Abstract
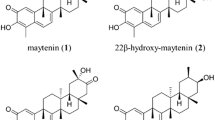
Peritassa campestris (Celastraceae) root bark accumulates potent antitumor quinonemethide triterpenes (QMTs). When grown in their natural habitat, plants of the family Celastraceae produce different QMTs such as celastrol (3) and pristimerin (4). However, when they are inserted in in vitro culture systems, they accumulate maytenin (1) as the main compound. Recently, Bacillus megaterium was detected as an endophytic microorganism (EM) living inside P. campestris roots cultured in vitro. We hypothesized that compound (1) controls EM growth more efficiently, and that the presence of EMs in the root culture causes compound (1) to accumulate. For the first time, this work has explored plant–microorganism interaction in a species of the family Celastraceae by co-culture with an EM. Live endophytic bacteria were used, and QMT accumulation in P. campestris adventitious roots was our main focus. The antimicrobial activity of the main QMTs against endophytic B. megaterium was also evaluated. Our results showed that compound (1) and maytenol (5) were more effective than their precursors QMTs (3) and (4) in controlling the EM. Co-culture of B. megaterium with roots significantly reduced bacterial growth whereas root development remained unaffected. Compound (1) production was 24 times higher after 48 hr in the presence of the highest B. megaterium concentration as compared to the control. Therefore, P. campestris adventitious roots affect the development of the endophyte B. megaterium through production of QMTs, which in turn can modulate production of compound (1).





Similar content being viewed by others
References
Brader G, Compant S, Mitter B et al (2014) Metabolic potential of endophytic bacteria. Curr Opin Biotechnol 27:30–37. https://doi.org/10.1016/J.COPBIO.2013.09.012
Carvalho PRF, Silva DHS, Bolzani VS, Furlan M (2005) Antioxidant quinonemethide triterpenes from Salacia campestris. Chem Biodivers 2:367–372. https://doi.org/10.1002/cbdv.200590016
Chakraborty U, Chakraborty B, Basnet M (2006) Plant growth promotion and induction of resistance in Camellia sinensis by Bacillus megaterium. J Basic Microbiol 46:186–195. https://doi.org/10.1002/jobm.200510050
Chinnaswamy A, Coba de la Peña T, Stoll A et al (2018) A nodule endophytic Bacillus megaterium strain isolated from Medicago polymorpha enhances growth, promotes nodulation by Ensifer medicae and alleviates salt stress in alfalfa plants. Ann Appl Biol 172:295–308. https://doi.org/10.1111/aab.12420
Coppede JS, Pina ES, Paz TA et al (2014) Cell cultures of Maytenus ilicifolia Mart. Are richer sources of quinone-methide triterpenoids than plant roots in natura. Plant Cell Tissue Organ Cult 118:33–43. https://doi.org/10.1007/s11240-014-0459-7
Ferreira DF (2011) Sisvar: a computer statistical analysis system. Ciênc agrotec 35:1039–1042. https://doi.org/10.1590/S1413-70542011000600001
Gaiero JR, McCall CA, Thompson KA et al (2013) Inside the root microbiome: bacterial root endophytes and plant growth promotion. Am J Bot 100:1738–1750. https://doi.org/10.3732/ajb.1200572
Gunatilaka AAL (1996) Triterpenoid quinonemethides and related compounds (Celastroloids). In: Herz W, Kirby GW, Moore RE et al (eds) Fortschritte der Chemie organischer Naturstoffe/Progress in the chemistry of organic natural products. Springer Vienna, Vienna, pp 1–123
Gunatilaka AAL (2006) Natural products from plant-associated microorganisms: distribution, structural diversity, bioactivity, and implications of their occurrence. J Nat Prod 69:509–526. https://doi.org/10.1021/np058128n
Hernandes C, Coppede J d S, Bertoni BW et al (2013) Flash microbiocide: a rapid and economic method for determination of MBC and MFC. Am J Plant Sci 4:850–852. https://doi.org/10.4236/ajps.2013.44104
Hernandes C, Pereira AMS, Severino P (2017) Compounds from Celastraceae targeting cancer pathways and their potential application in head and neck squamous cell carcinoma: a review. Curr Genomics 18:60–74. https://doi.org/10.2174/1389202917666160803160934
Inácio MC, Paz TA, Pereira AMS, Furlan M (2017) Endophytic Bacillus megaterium and exogenous stimuli affect the quinonemethide triterpenes production in adventitious roots of Peritassa campestris (Celastraceae). 131:15–26. https://doi.org/10.1007/s11240-017-1257-9
Kandel S, Joubert P, Doty S et al (2017) Bacterial endophyte colonization and distribution within plants. Microorganisms 5:77. https://doi.org/10.3390/microorganisms5040077
Kildea S, Ransbotyn V, Khan MR et al (2008) Bacillus megaterium shows potential for the biocontrol of Septoria tritici blotch of wheat. Biol Control 47:37–45. https://doi.org/10.1016/J.BIOCONTROL.2008.07.001
Li Y-C, Tao W-Y, Cheng L (2009) Paclitaxel production using co-culture of Taxus suspension cells and paclitaxel-producing endophytic fungi in a co-bioreactor. Appl Microbiol Biotechnol 83:233–239. https://doi.org/10.1007/s00253-009-1856-4
Lloyd G, McCown B (1980) Commercially-feasible micropropagation of mountain laurel, Kalmia latifolia, by use of shoot-tip culture. Int Plant Propag Soc Proc 30:421–427
López-Bucio J, Campos-Cuevas JC, Hernández-Calderón E et al (2007) Bacillus megaterium rhizobacteria promote growth and alter root-system architecture through an auxin- and ethylene-independent signaling mechanism in Arabidopsis thaliana. Mol Plant-Microbe Interact 20:207–217. https://doi.org/10.1094/MPMI-20-2-0207
Paz TA, dos Santos VAFFM, Inácio MC et al (2013) Production of the quinone-methide triterpene maytenin by in vitro adventitious roots of Peritassa campestris (Cambess.) A.C.Sm. (Celastraceae) and rapid detection and identification by APCI-IT-MS/MS. Biomed Res Int 2013:485837. https://doi.org/10.1155/2013/485837
Pina ES, Silva DB, Teixeira SP et al (2016) Mevalonate-derived quinonemethide triterpenoid from in vitro roots of Peritassa laevigata and their localization in root tissue by MALDI imaging. Sci Rep 6:22627. https://doi.org/10.1038/srep22627
Song X, Wu H, Yin Z et al (2017) Endophytic bacteria isolated from Panax ginseng improves ginsenoside accumulation in adventitious ginseng root culture. Molecules 22:837. https://doi.org/10.3390/molecules22060837
Vary PS (1994) Prime time for Bacillus megaterium. Microbiology 140:1001–1013. https://doi.org/10.1099/13500872-140-5-1001
Wang JW, Zhang Z, Tan RX (2001) Stimulation of artemisinin production in Artemisia annua hairy roots by the elicitor from the endophytic Colletotrichum sp. Biotechnol Lett 23:857–860. https://doi.org/10.1023/A:1010535001943
Wu J-Y, Ng J, Shi M, Wu S-J (2007) Enhanced secondary metabolite (tanshinone) production of Salvia miltiorrhiza hairy roots in a novel root–bacteria coculture process. Appl Microbiol Biotechnol 77:543–550. https://doi.org/10.1007/s00253-007-1192-5
Acknowledgements
The authors thank the São Paulo Research Foundation (FAPESP) for the CIBFar-2013/07600-3 grant. M. C. Inácio thanks FAPESP for her fellowships (2014/19362-2 and 2018/00143-0). T. A. Paz thanks the Coordination for the Improvement of Higher Education Personnel (CAPES) for providing his scholarship. A. M. S. Pereira and M. Furlan would also like to thank the National Council for Scientific and Technological Development (CNPq) for their research fellowships. We also thank Dr. Leslie Gunatilaka, Natural Products Center, The University of Arizona, USA, for kindly enabling the conduction of some chromatographic analyses in his laboratory.
Author information
Authors and Affiliations
Contributions
M. C. Inácio was responsible for the conception and design of all experiments, all data analysis, and drafting and editing of the manuscript. T. A. Paz contributed to the experiments performance, HPLC analyses and revision of the manuscript. A. M. S. Pereira supervised the experiments and contributed to data interpretation, drafting and final revision of the manuscript. M. Furlan contributed to data interpretation, drafting and final revision of the manuscript and supervised all experiments.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
ESM 1
(DOCX 269 kb)
Rights and permissions
About this article
Cite this article
Inácio, M.C., Paz, T.A., Pereira, A.M.S. et al. Maytenin Plays a Special Role in the Regulation of the Endophytic Bacillus megaterium in Peritassa campestris Adventitious Roots. J Chem Ecol 45, 789–797 (2019). https://doi.org/10.1007/s10886-019-01096-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-019-01096-y