Abstract
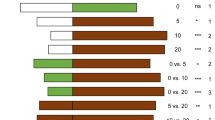
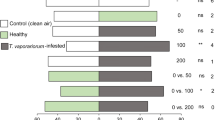
In response to attack by herbivorous insects, plants produce semiochemicals for intra- and interspecific communication. The perception of these semiochemicals by conspecifics of the herbivore defines their choice for oviposition and feeding. We aimed to investigate the role of herbivore-induced plant volatiles (HIPVs) by Tuta absoluta larvae on the oviposition choice of conspecific females on tomato plants. We performed two- choice and non-choice bioassays with plants damaged by larvae feeding and intact control plants. We also collected headspace volatiles of those plants and tested the response of female antennae on those blends with Gas Chromatography- Electro-Antennographical Detection (GC-EAD). In total 55 compounds were collected from the headspace of T. absoluta larvae-infested plants. Our results show that female moths preferred to oviposit on intact control plants instead of damaged ones. Herbivory induced the emission of hexanal, (Ζ)-3-hexen-1-ol, (E)-β-ocimene, linalool, (Z)-3-hexenyl butanoate, methyl salicylate, indole, nerolidol, guaidiene-6,9, β-pinene, β-myrcene, α-terpinene, hexenyl hexanoate, β-elemene, β-caryophyllene and (Ε-Ε)- 4,8,12-trimethyl-1,3,7,11-tridecatetraene (TMTT), one unidentified sesquiterpene and three unknown compounds. In Electroantennographic (EAG) assays, the antennae of T. absoluta females responded to hexanal, (Ζ)-3-hexen-1-ol, methyl salicylate and indole. The antennae of T. absoluta females exhibited a dose-response in EAG studies with authentic samples. Strong EAG responses were obtained for compounds induced on damaged tomato plants, as well as in nonanal, a compound emitted by both infested and control plants. These compounds could be utilized in integrated pest management of T. absoluta.



Similar content being viewed by others
References
Adams R (2007) Identification of essential oil components by gas-chromatography/mass spectrometry, 4th edn. Allured Business Media, Illinois
Allmann S et al (2013) Feeding-induced rearrangement of green leaf volatiles reduces moth oviposition. elife 2:e00421. https://doi.org/10.7554/eLife.00421
Anastasaki E, Balayannis G, Papanikolaou NE, Michaelakis AN, Milonas PG (2015) Oviposition induced volatiles in tomato plants. Phytochem Lett 13:262–266. https://doi.org/10.1016/j.phytol.2015.07.007
Angeles Lopez YI, Martinez-Gallardo NA, Ramirez-Romero R, Lopez MG, Sanchez-Hernandez C, Delano-Frier JP (2012) Cross-kingdom effects of plant-plant signaling via volatile organic compounds emitted by tomato (Solanum lycopersicum) plants infested by the greenhouse whitefly (Trialeurodes vaporariorum). J Chem Ecol 38:1376–1386. https://doi.org/10.1007/s10886-012-0201-z
Ataide LMS, Arce CCM, Curtinhas JN, da Silva DJH, DeSouza O, Lima E (2017) Flight behavior and oviposition of Tuta absoluta on susceptible and resistant genotypes of Solanum lycopersicum. Arthropod-Plant Inte 11:567–575. https://doi.org/10.1007/s11829-017-9500-1
Awmark C, Leather SR (2002) Host plant quality and fecundity in herbivorous insects. Annu Rev Entomol 47:817–844
Bawin T, De Backer L, Dujeu D, Legrand P, Megido RC, Francis F, Verheggen FJ (2014) Infestation level influences oviposition site selection in the tomato leafminer Tuta absoluta (Lepidoptera: Gelechiidae). Insects 5:877–884. https://doi.org/10.3390/insects5040877
Bawin T, Collard F, De Backer L, Yarou BB, Compere P, Francis F, Verheggen FJ (2017) Structure and distribution of the sensilla on the antennae of Tuta absoluta (Lepidoptera: Gelechiidae). Micron 96:16–28. https://doi.org/10.1016/j.micron.2017.01.008
Biondi A, Guedes RNC, Wan FH, Desneux N (2018) Ecology, worldwide wpread and management of the invasive south American tomato pinworm, Tuta absoluta: past, present, and future. Annu Rev Entomol 63. https://doi.org/10.1146/annurev-ento-031616-034933
Birkett MA et al (2000) New roles for cis-jasmone as an insect semiochemical and in plant defense. P Natl Acad Sci USA 97:9329–9334
Bruce TJ, Wadhams LJ, Woodcock CM (2005) Insect host location: a volatile situation. Trends Plant Sci 10:269–274. https://doi.org/10.1016/j.tplants.2005.04.003
Caparros Megido R et al (2014) Role of larval host plant experience and solanaceous plant volatile emissions in Tuta absoluta (Lepidoptera: Gelechiidae) host finding behavior. Arthropod-Plant Inte. https://doi.org/10.1007/s11829-014-9315-2
Cook SM, Khan ZR, Pickett JA (2007) The use of push-pull strategies in integrated pest management. Annu Rev Entomol 52:375–400
Copolovici L, Kännaste A, Pazouki L, Niinemets U (2012) Emissions of green leaf volatiles and terpenoids from Solanum lycopersicum are quantitatively related to the severity of cold and heat shock treatments. J Plant Physiol 169:664–672. https://doi.org/10.1016/j.jplph.2011.12.019
De Backer L, Megido RC, Fauconnier M-L, Brostaux Y, Francis F, Verheggen F (2015) Tuta absoluta-induced plant volatiles: attractiveness towards the generalist predator Macrolophus pygmaeus. Arthropod-Plant Inter 9:465–476. https://doi.org/10.1007/s11829-015-9388-6
De Moraes CM, Mescher MC, Tumlinson JH (2001) Caterpillar-induced nocturnal plant volatiles repel nonspecific females. Nature 410:577–580
Dicke M (2000) Chemical ecology of host plant selection by herbivorous arhtopods: a multitrophic perpective. Biochem Syst Ecol 28:601–617
Dicke M, Baldwin IT (2010) The evolutionary context for herbivore-induced plant volatiles: beyond the 'cry for help'. Trends Plant Sci 15:167–175. https://doi.org/10.1016/j.tplants.2009.12.002
Dicke M, van Poecke RMP, de Boer JG (2003) Inducible indirect defence of plants: from mechanisms to ecological functions. Basic Appl Ecol 4:27–42. https://doi.org/10.1078/1439-1791-00131
Erb M, Veyrat N, Robert CA, Xu H, Frey M, Ton J, Turlings TC (2015) Indole is an essential herbivore-induced volatile priming signal in maize. Nat Commun 6:6273. https://doi.org/10.1038/ncomms7273
Fatouros NE et al (2012) Plant volatiles induced by herbivore egg deposition affect insects of different trophic levels. PLoS One 7:e43607. https://doi.org/10.1371/journal.pone.0043607
Fatouros NE, Cusumano A, Danchin EGJ, Colazza S (2016) Prospects of herbivore egg-killing plant defenses for sustainable crop protection. Ecol Evol 6:6906–6918. https://doi.org/10.1002/ece3.2365
Furtado FB et al (2014) Seasonal variation of the chemical composition and antimicrobial and cytotoxic activities of the essential oils from Inga laurina (Sw.) Willd. Molecules 19:4560–4577. https://doi.org/10.3390/molecules19044560
Gripenberg S, Mayhew PJ, Parnell M, Roslin T (2010) A meta-analysis of preference–performance relationships in phytophagous insects. Ecol Lett 13. https://doi.org/10.1111/j.1461-0248.2009.01433.x
Heil M (2008) Indirect defence via tritrophic interactions. New Phytol 178:41–61. https://doi.org/10.1111/j.1469-8137.2007.02330.x
Hilker M, Fatouros NE (2015) Plant responses to insect egg deposition. Annu Rev Entomol 60:493–515. https://doi.org/10.1146/annurev-ento-010814-020620
IOFI (2011) Guidelines for the quantitative gas chromatography of volatile flavouring substances, from the working group on Methods of analysis of the International Organization of the Flavor Industry (IOFI). Flavour Frag J 26:297–299. https://doi.org/10.1002/ffj.2061
Kant MR, Ament K, Sabelis MW, Haring MA, Schuurink RC (2004) Differential timing of spider mite-induced direct and indirect defenses in tomato plants. Plant Physiol 135:483–495. https://doi.org/10.1104/pp.103.038315
Karban R (2011) The ecology and evolution of induced resistance against herbivores. Funct Ecol 25:339–347. https://doi.org/10.1111/j.1365-2435.2010.01789.x
Kessler A, Baldwin IT (2001) Defensive function of herbivore-induced plant volatile emissions in nature. Science 291:2141–2144. https://doi.org/10.1126/science.291.5511.2141
Khan ZR, James DG, Midega CAO, Pickett JA (2008) Chemical ecology and conservation biological control. Biol Control 45:210–224. https://doi.org/10.1016/j.biocontrol.2007.11.009
Pashalidou FG, Lucas-Barbosa D, van Loon JJA, Dicke M, Fatouros NE (2013) Phenotypic plasticity of plant response to herbivore eggs: effects on resistance to caterpillars and plant development. Ecology 94:702–713
Pereyra PC, Sánchez NE (2006) Effect of two solanaceous plants on developmental and population parameters of the tomato leaf miner, Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). Neotrop Entomol 35:565–574
Pinto-Zevallos DM, Strapasson P, Zarbin PHG (2016) Herbivore-induced volatile organic compounds emitted by maize: electrophysiological responses in Spodoptera frugiperda females. Phytochem Lett 16:70–74. https://doi.org/10.1016/j.phytol.2016.03.005
Poelman EH, Broekgaarden C, van Loon JJA, Dicke M (2008) Early season herbivore differentially affects plant defence responses to subsequently colonizing herbivores and their abundance in the field. Mol Ecol 17:3352–3365. https://doi.org/10.1111/j.1365-294X.2008.03838.x
Ponzio C et al (2016) Volatile-mediated foraging behaviour of three parasitoid species under conditions of dual insect herbivore attack. Anim Behav 111:197–206. https://doi.org/10.1016/j.anbehav.2015.10.024
Proffit M, Birgersson G, Bengtsson M, Reis R Jr, Witzgall P, Lima E (2011) Attraction and oviposition of Tuta absoluta females in response to tomato leaf volatiles. J Chem Ecol 37:565–574. https://doi.org/10.1007/s10886-011-9961-0
Raitanen J, Forsman JT, Kivela SM, Maenpaa MI, Valimaki P (2014) Attraction to conspecific eggs may guide oviposition site selection in a solitary insect. Behav Ecol 25:110–116. https://doi.org/10.1093/beheco/art092
Reisenman CE, Riffell JA, Duffy K, Pesque A, Mikles D, Goodwin B (2013) Species-specific effects of herbivory on the oviposition behavior of the moth Manduca sexta. J Chem Ecol 39:76–89. https://doi.org/10.1007/s10886-012-0228-1
Renwick JAA, Chew FS (1994) Species-specific effects of herbivory on the oviposition behavior of the moth Manduca sexta. J Chem Ecol 39:377–400
Scala A, Allmann S, Mirabella R, Haring MA, Schuurink RC (2013) Green leaf volatiles: a plant's multifunctional weapon against herbivores and pathogens. Int J Mol Sci 14:17781–17811. https://doi.org/10.3390/ijms140917781
Shiojiri K, Takabayashi J, Yano S, Takafuji A (2002) Oviposition preferences of herbivores are affected by tritrophic interaction webs. Ecol Lett 5:186–192
Silva DB, Bueno VHP, Lins JC Jr, van Lenteren JC (2015) Life history data and population growth of Tuta absoluta at constant and alternating temperatures on two tomato lines. B Instectol 68:223–232
Silva DB, Weldegergis BT, Van Loon JJ, Bueno VH (2017) Qualitative and quantitative differences in herbivore-induced plant volatile blends from tomato plants infested by either Tuta absoluta or Bemisia tabaci. J Chem Ecol 43:53–65. https://doi.org/10.1007/s10886-016-0807-7
Smith RM, Marshall JA, Davey MR, Lowe KC, Power JB (1996) Comparison of volatiles and waxes in leaves of genetically engineered tomatoes. Phytochemistry 43:753–758
Sokal RR, Rohlf FJ (2012) Biometry: the principles and practice of statistics in biological research. 4 edn. W. H. Freeman and Co, New York
Song C, Lai WC, Reddy KM, Wei B (2003) Temperature-programmed retention indices for GC and GC-MS of hydrocarbon fuels and simulateddistillation of GC of heavy oils. In: Hsu CS (ed) Analytical advances for hydrocarbon research, 1st edn. Springer Science & Business Media, New York, pp 147–210
Stenberg JA, Heil M, Åhman I, Björkman C (2015) Optimizing crops for biocontrol of pests and disease. Trends Plant Sci 20:698–712. https://doi.org/10.1016/j.tplants.2015.08.007
Strapasson P, Pinto-Zevallos DM, Paudel S, Rajotte EG, Felton GW, Zarbin PH (2014) Enhancing plant resistance at the seed stage: low concentrations of methyl jasmonate reduce the performance of the leaf miner Tuta absoluta but do not alter the behavior of its predator Chrysoperla externa. J Chem Ecol 40:1090–1098. https://doi.org/10.1007/s10886-014-0503-4
Teles Pontes WJ, Lima ER, Cunha EG, TDA PM, Lôbo AP, Barros R (2010) Physical and chemical cues affect oviposition by Neoleucinodes elegantalis. Physiol Entomol 35:134–139. https://doi.org/10.1111/j.1365-3032.2010.00720.x
Thompson JN (1988) Evolutionary ecology of the relationship between oviposition preference and performance of offspring in phytophagous insects. Entomol exp appl 47:3–14
Ulland S, Ian E, Mozuraitis R, Borg-Karlson AK, Meadow R, Mustaparta H (2008) Methyl salicylate, identified as primary odorant of a specific receptor neuron type, inhibits oviposition by the moth Mamestra brassicae L. (Lepidoptera, noctuidae). Chem Senses 33:35–46. https://doi.org/10.1093/chemse/bjm061
Zhuang X, Fiesselmann A, Zhao N, Chen H, Frey M, Chen F (2012) Biosynthesis and emission of insect herbivory-induced volatile indole in rice. Phytochemistry 73:15–22. https://doi.org/10.1016/j.phytochem.2011.08.029
Acknowledgements
We would like to thank two anonymous reviewers and the handling editor for their valuable comments that considerably improve the manuscript. We also thank Apostolos Kapranas for his comments and English language editing.
Funding
The present study was funded by the General Secretariat Research and Technology of the Greek Ministry of Education within the action “EXCELLENCE II” under the Operational Programme “Education and Lifelong Learning” 2007–2013 that is co-funded by the European Social Fund and National funds.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Anastasaki, E., Drizou, F. & Milonas, P.G. Electrophysiological and Oviposition Responses of Tuta absoluta Females to Herbivore-Induced Volatiles in Tomato Plants. J Chem Ecol 44, 288–298 (2018). https://doi.org/10.1007/s10886-018-0929-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-018-0929-1