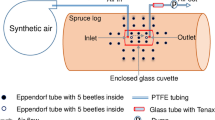
Abstract
The peach bark beetle (Phloeotribus liminaris Harris, PBB) affects the health, quality, and value of black cherry (Prunus serotina Ehrh.) within the Central Hardwoods Forest Region of North America. When colonized by adult beetles, black cherry trees produce a defensive exudate, or ‘gum’, staining the wood and decreasing its value up to 90%. Current management tactics are inadequate to avoid extensive damage to most veneer-sized black cherry in the region. We test the hypothesis that PBB colonization behavior is chemically-mediated and determine the extent to which PBB is attracted to compounds associated with wounded or PBB-infested cherry wood. Through olfactometer and field bioassays, we determined that adult PBB were attracted to cherry branches infested with female beetles. We then used dynamic headspace sampling to collect volatiles associated with wounded and infested bolts of black cherry. The volatile benzaldehyde dominated these collections and was more abundant in aerations of female-infested bolts than other odor sources. In subsequent field bioassays, we evaluated the bioactivity of benzaldehyde, as well as α-longipinene, in combination with several chemical carriers. Traps baited with benzaldehyde captured more PBB than all other treatments, irrespective of other lure components. Moreover, PBB were not attracted to traps baited solely with ethanol, a common attractant for bark beetles that colonize hardwood trees. This is the first report of benzaldehyde as an attractant for a species of bark beetle and could aid in developing semiochemical-based management tactics for this important pest.







Similar content being viewed by others
References
Bentz BJ, Régnière J, Fettig CJ, Hansen MH, Hayes JL, Hicke JA, Kelsey RG, Negrón JF, Seybold SJ (2010) Climate change and bark beetles of the western United States and Canada: direct and indirect effects. BioScience 60:602–613. https://doi.org/10.1525/bio.2010.60.8.6
Blomquist GJ, Figueroa-Teran R, Aw M, Song M, Gorzalski A, Abbott NA, Chang E, Tittiger C (2010) Pheromone production in bark beetles. Insect Biochem Mol Biol 40:699–712. https://doi.org/10.1016/j.ibmb.2010.07.013
Blum MS, Jones TH, House GJ, Tschinkel WR (1981) Defensive secretions of tiger beetles: cyanogenetic basis. Comp Biochem Physiol Part B Comp Biochem 69:903–904
Browne LE (1972) An emergence cage and refrigerated collector for wood-boring insects and their associates. J Econ Entomol 65:1499–1501. https://doi.org/10.1093/jee/65.5.1499
Cassens DL (2004) Factors determining the suitability of trees and logs for the face veneer industry. In: proceedings, 14th central hardwood Forest conference. Gen. Tech. Rep. NE-316. Newtown Square, PA: US Department of Agriculture, Forest Service. Pp 130–139
Deng J-Y, Wei H-Y, Huang Y-P (2004) Enhancement of attraction to sex pheromones of Spodoptera exigua by volatile compounds produced by host plants. J Chem Ecol 30:2037–2045
Dickens JC, Visser JH, Van Der Pers JNC (1993) Detection and deactivation of pheromone and plant odor components by the beet armyworm, Spodoptera exigua (Hübner) (Lepidoptera: Noctuidae). J Insect Physiol 39:503–516
Gitau CW, Bashford R, Carnegie AJ, Gurr GM (2013) A review of semiochemicals associated with bark beetle (Coleoptera: Curculionidae: Scolytinae) pests of coniferous trees: a focus on beetle interactions with other pests and their associates. For Ecol Manag 297:1–14. https://doi.org/10.1016/j.foreco.2013.02.019
Graham K (1968) Anaerobic induction of primary chemical attractancy for ambrosia beetles. Can J Zool 46:905–908
Hofstetter RW, Gaylord ML, Martinson S, Wagner MR (2012) Attraction to monoterpenes and beetle-produced compounds by syntopic Ips and Dendroctonus bark beetles and their predators. Agric For Entomol 14:207–215. https://doi.org/10.1111/j.1461-9563.2011.00560.x
Kenis M, Wermelinger B, Grégoire J-C (2004) Research on parasitoids and predators of Scolytidae – a review. In: Lieutier F, Day KR, Battisti A et al (eds) Bark and Wood boring insects in living trees in Europe, a synthesis. Springer Netherlands, Dordrecht, pp 237–290
Kimmerer TW, Kozlowski TT (1982) Ethylene, ethane, acetaldehyde, and ethanol production by plants under stress. Plant Physiol 69:840–847
Kruskal WH, Wallis WA (1952) Use of ranks in one-criterion variance analysis. J Am Stat Assoc 47:583–621
Kulman HM (1964) Defects in black cherry caused by bark beetles and agromizid cambium miners. For Sci 10:258–266
Leskey TC, Wright SE (2004) Influence of host tree proximity on adult plum curculio (Coleoptera: Curculionidae) responses to monitoring traps. Environ Entomol 33:389–396. https://doi.org/10.1603/0046-225X-33.2.389
McCormick AC, Gershenzon J, Unsicker SB (2014) Little peaks with big effects: establishing the role of minor plant volatiles in plant-insect interactions. Plant Cell Environ 37:1836–1844
Miller DR, Dodds KJ, Hoebeke ER, Poland TM, Willhite EA (2015) Variation in effects of conophthorin on catches of ambrosia beetles (Coleoptera: Curculionidae: Scolytinae) in ethanol-baited traps in the United States. J Econ Entomol 108:183–191. https://doi.org/10.1093/jee/tou043
Miller DR, Lindgren BS (2000) Comparison of a-pinene and myrcene on attraction of mountain pine beetle, Dendroctonus ponderosae (Coleoptera: Scolytidae) to pheromones in stands of western white pine. J Entomol Soc Br Columbia 97:41–46
Miller DR, Rabaglia RJ (2009) Ethanol and (−)-α-pinene: attractant kairomones for bark and ambrosia beetles in the southeastern US. J Chem Ecol 35:435–448. https://doi.org/10.1007/s10886-009-9613-9
Moore BP (1967) Hydrogen cyanide in the defensive secretions of larval Paropsini (Coleoptera: Chrysomelidae). Aust J Entomol 6:36–38. https://doi.org/10.1111/j.1440-6055.1967.tb02135.x
Pereira AE, Vilela EF, Tinoco RS, de Lima JOG, Fantine AK, Morais EGF, França CFM (2012) Correlation between numbers captured and infestation levels of the coffee berry-borer, Hypothenemus hampei : a preliminary basis for an action threshold using baited traps. Int J Pest Manag 58:183–190. https://doi.org/10.1080/09670874.2012.676219
Piñero JC, Wright SE, Prokopy RJ (2001) Response of plum curculio (Coleoptera: Curculionidae) to odor-baited traps near woods. J Econ Entomol 94:1386–1397. https://doi.org/10.1603/0022-0493-94.6.1386
Plaza MT, Rodriguez M, Izquierdo I et al (2000) Analysis and identification of volatiles associated to olive bark beetle Phloeotribus scarabaeoides (Bern.). Contribution to the determination of the semiochemicals involved. Ars Pharm 41:405–413
Power FB, Moore CW (1909) XXXII.—the constituents of the bark of Prunus Serotina. Isolation of l-mandelonitrile glucoside. J Chem Soc Trans 95:243–261
Prokopy RJ, Chandler BW, Dynok SA, Ero JCP (2003) Odor-baited trap trees: a new approach to monitoring plum curculio (Coleoptera: Curculionidae). J Econ Entomol 96:9
Pureswaran DS, Gries R, Borden JH, Pierce, Jr. HD (2000) Dynamics of pheromone production and communication in the mountain pine beetle, Dendroctonus ponderosae Hopkins, and the pine engraver, Ips pini (Say) (Coleoptera: Scolytidae): Chemoecology 10:153–168. https://doi.org/10.1007/PL00001818
Ranger CM, Reding ME, Schultz PB et al (2016) Biology, ecology, and management of nonnative ambrosia beetles (Coleoptera: Curculionidae: Scolytinae) in ornamental plant nurseries. J Integr Pest Manag 7:1–23
Raty L, Drumont A, De Windt N, Grégoire J-C (1995) Mass trapping of the spruce bark beetle Ips typographus L.: traps or trap trees? For Ecol Manag 78:191–205. https://doi.org/10.1016/0378-1127(95)03582-1
Reddy GVP, Guerrero A (2004) Interactions of insect pheromones and plant semiochemicals. Trends Plant Sci 9:253–261. https://doi.org/10.1016/j.tplants.2004.03.009
Rexrode CO (1982) Bionomics of the peach bark beetle, Phloeotribus liminaris (Coleoptera: Scolytidae) in black cherry. J Ga Entomol Soc 17:388–398
Rexrode CO (1981) Gum spots in black cherry caused by natural attacks of peach bark beetle. US Department of Agriculture, Forest Service, northeastern Forest Experiment Station. NE-474: 5
Rexrode CO, Baumgras JE (1984) Distribution of gum spots by causal agent in black cherry and effect on log and tree quality. South J Appl For 8:22–28
Rexrode CO, Krause CR (1981) Sexing Phloeotribus liminaris adults (Coleoptera: Scolytidae) [morphological differences on head, propygidium, and pygidium]. Proc Entomol Soc Wash 83:759–762
Rexrode CO, Smith HC (1990) Occurrence of gum spots in black cherry after partial harvest cutting. US Department of Agriculture, Forest Service, northeastern Forest Experiment Station. NE-634: 7
Ross DW, Daterman GE (1997) Using pheromone-baited traps to control the amount and distribution of tree mortality during outbreaks of the Douglas-fir beetle. For Sci 43:65–70
Ruther J (2004) Male-biassed response of garden chafer, Phyllopertha horticola L., to leaf alcohol and attraction of both sexes to floral plant volatiles. Chemoecology 14:187–192. https://doi.org/10.1007/s00049-004-0271-7
Sankar M, Nowicka E, Carter E, Murphy DM, Knight DW, Bethell D, Hutchings GJ (2014) The benzaldehyde oxication paradox explained by the interception of peroxy radical by benzyl alcohol. Nat Commun 5:3332. https://doi.org/10.1038/ncomms4332
Schlyter F, Zhang Q-H, Liu G-T, Ji L-Z (2001) A successful case of pheromone mass trapping of the bark beetle Ips duplicatus in a forest island, analysed by 20-year time-series data. Integr Pest Manag Rev 6:185–196
Sokal RR, Rohlf FJ (1995) Biometry: the principles of statistics in biological research. WH Freeman and Co., New York, NY
Tillman JA, Holbrook GL, Dallara PL, Schal C, Wood DL, Blomquist GJ, Seybold SJ (1998) Endocrine regulation of de novo aggregation pheromone biosynthesis in the pine engraver, Ips pini (say) (Coleoptera: Scolytidae). Insect Biochem Mol Biol 28:705–715. https://doi.org/10.1016/S0965-1748(97)00117-3
VanDerLaan NR, Ginzel MD (2013) The capacity of conophthorin to enhance the attraction of two Xylosandrus species (Coleoptera: Curculionidae: Scolytinae) to ethanol and the efficacy of verbenone as a deterrent: semiochemicals of two Xylosandrus spp. Agric For Entomol 15:391–397. https://doi.org/10.1111/afe.12026
Weed AS, Ayres MP, Hicke JA (2013) Consequences of climate change for biotic disturbances in north American forests. Ecol Monogr 83:441–470
Wermelinger B (2004) Ecology and management of the spruce bark beetle Ips typographus—a review of recent research. For Ecol Manag 202:67–82. https://doi.org/10.1016/j.foreco.2004.07.018
Wiedenbeck J, Wiemann M, Alderman D, Baumgras J, Luppold W (2004) Defining hardwood veneer log quality attributes. USDA Forest Service, northeastern Research Station. Newtown Square, Pennsylvania
Witzgall P, Kirsch P, Cork A (2010) Sex pheromones and their impact on pest management. J Chem Ecol 36:80–100. https://doi.org/10.1007/s10886-009-9737-y
Wood DL (1982) The bark and ambrosia beetles of North and Central America (Coleoptera: Scolytidae), a taxonomic monograph. Gt Basin Nat Mem 6:1359
Yang CY, Kim J, Kim H-H (2018) Benzaldehyde synergizes the response of female Xyleborinus saxesenii (Coleoptera: Curculionidae: Scolytinae) to ethanol. J Econ Entomol 111:1691–1695. https://doi.org/10.1093/jee/toy131
Zagatti P, Kunesch G, Ramiandrosoa F et al (1987) Sex pheromones of rice moth, Corcyra cephalonica Stainton. J Chem Ecol 13:1561–1573
Acknowledgments
We would like to acknowledge Hannah Quellhorst and Bridget Blood for their technical assistance in field and laboratory experiments. A portion of this research contributed towards a M.S. degree for Nicole VanDerLaan and Ph.D. for Matthew Ethington.
Funding
This research was funded by the Fred M. Van Eck Forestry Foundation.
Author information
Authors and Affiliations
Contributions
Matthew W. Ethington: Conceived and designed the research, conducted lab and field experiments, statistically analyzed the data, and wrote the manuscript. Gabriel P. Hughes: Codesigned research and conducted lab and field experiments. Nikki Van Der Laan: Codesigned research and conducted lab and field experiments. Matthew D. Ginzel: Codesigned research and helped write the manuscript. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
All the procedures performed in studies involving insects were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
This article does not contain any studies with human participants or vertebrate animals performed by any of the authors.
Conflict of Interest
None.
Rights and permissions
About this article
Cite this article
Ethington, M.W., Hughes, G.P., VanDerLaan, N.R. et al. Chemically-mediated colonization of black cherry by the peach bark beetle, Phloeotribus liminaris. J Chem Ecol 47, 303–312 (2021). https://doi.org/10.1007/s10886-021-01256-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-021-01256-z