbon on their surface. J. Solid State Chem. 184, 387–390 (2011)
A. Dolgormaa, C.J. Lv, Y. Li, J. Yang, J.X. Yang, P. Chen, H.P. Wang, J. Huang, Adsorption of Cu(II) and Zn(II) Ions from Aqueous Solution by Gel/PVA-Modified Super-Paramagnetic Iron Oxide Nanoparticles. Molecules 23(11), 2982 (2018)
Buhani, Narsito, Nuryono, E.S. Kunarti, Production of metal ion imprinted polymer from mercapto-silica through so-gel process as selective adsorbent of cadmium. Desalination 251, 83–89 (2010)
A. Aklil, M. Mouflih, S. Sebti, Removal of heavy metal ions from water by using calcined phosphate as a new adsorbent. J. Hazard. Mater. 112, 183–190 (2004)
Buhani, Suharso, Sumadi, Adsorption kinetics and isotherm of Cd(II) ion on Nannochloropsis sp biomass imprinted ionic polymer. Desalination 259, 140–146 (2010)
Y.S. Ho, Second-order kinetic model for the sorption of cadmium onto tree fern: a comparison of linear and non-linear methods. Water Res. 40, 119–125 (2006)
Y.S. Ho, G. Mckay, A comparison of chemisorption kinetic models applied to pollutant removal on various sorbents. Process Saf. Environ. 76, 332–340 (1998)
Z. Popovii, G. Pavlovii, M. Vinkovii, D. Vikiivii, M.R. Linarii, Coordination modes of 3-hydroxypicolinic acid (OH-picH): synthesis and characterization of cadmium(II) complexes. Crystal and molecular structures of [CdX(OH-pic)(OH-picH)(H2O)]2 X = Cl-, Br-. Polyhedron 25(12), 2353–2362 (2006)
C.W. Belock, A. Cetin, N.V. Barone, C.J. Ziegler, Transition Metal Coordination Chemistry of N, N-Bis(2-{pyrid-2-ylethyl})hydroxylamine. Inorg. Chem. 47(16), 7114–7120 (2008)
H. Soltani, A. Belmokhar, F.Z. Zeggal, A. Benyoucef, S. Bousalem, K. Bachari, Copper(II) removal from aqueous solutions by PANI-clay hybrid material: fabrication, characterization, adsorption and kinetics study. J. Inorg. Organomet. Polym. Mater. (2019). https://doi.org/10.1007/s10904-018-01058-z
B. Zhang, Y. Wu, Y. Fan, Synthesis of Novel Magnetic NiFe2O4 Nanocomposite Grafted Chitosan and the Adsorption Mechanism of Cr(VI). J. Inorg. Organomet. Polym Mater. 29, 290–301 (2019)
M.H. Karaoglu, S. Zor, M. Ugurlu, Biosorption of Cr(III) from solutions using vineyard pruning waste. Chem. Eng. J. 159, 98–106 (2010)
Y. Liu, X. Hu, M. Meng, Z. Liu, L. Ni, X. Meng, J. Qiu, RAFT-mediated microemulsion polymerization to synthesize a novel high-performance graphene oxide-based cadmium imprinted polymer. Chem. Eng. J. 302, 609–618 (2016)
M. Imamoglu, Adsorption of Cd(II) Ions onto activated carbon prepared from hazelnut husks. J. Disper. Sci. Technol. 34(9), 1183–1187 (2013)
T.M. Alslaibi, I. Abustan, M.A. Ahmad, A.A. Foul, Kinetics and equilibrium adsorption of iron (II), lead (II), and copper (II) onto activated carbon prepared from olive stone waste. Desalin. Water Treat. 52(40–42), 7887–7897 (2014)
J. Zhang, Preparation, characterization and application of thiosemicarbazide grafted multiwalled carbon nanotubes for solid-phase extraction of Cd(II), Cu(II) and Pb(II) in environmental samples. J. Environ. Sci. 25(11), 2331–2337 (2013)
J. Goel, K. Kadirvelu, A.C. Rajagopal, V.K. Garg, Cadmium(II) Uptake from aqueous solution by adsorption onto carbon aerogel using a response surface methodological approach. Ind. Eng. Chem. Res. 45(19), 6531–6537 (2006)
K. Yang, J. Fox, DPF soot as an adsorbent for Cu(II), Cd(II), and Cr(VI) compared with commercial activated carbon. Environ. Sci. Pollut. Res. 25(9), 8620–8635 (2018)
D. Kołodyńska, J. Krukowska, P. Thomas, Comparison of sorption and desorption studies of heavy metal ions from biochar and commercial active carbon. Chem. Eng. J. 307, 353–363 (2017)
P. Brown, I.A. Jefcoat, D. Parrish, S. Gill, E. Graham, Evaluation of the adsorptive capacity of peanut hull pellets for heavy metals in solution. Adv. Environ. Res. 4(1), 19–29 (2000)
M. Ciopec, C. Davidescu, A. Negrea, L. Lupa, P. Negrea, A. Popa, C. Muntean, Use of D2EHPA-impregnated XAD7 resin for the removal of Cd(II) and Zn(II) from aqueous solutions. Environ. Eng. Manag. J. 10(10), 1597–1608 (2011)
A.S.A. Aziz, L.A. Manaf, H.C. Man, N.S. Kumar, Equilibrium studies and dynamic behavior of cadmium adsorption by palm oil boiler mill fly ash (POFA) as a natural low-cost adsorbent. Desalin. Water Treat. 54(7), 1956–1968 (2015)
S.A. Ong, M. Hirata, T. Hano, Comparative study on kinetic adsorption of Cu(II), Cd(II) and Ni(II) ions from aqueous solutions using activated sludge and dried sludge. Appl. Water Sci. 3(1), 321–325 (2013)
Acknowledgements
We are grateful to the National Natural Science Foundation of China (Grant No. 41771509), Zhejiang Science and Technology Project (Grant Nos. 2018C37052, 2016C37078, 2008C14067) and SRF for ROCS, SEM.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, H., Lin, Y., Li, Y. et al. A Novel Magnetic Cd(II) Ion-Imprinted Polymer as a Selective Sorbent for the Removal of Cadmium Ions from Aqueous Solution. J Inorg Organomet Polym 29, 1874–1885 (2019). https://doi.org/10.1007/s10904-019-01148-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-019-01148-6