Abstract
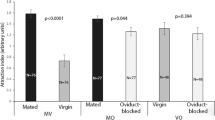
Honey bee (Apis mellifera) queens produce pheromones responsible for mediating both male mating behavior and many critical facets of worker social organization within their colony. These pheromones are dynamic multi-component blends, allowing the communication of detailed information. Indeed, variation in the queen’s mating and reproductive state is associated with significant changes in her pheromone profiles, and these different pheromone profiles elicit different behavioral and physiological responses in female workers. Here we evaluate behavioral responses of male drones to the chemical blends produced by two exocrine glands in queens, and determine if the blends and responses are altered by the queen’s mating and reproductive state. We find that drone attraction to the chemical blends of mandibular glands produced by mated, laying queens versus virgin queens is reduced, suggesting that the queens produce a reliable signal of their mating receptivity. Interestingly, while the chemical blends of mating, laying queens and virgins queens largely overlap, mated, laying queens produce a greater number of chemicals and greater quantities of certain chemicals than virgin queens, suggesting that these chemicals may serve to inhibit behavioral responses of drones to mated, laying queens. Thus, our results highlight the importance of considering chemical cues and signals that serve to both stimulate and inhibit behavioral responses during social interactions in animals.




Similar content being viewed by others
References
Billen JPJ, Dumortier KT, Velthuis HHW (1986) Plasticity of honey bee castes: occurrence of tergal glands in workers. Naturwissenschaften 73:332–333. https://doi.org/10.1007/BF00451486
Blum MS, Jones TH, Rinderer TE, Sylvester HA (1988) Oxygenated compounds in beeswax: Identification and possible significance. Comp Biochem Physiol B Biochem Mol Biol 91(3), 581–583.
Boch R, Shearer DA (1971) Chemical releasers of alarm behaviour in the honey-bee, Apis mellifera. J Insect Physiol 17:2277–2285. https://doi.org/10.1016/0022-1910(71)90077-1
Boch R, Shearer DA, Young JC (1975) Honey bee pheromones: field tests of natural and artificial queen substance. J Chem Ecol 1:133–148. https://doi.org/10.1007/BF00987726
Brandstaetter AS, Bastin F, Sandoz J-C (2014) Honeybee drones are attracted by groups of consexuals in a walking simulator. J Exp Biol 217:1278–1285. https://doi.org/10.1242/jeb.094292
Brockmann A, Dietz D, Spaethe J, Tautz J (2006) Beyond 9-ODA: sex pheromone communication in the European honey bee Apis mellifera L. J Chem Ecol 32:657–667. https://doi.org/10.1007/s10886-005-9027-2
Butler CG (1967) A sex attractant acting as an aphrodisiac in honeybee (Apis Mellifera L.). proc R Entomol Soc London a 42:71-
Christensen TA, Mustaparta H, Hildebrand JG (1995) Chemical communication in heliothine moths: antennal lobe projection patterns of pheromone-detecting olfactory receptor neurons in the male Heliothis virescens. J Comp Physiol A 177:275–280. https://doi.org/10.1007/BF00207184
Cobey SW (2007) Comparison studies of instrumentally inseminated and naturally mated honey bee queens and factors affecting their performance*. Apidologie 38:390–410. https://doi.org/10.1051/apido:2007029
Connor JC (2009) Queen rearing essentials. Wcwas Press, Kalamazoo
Engels W, Rosenkranz P, Adler A et al (1997) Mandibular gland volatiles and their ontogenetic patterns in queen honey bees, Apis mellifera carnica. J Insect Physiol 43:307–313. https://doi.org/10.1016/S0022-1910(96)00110-2
Espelie K, Butz Huryn VM, Dietz A (1990) Decyl decanoate: a major component of the tergite glands of honeybee queens (Apis mellifera L.). J Apic Res 29:15–19
Grozinger CM, Fischer P, Hampton JE (2007) Uncoupling primer and releaser responses to pheromone in honey bees. Naturwissenschaften 94:375–379. https://doi.org/10.1007/s00114-006-0197-8
Holt H, Villar G, Cheng W et al (2018) Molecular, physiological and behavioral responses of honey bee (Apis mellifera) drones to infection with microsporidian parasites. J Invertebr Pathol 155:14–24
Hoover SER, Keeling CI, Winston ML, Slessor KN (2003) The effect of queen pheromones on worker honey bee ovary development. Naturwissenschaften 90:477–480. https://doi.org/10.1007/s00114-003-0462-z
Ikeno H, Akamatsu T, Hasegawa Y, Ai H (2014) Effect of olfactory stimulus on the flight course of a honeybee, Apis mellifera, in a wind tunnel. Insects 5:92–104. https://doi.org/10.3390/insects5010092
Keeling CI, Slessor KN, Higo HA, Winston ML (2003) New components of the honey bee (Apis mellifera L.) queen retinue pheromone. Proc Natl Acad Sci U S A 100:4486–4491. https://doi.org/10.1073/pnas.0836984100
Kocher SD, Grozinger CM (2011) Cooperation, conflict, and the evolution of queen pheromones. J Chem Ecol 37:1263–1275. https://doi.org/10.1007/s10886-011-0036-z
Kocher SD, Richard FJ, Tarpy DR, Grozinger CM (2009) Queen reproductive state modulates pheromone production and queen-worker interactions in honeybees. Behav Ecol 20:1007–1014. https://doi.org/10.1093/beheco/arp090
Koeniger N, Koeniger G (2000) Reproductive isolation among species of the genus Apis. Apidologie 31:313–339. https://doi.org/10.1051/apido:2000125
Laidlaw HH, Page RE (1996) Queen rearing and bee breeding. Wicwas Press, Kalamazoo
Le Conte Y, Hefetz A (2008) Primer pheromones in social Hymenoptera. Annu Rev Entomol 53:523–542. https://doi.org/10.1146/annurev.ento.52.110405.091434
Loper GM, Wolf WW, Taylor OR (1992) Honey bee drone flyways and congregation areas : radar observations. J Kansas Entomol Soc 65:223–230
Loper GM, Taylor OR, Foster LJ, Kochansky J (1996) Relative attractiveness of queen mandibular pheromone components to honey bee (Apis mellifera) drones. J Apic Res 35:122–123
Maisonnasse A (2010) New insights into honey bee (Apis mellifera) pheromone communication. Is the queen mandibular pheromone alone in colony regulation? Front Zool 7:18. https://doi.org/10.1186/1742-9994-7-18
Melathopoulos AP, Winston ML, Pettis JS, Pankiw T (1996) Effect of queen mandibular pheromone on initiation and maintenance of queen cells in the honey bee (Apis mellifera L.). Can Entomol 128:263–272
Mustaparta H (1996) Central mechanisms of pheromone information processing. Chem Senses 21:269–275. https://doi.org/10.1093/chemse/21.2.269
Niño EL, Tarpy DR, Grozinger C (2011) Genome-wide analysis of brain transcriptional changes in honey bee ( Apis mellifera L .) queens exposed to. 20:387–398. https://doi.org/10.1111/j.1365-2583.2011.01072.x
Niño EL, Malka O, Hefetz A et al (2013) Chemical profiles of two pheromone glands are differentially regulated by distinct mating factors in honey bee queens (Apis mellifera L.). PLoS one 8. https://doi.org/10.1371/journal.pone.0078637
Nirazawa T, Fujii T, Seki Y, Namiki S, Kazawa T, Kanzaki R, Ishikawa Y (2017) Morphology and physiology of antennal lobe projection neurons in the hawkmoth Agrius convolvuli. J Insect Physiol 98:214–222. https://doi.org/10.1016/j.jinsphys.2017.01.010
Okosun OO, Yusuf AA, Crewe RM, Pirk CWW (2019) Tergal gland components of reproductively dominant honey bee workers have both primer and releaser effects on subordinate workers. Apidologie. 50:173–182. https://doi.org/10.1007/s13592-018-0628-5
Pankiw T, Huang ZY, Winston ML, Robinson GE (1998) Queen mandibular gland pheromone influences worker honey bee (Apis mellifera L.) foraging ontogeny and juvenile hormone titers. J Insect Physiol 44:685–692. https://doi.org/10.1016/S0022-1910(98)00040-7
Plettner E, Otis GW, Wimalaratne PDC, Winston ML, Slessor KN, Pankiw T, Punchihewa PWK (1997) Species- and caste-determined mandibular gland signals in honeybees (Apis). J Chem Ecol 23:363–377. https://doi.org/10.1023/B:JOEC.0000006365.20996.a2
Renner M, Vierling G (1977) Die rolle des taschendrusenpheromons beim hochzeitsflug der bienenkonigin. Behav Ecol Sociobiol 2:329–338
Rhodes JW, Lacey MJ, Harden S (2007) Changes with age in queen honey bee (Apis mellifera) head chemical constituents (Hymenoptera: Apidae). Sociobiology 50:11–22
Richard FJ, Tarpy DR, Grozinger CM (2007) Effects of insemination quantity on honey bee queen physiology. PLoS One 2:e980. https://doi.org/10.1371/journal.pone.0000980
Rueppell O, Page RE, Fondrk MK (2006) Male behavioural maturation rate responds to selection on pollen hoarding in honeybees. Anim Behav 71:227–234. https://doi.org/10.1016/j.anbehav.2005.05.008
Sandoz J-C (2006) Odour-evoked responses to queen pheromone components and to plant odours using optical imaging in the antennal lobe of the honey bee drone Apis mellifera L. J Exp Biol 209:3587–3598. https://doi.org/10.1242/jeb.02423
Slessor KN, Kaminski L-A, King GGS, Borden JH, Winston ML (1988) Semiochemical basis of the retinue response to queen honey bees. Nature 332:354–356
Slessor KN, Kaminski LA, King GGS, Winston ML (1990) Semiochemicals of the honeybee queen mandibular glands. J Chem Ecol 16:851–860. https://doi.org/10.1007/BF01016495
Strang GE (1970) A study of honey bee drone attraction in the mating response. J Econ Entomol 63:641–645
Strauss K, Scharpenberg H, Crewe RM, Glahn F, Foth H, Moritz RFA (2008) The role of the queen mandibular gland pheromone in honeybees (Apis mellifera): honest signal or suppressive agent? Behav Ecol Sociobiol 62:1523–1531. https://doi.org/10.1007/s00265-008-0581-9
Trhlin M, Rajchard J (2011) Chemical communication in the honeybee ( Apis mellifera L .): a review. Ecology 2011:265–273
Varela N, Couton L, Gemeno C, Avilla J, Rospars JP, Anton S (2009) Three-dimensional antennal lobe atlas of the oriental fruit moth, Cydia molesta (Busck) (Lepidoptera: Tortricidae): comparison of male and female glomerular organization. Cell Tissue Res 337:513–526. https://doi.org/10.1007/s00441-009-0839-1
Villar G, Grozinger CM (2017) Primer effects of the honeybee, Apis mellifera, queen pheromone 9-ODA on drones. Anim Behav 127:271–279. https://doi.org/10.1016/j.anbehav.2017.03.023
Villar G, Wolfson MD, Hefetz A, Grozinger CM (2018) Evaluating the role of drone-produced chemical signals in mediating social interactions in honey bees (Apis mellifera). J Chem Ecol 44(1), 1–8. https://doi.org/10.1007/s10886-017-0912-2
Wanner KW, Nichols AS, Walden KKO, Brockmann A, Luetje CW, Robertson HM (2007) A honey bee odorant receptor for the queen substance 9-oxo-2-decenoic acid. Proc Natl Acad Sci U S A 104:14383–14388. https://doi.org/10.1073/pnas.0705459104
Wittmann D, Radtke R, Zeil J, Lübke G, Francke W (1990) Robber bees (Lestrimelitta limao) and their host chemical and visual cues in nest defense by Trigona (Tetragonisca) angustula (Apidae: Meliponinae). J Chem Ecol 16:631–641. https://doi.org/10.1007/BF01021793
Wossler TC, Crewe RM (1999a) The releaser effects of the tergal gland secretion of queen honeybees (Apis mellifera). J Insect Behav 12:343–351
Wossler TC, Crewe RM (1999b) Mass spectral identification of the tergal gland secretions of female castes of two African honey bee races (Apis mellifera). J Apic Res 38:137–148. https://doi.org/10.1080/00218839.1999.11101004
Acknowledgments
We are grateful to Mario Padilla for expert beekeeping assistance, the Grozinger laboratory members for helpful comments, and to Dr. Margarita Orlova for her assistance with our chemical analysis. This work was supported by a USDA-NIFA Predoctoral Fellowship to G.V. (2015-67011-22802), a USDA-SARE student research grant awarded to G.V. (GNE14-090), an Alfred P. Sloan Foundation Fellowship awarded to G.V., the United States-Israel Binational Science Foundation (grant # 2013104) to C.MG., A.H., N. Altman.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(XLSX 26 kb)
Rights and permissions
About this article
Cite this article
Villar, G., Hefetz, A. & Grozinger, C.M. Evaluating the Effect of Honey Bee (Apis mellifera) Queen Reproductive State on Pheromone-Mediated Interactions with Male Drone Bees. J Chem Ecol 45, 588–597 (2019). https://doi.org/10.1007/s10886-019-01086-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-019-01086-0