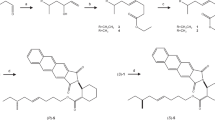
Abstract
In a previous study, we reported the identification and synthesis of a male-specific sex pheromone component of the stink bug, Pellaea stictica, as the alcohol 2,4,8,13-tetramethyltetradecan-1-ol (1). To establish the correlation between the stereochemistry of the pheromone and its bioactivity, it first was necessary to determine its absolute configuration. For this purpose, a series of syntheses were designed to: (a) furnish a mixture of all possible stereoisomers; (b) a narrowed down group of diastereomers, and (c) one specific enantiomer. A crucial step in the syntheses involved a coupling reaction between two key intermediates: a phosphonium salt and an aldehyde, through a Wittig olefination. Nuclear magnetic resonance data of a mixture of the synthetic pheromone diastereomers and further comparison of GC retention times with that of the natural product by gas chromatography suggested that the methyl branches at C2 and C4 were in a syn relationship, reducing the possibilities to only four of the eight possible stereoisomers. Employing GC analysis, chiral derivatization reagents and synthetic (8R)-2,4-syn-1 it was possible to confirm the configuration of the methyl branch at C8 as R, reducing the number of possible stereoisomers to two. After enantioselective synthesis of (2R,4R,8R)-1, the absolute configurations of all methyl branches of the natural compound were confirmed as R, fully identifying the male-produced sex pheromone of P. stictica as (2R,4R,8R)-2,4,8,13-tetramethyltetradecan-1-ol.












Similar content being viewed by others
References
Aldrich JR, Oliver JE, Lusby WR, Kochansky JP, Lockwood JA (1987) Pheromone strains of the cosmopolitan pest, Nezara viridula (Heteroptera: Pentatomidae). J Exp Zool 244:171–175. https://doi.org/10.1002/jez.1402440121
Anelli PL, Biffi C, Montanari F, Quici S (1987) Fast and selective oxidation of primary alcohols to aldehydes or to carboxylic acids and of secondary alcohols to ketones mediated by oxoammonium salts under two-phase conditions. J Org Chem 52:2559–2562. https://doi.org/10.1021/jo00388a038
Appel R (1975) Tertiary phosphane/tetrachloromethane: a versatile reagent for chlorination, dehydration and P-N linkage. Angew Chem 14:801–811. https://doi.org/10.1002/anie.197508011
Bång J, Hedenströn E, Sjödin K (2011) Purification, stereoisomeric analysis and quantification of sex pheromone precursors in female whole body extracts from pine sawfly species. J Chem Ecol 37:125–133. https://doi.org/10.1007/s10886-010-9886-z
Baughman TW, Sworen JC, Wagener KB (2004) The facile preparation of alkenyl metathesis synthons. Tetrahedron 60:10943–10948. https://doi.org/10.1016/j.tet.2004.09.021
Bellota F, D’Auria MV, Sepe V, Zampella A (2009) Synthetic studies on homophymine A: Stereoselective synthesis of (2R,3R,4R,6R)-3-hydroxy-2,4,6-trimethyloctanoic acid. Tetrahedron 65:3659–3663. https://doi.org/10.1016/j.tet.2009.02.069
Bernady KF, Floyd MB, Poletto JF, Weiss MJ (1979) Prostaglandins and congeners. 20. Synthesis of prostaglandins via conjugate addition of lithium trans-1-alkenyltrialkylalanate reagents. A novel reagent for conjugate 1,4-additions. J Org Chem 44:1438–1447. https://doi.org/10.1021/jo01323a017
Borges M, Zarbin PHG, Ferreira JTB, Da Costa MLM (1999) Pheromone sharing: blends based on the same compounds for Euschistus heros and Piezodorus guildinii. J Chem Ecol 25:629–639. https://doi.org/10.1023/A:1020914222769
Borges M, Millar JG, Laumann RA, Moraes MCB (2007) A male-produced sex pheromone from the neotropical redbanded stink bug, Piezodorus guildinii (W.). J Chem Ecol 33:1235–1248. https://doi.org/10.1007/s10886-007-9294-1
Byrne PA, Gilheany DG (2013) The modern interpretation of the Wittig reaction mechanism. Chem Soc Rev 42:6670–6696. https://doi.org/10.1039/C3CS60105F
Chaudhary SK, Hernandez O (1979) 4-Dimethylaminopyridine: an efficient and selective catalyst for the silylation of alcohols. Tetrahedron Lett 20:99–102. https://doi.org/10.1016/S0040-4039(01)85893-7
Corey EJ, Venkateswarlu A (1978) Protection of hydroxyl groups as tert-butyldimethylsilyl derivatives. J Am Chem Soc 94:6190–6191. https://doi.org/10.1021/ja00772a043
Corey EJ, Niwa H, Knolle J (1978) Total synthesis of (S)-12-hydroxy-5,8,14-cis-10-trans-eicosatetraenoic acid (Samuelsson’s HETE). J Am Chem Soc 100:1942–1943. https://doi.org/10.1021/ja00474a058
Crouse GD, Terando NH (1989) Synthesis of 14 C-radiolabelled tilmicosin. J Label Compd Radiopharm 27:465–471. https://doi.org/10.1002/jlcr.2580270414
Dale JA, Dull DL, Mosher HS (1969) α-Methoxy-a-trifluoromethylphenylacetic acid, a versatile Reagent for the determination of enantiomeric composition of alcohols and amines. J Org Chem 34:2543–2549. https://doi.org/10.1021/jo01261a013
Dewick M (2009) Medicinal Natural Products – A Biosynthetic Approach, Third Edition. John Wiley & Sons
Fávaro CF, Rodrigues MACD, Aldrich JR, Zarbin PHG (2011) Identification of semiochemicals in adults and nymphs of the stink bug Pallantia macunaima Grazia (Hemiptera: Pentatomidae). J Braz Chem Soc 22:58–64. https://doi.org/10.1590/S0103-50532011000100007
Fávaro CF, Zarbin PHG (2012) Identification of the defensive compounds in metathoracic and dorsal abdominal glands of the stink bugs Loxa deducta and Pellaea stictica (Heteroptera: Pentatomidae). Quim Nov 35:1582–1586. https://doi.org/10.1590/S0100-40422012000800017
Fávaro CF, Santos TB, Zarbin PHG (2012) Defensive Compounds and Male-Produced Sex Pheromone of the Stink Bug, Agroecus griseus. J Chem Ecol 38:1124–1132. https://doi.org/10.1007/s10886-012-0172-0
Fávaro CF, Soldi RA, Ando T, Aldrich JR, Zarbin PHG (2013) (6R,10S)–Pallantione: The first ketone identified as a sex pheromone in stink bugs. Org Lett 15:1822–1825. https://doi.org/10.1021/ol400413w
Fávaro CF, Millar JG, Zarbin PHG (2015) Identification and synthesis of the male-produced sex pheromone of the stink bug, Pellaea stictica. J Chem Ecol 41:859–868. https://doi.org/10.1007/s10886-015-0621-7
Ferreira JTB, Zarbin PHG (1996) Pheromone syntheses: A tropical approach. Enantioselective synthesis of the (2R,6S,10S) and (2S,6S,10S) isomers of methyl 2,6,10-trimethyldodecanoate. Bioorg Med Chem 4:381–388. https://doi.org/10.1016/0968-0896(96)00015-6
Fouquet G, Schlosser M (1974) Improved carbon-carbon linking by controlled copper catalysis. Angew Chem 13:82–83. https://doi.org/10.1002/anie.197400821
Fujita K, Mori K (2001) Synthesis of (2R,4R)-supellapyrone, the sex pheromone of the brownbanded cockroach, Supella longipalpa, and its three stereoisomers. Eur J Org Chem 2001:493-502. https://doi.org/10.1002/1099-0690(200102)2001:3<493::AID-EJOC493>3.0.CO;2-B
Hartung WH, Simonoff R (2004) Hydrogenolysis of benzyl groups attached to oxygen, nitrogen or sulfur. Org React 7:263–326. https://doi.org/10.1002/0471264180.or007.05
Heathcock CH, Ratcliffe R (1971) Stereoselective total synthesis of the guaiazulenic sesquiterpenoids. α-bulnesene and bulnesol. J Am Chem Soc 93:1746–1757. https://doi.org/10.1021/ja00736a029
Henry TJ (1984) New United States records for two Heteroptera: Pellaea stictica (Pentatomidae) and Rhinacloa pallidipes (Miridae). Proc Entomol Soc Wash 86:519–520
Herber C, Breit B (2006) Iterative deoxypropionate synthesis based on a copper-mediated directed allylic substitution: formal total synthesis of borrelidin (C3-C11 fragment). Chem Eur J 12:6684–6691. https://doi.org/10.1002/chem.200600343
Hoye TR, Jeffrey CS, Shao F (2007) Mosher ester analysis for the determination of absolute configuration of stereogenic (chiral) carbinol carbons. Nat Protoc 2:2451–2458. https://doi.org/10.1038/nprot.2007.354
Lautens M, Colucci JT, Hiebert S, Smith ND, Bouchain G (2002) Total synthesis of ionomycin using ring-opening strategies. Org Lett 4:1879–1882. https://doi.org/10.1021/ol025872f
Leal WS, Zarbin PHG, Wojtasek H, Ferreira JT (1999) Biosynthesis os scarab beetle pheromones – enantioselective 8-hydroxylation of fatty acids. Eur J Biochem 259:175–180. https://doi.org/10.1046/j.1432-1327.1999.00018.x
Li S, Liang S, Tan W, Xu Z, Ye T (2009) Total synthesis of emericellamides A and B. Tetrahedron 65:2695–2702. https://doi.org/10.1016/j.tet.2009.01.082
Lin G, Xu W (1996) Enzymatic synthesis of (2E,4E)-(6R,10R)-4,6,10,12-tetramethyl-2,4-tridecadien-7-one, the sex pheromone of Matsucoccus matsumurae Japanese pine bast scale. Bioorg Med Chem 4:375–380. https://doi.org/10.1016/0968-0896(96)00014-4
McDougal PG, Rico JG, Oh YI, Condon BD (1986) A convenient procedure for the monosilylation of symmetric 1,n-diols. J Org Chem 51:3388–3390. https://doi.org/10.1021/jo00367a033
McPherson JE (2018) Invasive stink bugs and related species (Pentatomoidea): biology, higher systematics, semiochemistry, and management. CRC Press, Boca Raton FL, USA
Moreira JA, Corrêa AG (2003) Enantioselective synthesis of three stereoisomers of 5,9- dimethylpentadecane, sex pheromone component of Leucoptera coffeella, from (-)-isopulegol. Tetrahedron: Asymm 14:3787–3795. https://doi.org/10.1016/j.tetasy.2003.09.030
Morgan ED (2004) Biosynthesis in Insects. Royal Society of Chemistry
Mori K (1989) Synthesis of optically active pheromones. Tetrahedron 45:3233–3298. https://doi.org/10.1016/S0040-4020(01)81007-3
Mori K, Murata N (1994) Synthesis of methyl 2,6,10-trimethyltridecanoate, the male-produced pheromone of the stink bugs, Euschistus heros and E. obscurus, as a stereoisomeric mixture. Liebigs Ann Chem 1994:637–639
Mori K (2007) Significance of chirality in pheromone science. Bioorg Med Chem 15:7505–7523. https://doi.org/10.1016/j.bmc.2007.08.040
Mundy BP, Ellerd MG, Favaloro FG Jr (2005) Name reactions and reagents in organic synthesis. John Wiley & Sons, Hoboken NJ, USA
Negishi E, Tan Z, Liang B, Novak T (2004) An efficient and general route to reduced polypropionates via Zr-catalyzed asymmetric C-C bond formation. Proc Natl Acad Sci 101:5782–5787. https://doi.org/10.1073/pnas.0307514101
Paquette LA, Boulet SL (2002) Toward a total synthesis of okilactomycin. 1. A direct enantiocontrolled route for the western sector. Synthesis 2002:0888–0894. https://doi.org/10.1055/s-2002-28510
Roos J, Effenberger F (1999) Hydroxynitrile lyase catalyzed enantioselective HCN addition to O-protected α-hydroxyaldehydes. Tetrahedron: Asymmetry 10:2817–2828. https://doi.org/10.1002/chin.199952049
Soldi RA, Szczerbowski D, Zarbin PHG (2018) A new enantioselective synthesis of the four stereoisomers of pallantione, the male-produced sex pheromone of Pallantia macunaima (Heretoptera: Pentatomidae). Tetrahedron 74:88–98. https://doi.org/10.1016/j.tet.2017.11.043
Szczerbowski D, Schulz S, Zarbin PHG (2020) Total synthesis of four stereoisomers of methyl 4,8,12-trimethylpentadecanoate, a major component of the sex pheromone of the stink bug Edessa meditabunda. Org Biomol Chem 18:5034–5044. https://doi.org/10.1039/D0OB00862A
Tamura M, Kochi J (1971) Coupling of Grignard reagents with organic halides. Synthesis 1971:303–305
Wang Y, Dong X, Larock RC (2003) Synthesis of naturally occurring pyridine alkaloids via palladium-catalyzed coupling/migration chemistry. J Org Chem 68:3090–3098. https://doi.org/10.1021/jo026716p
Zarbin PHG, Cruz WO, Ferreira JTB (1998) Stereospecific synthesis of two isomers of (4,8)-dimethyldecanal, the aggregation pheromone of Tribolium spp. J Braz Chem Soc 9:511–513. https://doi.org/10.1590/S0103-50531998000500018
Zarbin PHG, Fávaro CF, Vidal DM, Rodrigues MACM (2012) Male-produced sex pheromone of the stink bug Edessa meditabunda. J Chem Ecol 38:825–835. https://doi.org/10.1007/s10886-012-0144-4
Zhai H, Hrabar M, Gries R, Gries G, Britton R (2016) Total synthesis, stereochemical assignment, and field-testing of the sex pheromone of the strepsipteran Xenos peckii. Chem Eur J 22:6190–6193. https://doi.org/10.1002/chem.201505097
Zheng JF, Lan HQ, Yang RF, Peng QL, Xiao ZH, Tuo SC, Hu KZ, Xiang YG, Wei Z, Zhang Z, Huang PQ (2012) Asymmetric syntheses of the sex pheromones of pine sawflies, their homologs and stereoisomers. Helv Chim Acta 95:1799–1808. https://doi.org/10.1002/hlca.201200341
Zhu G, Liang B, Negishi E (2008) Efficient and selective synthesis of (S,R,R,S,R,S)-4,6,8,10,16,18-hexamethyldocosane via Zr-catalyzed asymmetric carboalumination of alkenes (ZACA reaction). Org Lett 10:1099–1101. https://doi.org/10.1021/ol703056u
Acknowledgements
The authors thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Brazil), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and the Instituto Nacional de Ciências e Tecnologia de Semioquímicos na Agricultura (INCT) for financial support. We thank the Programa de Doutorado Sanduíche no Exterior (PDSE) for funding a visiting fellowship for Carla M. B. Gomes to the University of California, Riverside CA, USA.
Funding
This research was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Brazil), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) (for fellowships of Gomes CMB and Souza JPA) and the Instituto Nacional de Ciências e Tecnologia de Semioquímicos na Agricultura (INCT).
Author information
Authors and Affiliations
Contributions
The synthesis and structural characterizations were performed by Gomes CMB and Souza JPA, supervised by Prof. Dr. Zarbin PHG and Prof. Dr. Millar JG. All authors contributed to the writing of manuscript. Prof. Dr. Zarbin PHG and Prof. Dr. Millar JG contributed with the laboratory facilities and financial structure for the development of the experiments.
Corresponding author
Ethics declarations
Conflicts of Interest/Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gomes, C.M.B., Souza, J.P.A., Millar, J.G. et al. Determination of the Absolute Configuration of the Male-Produced Sex Pheromone of the Stink Bug Pellaea stictica, (2R,4R,8R)-2,4,8,13-Tetramethyltetradecan-1-ol by Stereoselective Synthesis Coupled with Enantiomeric Resolution. J Chem Ecol 48, 502–517 (2022). https://doi.org/10.1007/s10886-022-01371-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-022-01371-5