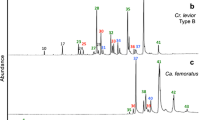
Abstract
Termite nests often are referred to as the most elaborate constructions of animals. However, some termite species do not build a nest at all and instead found colonies inside the nests of other termites. Since these so-called inquilines do not need to be in direct contact with the host population, the two colonies usually live in separate parts of the nest. Adaptations of both the inquiline and its host are likely to occur to maintain the spatial exclusion and reduce the costs of potential conflicts. Among them, mutual avoidance, based on chemical cues, is expected. We investigated chemical aspects of cohabitation between Constrictotermes cavifrons (Nasutitermitinae) and its obligatory inquiline Inquilinitermes inquilinus (Termitinae). Inquiline soldiers produce in their frontal glands a blend of wax esters, consisting of the C12 alcohols (3Z)-dodec enol, (3Z,6Z)-dodecadienol, and dodecanol, esterified with different fatty acids. The C12 alcohols appear to be cleaved gradually from the wax esters, and they occur in the frontal gland, in soldier headspace, and in the walls of the inquiline part of the nest. Electrophysiological experiments revealed that (3Z)-dodecenol and (3Z,6Z)-dodecadienol are perceived by workers of both species. Bioassays indicated that inquiline soldier heads, as well as the two synthetic compounds, are attractive to conspecific workers and elicit an arresting behavior, while host soldiers and workers avoid these chemicals at biologically relevant amounts. These observations support the hypothesis that chemically mediated spatial separation of the host and the inquiline is an element of a conflict-avoidance strategy in these species.




Similar content being viewed by others
References
Bagnères A-G, Hanus R (2015) Communication and social regulation in termites. In: Aquiloni L, Tricarico E (eds) Social recognition in invertebrates: the Knowns and the unknowns. Springer, Dordrecht, pp. 193–248
Bordereau C, Pasteels JM (2011) Pheromones and chemical ecology of dispersal and foraging in termites. In: Bignell DE, Roisin Y, Lo N (eds) Biology of termites: a modern synthesis. Springer, Dordrecht, pp. 279–320
Bourguignon T, Šobotník J, Lepoint G, Martin JM, Hardy OJ, Dejean A, Roisin Y (2011) Feeding ecology and phylogenetic structure of a complex neotropical termite assemblage, revealed by nitrogen stable isotope ratios. Ecol Entomol 36:261–269
Costa DA, Carvalho RA, Lima-Filho GF, Brandão D (2009) Inquilines and invertebrate fauna associated with termite nests of Cornitermes cumulans (Isoptera, Termitidae) in the Emas National Park, Mineiros, Goiás, Brazil. Sociobiology 53:443–453
Cristaldo PF, DeSouza O, Krasulová J, Jirošová A, Kutalová K, Rodrigues Lima E, Šobotník J, Sillam-Dussès D (2014) Mutual use of trail-following chemical cues by a termite host and its inquilines. PLoS One 9:e85315
Cristaldo PF, Rosa CS, Florencio DF, Marins A, DeSouza O (2012) Termitarium volume as a determinant of invasion by obligatory termitophiles and inquilines in the nests of Constrictotermes cyphergaster (Termitidae, Nasutitermitinae). Insect Soc 59:541–548
Cunha HF, Costa DA, Filho KE, Silva LO, Brandão D (2003) Relationship between Constrictotermes cyphergaster and inquiline termites in the Cerrado (Isoptera: Termitidae). Sociobiology 42:761–770
Deligne J, Quennedey A, Blum MS (1981) The enemies and defense mechanisms of termites. In: Hermann HR (ed) Social insects, vol Volume 2. Academic Press, New York, pp. 1–76
Devigne C, Detrain C (2002) Collective exploration and area marking in the ant Lasius niger. Insect Soc 49:357–362
Florencio DF, Marins A, Rosa CS, Cristaldo PF, Araujo APA, Silva IR, DeSouza O (2013) Diet segregation between cohabiting builder and inquiline termite species. PLoS One 8:e66535
Fuller CA (2007) Fungistatic activity of freshly killed termite, Nasutitermes acajutlae, soldiers in the Caribbean. J Insect Sci 7:1–8
Higashi M, Yamamura N, Abe T, Burns TP (1991) Why don’t all termite species have a sterile worker caste? Proc R Soc B 246:25–29
Hölldobler B (1976) Recruitment behavior, home range orientation and territoriality in harvester ants, Pogonomyrmex. Behav Ecol Sociobiol 1:3–44
Hölldobler B, Wilson EO (1977) Colony-specific territorial pheromone in the African weaver ant Oecophylla longinoda (Latreille). Proc Natl Acad Sci U S A 74:2072–2075
Kilner RM, Langmore NE (2011) Cuckoos versus hosts in insects and birds: adaptations, counter-adaptations and outcomes. Biol Rev Camb Philos Soc 86:836–852
Kyjaková P, Dolejšová K, Krasulová J, Bednárová L, Hadravová R, Pohl R, Hanus R (2015) The evolution of symmetrical snapping in termite soldiers need not lead to reduced chemical defence. Biol J Linnean Soc 115:818–825
Kyjaková P, Roy V, Jirošová A, Krasulová J, Dolejšová K, Křivánek J, Hadravová R, Rybáček J, Pohl R, Roisin Y, Sillam-Dussès D, Hanus R (2016) Chemical systematics of neotropical termite genera with symmetrically snapping soldiers (Termitidae: Termitinae). Zool J Linnean Soc. doi:10.1111/zoj.12486
Langen TA, Tripet F, Nonacs P (2000) The red and the black: habituation and the dear-enemy phenomenon in two desert Pheidole ants. Behav Ecol Sociobiol 48:285–292
Martius C, Amelung W, Garcia MVB (2000) The Amazonian forest termite (Isoptera: Termitidae) (Constrictotermes cavifrons) feeds on microepiphytes. Sociobiology 35:379–383
Millar JG, Haynes KF (1998) Methods in chemical ecology: chemical methods. Springer, New York
Noirot C (1970) The nests of termites. In: Krishna K, Weesner FM (eds) Biology of termites, vol Volume II. Academic Press, New York, pp. 73–125
Peppuy A, Robert A, Sémon E, Bonnard O, Son NT, Bordereau C (2001a) Species specificity of trail pheromones of fungus-growing termites from northern Vietnam. Insect Soc 48:245–250
Peppuy A, Robert A, Sémon E, Giniès C, Letteré M, Bonnard O, Bordereau C (2001b) (Z)-dodec-3-en-1-ol, a novel termite trail pheromone identified after solid phase microextraction from Macrotermes annandalei. J Insect Physiol 47:445–453
Prestwich GD (1984) Defense mechanisms of termites. Annu Rev Entomol 29:201–232
Quennedey A (1984) Morphology and ultrastructure of termite defense glands. In: Hermann HR (ed) Defensive mechanisms in social insects. Praeger, New York, pp. 151–200
Quennedey A, Sillam-Dussès D, Robert A, Bordereau C (2008) The fine structural organization of sternal glands of pseudergates and workers in termites (Isoptera): a comparative survey. Arthropod Struct Dev 37:168–185
Robert A, Peppuy A, Sémon E, Boyer FD, Lacey MJ, Bordereau C (2004) A new C12 alcohol identified as a sex pheromone and a trail following pheromone in termites: the diene (Z,Z)-dodeca-3,6-dien-1-ol. Naturwissenschaften 91:34–39
Rosengaus RB, Lefebvre ML, Traniello JFA (2000) Inhibition of fungal spore germination by Nasutitermes: evidence for a possible antiseptic role of soldier defensive secretions. J Chem Ecol 26:21–39
Shellman-Reeve JS (1997) The spectrum of eusociality in termites. In: Choe J, Crespi BJ (eds) The evolution of social behavior in insects and arachnids. Cambridge University Press, Cambridge, pp. 52–93
Sillam-Dussès D (2010) Trail pheromones and sex pheromones in termites. Nova Publishers, New York
Sillam-Dussès D (2011) Trail pheromones and sex pheromones in termites: glandular origin, chemical nature, and potential use in pest management. In: Gregory IM (ed) Pheromones: theories. Types and Uses. Nova Publishers, New York, pp. 39–92
Sillam-Dussès D, Kalinová B, Jiroš P, Březinová A, Cvačka J, Hanus R, Šobotník J, Bordereau C, Valterová I (2009a) Identification by GC-EAD of the two-component trail-following pheromone of Prorhinotermes simplex (Isoptera, Rhinotermitidae, Prorhinotermitinae). J Insect Physiol 55:751–757
Sillam-Dussès D, Sémon E, Lacey MJ, Robert A, Lenz M, Bordereau C (2007) Trail-following pheromones in basal termites, with special reference to Mastotermes darwiniensis. J Chem Ecol 33:1960–1977
Sillam-Dussès D, Sémon E, Robert A, Bordereau C (2009b) (Z)-dodec-3-en-1-ol, a common major component of the trail-following pheromone in the termites Kalotermitidae. Chemoecology 19:103–108
Šobotník J, Jirošová A, Hanus R (2010) Chemical warfare in termites. J Insect Physiol 56:1012–1021
Stránsky K, Jursik T (1996) Simple quantitative transesterification of lipids 1. Introduction. Fett-Lipid 98:65–71
Urbanová K, Vrkoslav V, Valterová I, Háková M, Cvačka J (2012) Structural characterization of wax esters by electron ionization mass spectrometry. J Lipid Res 53:204–213
Wen P, Ji BZ, Liu SW, Liu C, Sillam-Dussès D (2012) Sex-pairing pheromone in the Asian termite pest species Odontotermes formosanus (Isoptera, Termitidae, Macrotermitinae). J Chem Ecol 38:566–575
Wen P, Ji BZ, Sillam-Dussès D (2014) Trail communication is regulated by two trail pheromone components in the fungus-growing termite Odontotermes formosanus Shiraki. PLoS One 9:e90906
Wen P, Mo J, Lu C, Tan K, Šobotník J, Sillam-Dussès D (2015) Sex-pairing pheromone of Ancistrotermes dimorphus (Isoptera: Macrotermitinae). J Insect Physiol 83:81–14
Acknowledgments
This research was supported by the Czech Science Foundation (13-25354P) and by the Institute of Organic Chemistry and Biochemistry AS CR (RVO 61388963). We are grateful to P. Cerdan and the staff of the Laboratoire Environnement HYDRECO of Petit Saut (EDF-CNEH) and to J. Orivel for logistical support as well as to Jarmila Titzenthalerová (IOCB) for her great efforts in electrophysiological experiments and to Lucie Bednárová and Soňa Vašíčková (IOCB) for GC-FTIR analyses. We thank Edward Curtis for proofreading.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Anna Jirošová and David Sillam-Dussès contributed equally to this study.
Electronic Supplementary Material
ESM 1
(PDF 855 kb)
Rights and permissions
About this article
Cite this article
Jirošová, A., Sillam-Dussès, D., Kyjaková, P. et al. Smells Like Home: Chemically Mediated Co-Habitation of Two Termite Species in a Single Nest. J Chem Ecol 42, 1070–1081 (2016). https://doi.org/10.1007/s10886-016-0756-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-016-0756-1