Abstract
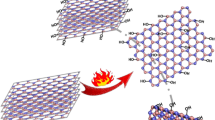
Using density functional theory calculations (based on the B3LYP/6-31G(d) method), we investigated the functionalization of a BN nanosheet (B33N33H22) by different α-amidino carboxylic acids (ACA). It was found that the pristine BN sheet can be noncovalently functionalized by the ACA with an adsorption energy of about − 2.7 kcal/mol. Also, the results showed that structural Stone–Wales (SW) defects significantly increase the adsorption energy. The SW-BN nanosheet transforms from an insulator to a semiconductor by covalent functionalization. Incorporating –NH2 and –NO2 groups in the structure of the ACA molecule increases and decreases the adsorption energy, respectively. The –NH2 group compared to the –NO2 was found to be more favorable for covalent functionalization especially when it is substituted at the meta position of phenyl group of ACA molecule. Investigating the effect of some other electron donating groups showed that the favorability order of applied groups for functionalization is as follows: –N(CH3)2 > –OH > –NH2 > –OCH3 > –Phenyl > –F. Furthermore, it was found that the solubility of BN nanosheets significantly increases, depending on the type of functional group.






Similar content being viewed by others
References
M.J. Allen, V.C. Tung, R.B. Kaner, Honeycomb carbon: a review of graphene. Chem. Rev. 110, 132–145 (2009)
Y. Kubota, K. Watanabe, O. Tsuda, T. Taniguchi, Deep ultraviolet light-emitting hexagonal boron nitride synthesized at atmospheric pressure. Science 317, 932–934 (2007)
A. Ahmadi Peyghan, H. Soleymanabadi, Z. Bagheri, Hydrogen release from NH3 in the presence of BN graphene: DFT studies. J. Mex. Chem. Soc. 59, 67–73 (2015)
L. Wang, J. Huo, H. Yu, T. Chen, L. Deng, A Novel approach for preparation of nano-gold particles/carbon nanotube composites from gold film, poly(ferrocenylsilane) and acetylene. J. Inorg. Organomet. Polym Mater. 17, 121–125 (2007)
G. Selvaduray, L. Sheet, Aluminium nitride: review of synthesis methods. Mater. Sci. Technol. 9, 463–473 (1993)
M. Samadizadeh, A.A. Peyghan, S.F. Rastegar, Sensing behavior of BN nanosheet toward nitrous oxide: a DFT study. Chin. Chem. Lett. 26, 1042–1045 (2015)
J. Nie, Y. Jia, P. Qu, Q. Shi, Carbon nanotube/carbon fiber multiscale composite: influence of interfacial strength on mechanical properties. J. Inorg. Organomet. Polym. Mater. 21, 937–940 (2011)
D. Golberg, Y. Bando, Y. Huang, T. Terao, M. Mitome, C. Tang, C. Zhi, Boron nitride nanotubes and nanosheets. ACS Nano 4, 2979–2993 (2010)
X. Chen, P. Wu, M. Rousseas, D. Okawa, Z. Gartner, A. Zettl, C.R. Bertozzi, Boron nitride nanotubes are noncytotoxic and can be functionalized for interaction with proteins and cells. J. Am. Chem. Soc. 131, 890–891 (2009)
K.K. Kim, A. Hsu, X. Jia, S.M. Kim, Y. Shi, M. Hofmann, D. Nezich, J.F. Rodriguez-Nieva, M. Dresselhaus, T. Palacios, Synthesis of monolayer hexagonal boron nitride on Cu foil using chemical vapor deposition. Nano Lett. 12, 161–166 (2011)
G. Ciofani, G.G. Genchi, I. Liakos, A. Athanassiou, D. Dinucci, F. Chiellini, V. Mattoli, A simple approach to covalent functionalization of boron nitride nanotubes. J. Colloid Interface Sci. 374, 308–314 (2012)
H. Zeng, C. Zhi, Z. Zhang, X. Wei, X. Wang, W. Guo, Y. Bando, D. Golberg, White graphenes”: boron nitride nanoribbons via boron nitride nanotube unwrapping. Nano Lett. 10, 5049–5055 (2010)
C.R. Dean, A.F. Young, I. Meric, C. Lee, L. Wang, S. Sorgenfrei, K. Watanabe, T. Taniguchi, P. Kim, K. Shepard, Boron nitride substrates for high-quality graphene electronics. Nat. Nanotechnol. 5, 722–726 (2010)
J. Sławińska, I. Zasada, Z. Klusek, Energy gap tuning in graphene on hexagonal boron nitride bilayer system. Phys. Rev. B 81, 155433–155439 (2010)
J. Jalilian, H. Zahrabi, J. Jalilian, F. Soofivand, S. Farshadfar, S. Naderizadeh, N. Rahimi, Electronic and magnetic properties of Fe, Co and Ni atomic chains encapsulated in BN nanotube bundle. Comput. Theor. Chem. 979, 10–16 (2012)
C. Zhi, Y. Bando, C. Tang, S. Honda, K. Sato, H. Kuwahara, D. Golberg, Covalent functionalization: towards soluble multiwalled boron nitride nanotubes. Angew. Chem. Int. Ed. 44, 7932–7935 (2005)
A. Bahrami, M.B. Qarai, N.L. Hadipour, The electronic and structural responses of B12N12 nanocage toward the adsorption of some nonpolar X2 molecules: X=(Li, Be, B, N, O, F. Cl, Br, I): a DFT approach, Comput Theor Chem. 1108, 63–69 (2017)
C. Zhi, Y. Bando, C. Tang, Q. Huang, D. Golberg, Boron nitride nanotubes: functionalization and composites. J. Mater. Chem. 18, 3900–3908 (2008)
J. Beheshtian, A.A. Peyghan, M.B. Tabar, Z. Bagheri, DFT study on the functionalization of a BN nanotube with sulfamide. Appl. Surf. Sci. 266, 182–187 (2013)
C.H. Lee, S. Bhandari, B. Tiwari, N. Yapici, D. Zhang, Y.K. Yap, Boron nitride nanotubes: recent advances in their synthesis, functionalization, and applications. Molecules 21, 922 (2016)
S. Danti, G. Ciofani, S. Moscato, D. D’Alessandro, E. Ciabatti, C. Nesti, R. Brescia, G. Bertoni, A. Pietrabissa, M. Lisanti, Boron nitride nanotubes and primary human osteoblasts: in vitro compatibility and biological interactions under low frequency ultrasound stimulation. Nanotechnology 24, 465102 (2013)
S. Pal, S. Vivekchand, A. Govindaraj, C. Rao, Functionalization and solubilization of BN nanotubes by interaction with Lewis bases. J. Mater. Chem. 17, 450–452 (2007)
E.R. Pérez, R.H. Santos, M.T. Gambardella, L.G. De Macedo, U.P. Rodrigues-Filho, J.-C. Launay, D.W. Franco, Activation of carbon dioxide by bicyclic amidines. J. Org. Chem. 69, 8005–8011 (2004)
M. Schmidt et al., General atomic and molecular electronic structure system. J. Comput. Chem. 14, 1347–1363 (1993)
D. Hossain, C.U. Pittman, S.R. Gwaltney, Structures and stabilities of the metal doped gold nano-clusters: M@Au10 (M = W, Mo, Ru, Co). J. Inorg. Organomet. Polym. Mater. 24, 241–249 (2014)
F. Mashhadban, A.S. Ghasemi, F. Ravari, The Effects of Zn doping on the interaction of a single walled carbon nanotube with penicillamine drug: a DFT study. J. Inorg. Organomet. Polym. Mater. 28, 954–961 (2018)
G.W. Waldhart, A.J. Webster, B.M. Schreiber, R.B. Siedschlag, J.S. D’Acchioli, The curious case of the allyl ligand: a study in applying the 18-electron rule. J. Inorg. Organomet. Polym. Mater. 24, 87–94 (2014)
K.M. Eid, H.Y. Ammr, Adsorption of SO2 on Li atoms deposited on MgO (1 0 0) surface: DFT calculations. Appl. Surf. Sci. 257, 6049–6058 (2011)
I.K. Petrushenko, K.B. Petrushenko, Physical adsorption of N-containing heterocycles on graphene-like boron nitride-carbon heterostructures: a DFT study. Comput. Theor. Chem. 1117, 162–168 (2017)
S. Tomić, B. Montanari, N.M. Harrison, The group III–V’s semi-conductor energy gaps predicted using the B3LYP hybrid functional. Physica E 40, 2125–2127 (2008)
T. Koopmans, Ordering of wave functions and eigenenergies to the individual electrons of an atom. Physica 1, 104–113 (1933)
L. Song, L. Ci, H. Lu, P.B. Sorokin, C. Jin, J. Ni, A.G. Kvashnin, D.G. Kvashnin, J. Lou, B.I. Yakobson, Large scale growth and characterization of atomic hexagonal boron nitride layers. Nano Lett. 10, 3209–3215 (2010)
W. Chen, Y. Li, G. Yu, Z. Zhou, Z. Chen, Electronic structure and reactivity of boron nitride nanoribbons with stone-wales defects. J. Chem. Theory Comput. 5, 3088–3095 (2009)
Y. Li, Z. Zhou, D. Golberg, Y. Bando, P.V.R. Schleyer, Z. Chen, Stone—wales defects in single-walled boron nitride nanotubes: formation energies, electronic structures, and reactivity. J. Phys. Chem. C 112, 1365–1370 (2008)
J. Aihara, Reduced HOMO – LUMO gap as an index of kinetic stability for polycyclic aromatic hydrocarbons. J. Phys. Chem. A 103, 7487–7495 (1999)
C. Hansch, A. Leo, R. Taft, A survey of Hammett substituent constants and resonance and field parameters. Chem. Rev. 91, 165–195 (1991)
B. Mennucci, Polarizable continuum model. Wiley Interdiscip. Rev.: Comput. Mol. Sci. 2, 386–404 (2012)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zaghari, Z., Azizian, J. Functionalization of B33N33H22 Nanosheets with α-Amidino Carboxylic Acids: A DFT Study. J Inorg Organomet Polym 29, 1076–1083 (2019). https://doi.org/10.1007/s10904-018-0996-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-018-0996-3