Abstract
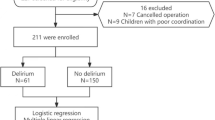
Studies have associated electroencephalogram (EEG) suppression with postoperative delirium (POD) and postoperative cognitive decline (POCD). Otherwise, improving cerebral tissue oxygen saturation (rScO2) seems beneficial. No study has evaluated the impact of EEG suppression and decreased rScO2 on the incidence of POD and POCD when the intraoperative management of patients is performed with a depth-of-anesthesia (DOA) monitor and a cerebral oximetry. In this prospective study patients undergoing cardiac interventions were monitored with the NeuroSENSE® DOA monitor and bilateral cerebral oximetry. An algorithm was used to optimize cerebral oxygenation. EEG suppression was presented as total area under the curve (AUC) of suppression ratio (SR) > 0 s (AUCEEGSR>0s). Cerebral desaturation was defined as AUC of 25% drop of oximetry values as compared to baseline. POD was evaluated by the chart review method. POCD was defined as a Z-score ≤ 2 based on Mini Mental State Examination at baseline and day 5 or if the patient reported any cognitive decline at 3 and at 6 months postoperatively. Among the 1616 patients, 1513 underwent normothermic surgery and were further analyzed. POD and POCD were respectively evaluated in 1504 and 1350 patients of whom 303 (20%) and 270 (20%) were respectively diagnosed positive. Having experienced high magnitudes of EEG suppression (fourth quartile of AUCEEGSR>0s) was significantly associated with POD (OR = 2.247; 95% CI = 1.414–3.571; P = 0.001). Low rScO2 at the end of surgery was statistically associated with POCD (OR = 0.981; 95% CI = 0.965–0.997; P = 0.018). The results of our study show that the degree of intraoperative EEG suppression on one hand, and low rScO2 at the end of procedure on the other hand, are associated with respectively POD and POCD in patients undergoing cardiac interventions.

Similar content being viewed by others
References
Lei L, Katznelson R, Fedorko L, Carroll J, Poonawala H, Machina M, Styra R, Rao V, Djaiani G. Cerebral oximetry and postoperative delirium after cardiac surgery: a randomised, controlled trial. Anaesthesia. 2017;72:1456–66.
Rudolph JL, Marcantonio ER. Review articles: postoperative delirium: acute change with long-term implications. Anesth Analg. 2011;112:1202–11.
Gleason LJ, Schmitt EM, Kosar CM, Tabloski P, Saczynski JS, Robinson T, Cooper Z, Rogers SO Jr, Jones RN, Marcantonio ER, Inouye SK. Effect of delirium and other major complications on outcomes after elective surgery in older adults. JAMA Surg. 2015;150:1134–40.
Witlox J, Eurelings LS, de Jonghe JF, Kalisvaart KJ, Eikelenboom P, van Gool WA. Delirium in elderly patients and the risk of postdischarge mortality, institutionalization, and dementia: a meta-analysis. JAMA. 2010;304:443–51.
Saczynski JS, Marcantonio ER, Quach L, Fong TG, Gross A, Inouye SK, Jones RN. Cognitive trajectories after postoperative delirium. N Engl J Med. 2012;367:30–9.
Sauër AC, Veldhuijzen DS, Ottens TH, Slooter AJC, Kalkman CJ, van Dijk D. Association between delirium and cognitive change after cardiac surgery. Br J Anaesth. 2017;119:308–15.
Sprung J, Roberts RO, Weingarten N, Cavalcante A, Knopman DS, Petersen RC, Hanson AC, Schroeder DR, Warner DO. Postoperative delirium in elderly patients is associated with subsequent cognitive impairment. Br J Anaesth. 2017;119:316–23.
Brown CH 4th, Probert J, Healy R, Parish M, Nomura Y, Yamaguchi A, Tian J, Zehr K, Mandal K, Kamath V, Neufeld KJ, Hogue CW. Cognitive decline after delirium in patients undergoing cardiac surgery. Anesthesiology. 2018;129:406–16.
Steiner LA. Postoperative delirium. Part I: pathophysiology and risk factors. Eur J Anaesthesiol. 2011;28:628–36.
Berger M, Terrando N, Smith K, Browndyke JN, Newman MF, Mathew JP. Neurocognitive function after cardiac surgery: from phenotypes to mechanisms. Anesthesiology 2018; 129:829–51.
Soehle M, Dittmann A, Ellerkmann RK, Baumgarten G, Putensen C, Guenther U. Intraoperative burst suppression is associated with postoperative delirium following cardiac surgery: a prospective, observational study. BMC Anesthesiol. 2015;15:61.
Whitlock EL, Torres BA, Lin N, Helsten DL, Nadelson MR, Mashour MR, Avidan MS. Postoperative delirium in a substudy of cardiothoracic surgical patients in the BAG-RECALL clinical trial. Anesth Analg. 2014;118:809–17.
Fritz BA, Kalarickal PL, Maybrier HR, Muench MR, Dearth D, Chen Y, Escallier KE, Ben Abdallah A, Lin N, Avidan MS. Intraoperative electroencephalogram suppression predicts postoperative delirium. Anesth Analg. 2016;122:234–42.
Radtke FM, Franck M, Lendner J, Krüger S, Wernecke KD, Spies CD. Monitoring depth of anesthesia in a randomized trial decreases the rate of postoperative delirium but not postoperative cognitive dysfunction. Br J Anaesth. 2013;110:98–105.
Sieber FE, Zakriya KJ, Gottschalk A, Blute MR, Lee HB, Rosenberg PB, Mears SC. Sedation depth during spinal anesthesia and the development of postoperative delirium in elderly patients undergoing hip fracture repair. Mayo Clin Proc. 2010;85:18–26.
Chan MT, Cheng BC, Lee TM, Gin T, CODA Trial Group. BIS-guided anesthesia decreases postoperative delirium and cognitive decline. J Neurosurg Anesthesiol. 2013;25:33–42.
MacKenzie KK, Britt-Spells AM, Sands LP, Leung JM. Processed electroencephalogram monitoring and postoperative delirium. A systematic review and meta-analysis. Anesthesiology 2108; 129:417–27.
Murkin JM, Adams SJ, Novick RJ, Quantz M, Bainbridge D, Iglesias I, Cleland A, Schaefer B, Irwin B, Fox S. Monitoring brain oxygen saturation during coronary bypass surgery: a randomized, prospective study. Anesth Analg. 2007;104:51–8.
Zheng F, Sheinberg R, Yee MS, Ono M, Zheng Y, Hogue CW. Cerebral near-infrared spectroscopy monitoring and neurologic outcomes in adult cardiac surgery patients: a systematic review. Anesth Analg. 2013;116:663–76.
Serraino GF, Murphy GJ. Effects of cerebral near-infrared spectroscopy on the outcome of patients undergoing cardiac surgery: a systematic review of randomised trials. BMJ Open 2017; 7(9).
Denault A, Deschamps A, Murkin JM. A proposed algorithm for the intraoperative use of cerebral near-infrared spectroscopy. Semin Cardiothorac Vasc Anesth. 2007;11:274–81.
Purdon PL, Sampson A, Pavone KJ, Brown EN. Clinical electroencephalography for anesthesiologists: part I: background and basic signatures. Anesthesiology. 2015;123:937–60.
Bibian S, Dumont GA, Zikov T. Dynamic behavior of BIS, M-entropy and neuroSENSE brain function monitors. J Clin Monit Comput. 2011;25:81–7.
Momeni M, Baele P, Jacquet LM, Peeters A, Noirhomme P, Rubay J, Docquier MA. Detection by NeuroSENSE cerebral monitor of two major neurologic events during cardiac surgery. J Cardiothorac Vasc Anesth. 2015;29:1013–15.
Zikov T, Bibian S, Dumont GA, Huzmezan M, Ries CR. Quantifying cortical activity during general anesthesia using wavelet analysis. IEEE Trans Biomed Eng. 2006;53:617–32.
Inouye SK, Leo-Summers L, Zhang Y, Bogardus ST Jr, Leslie DL, Agostini JV. A chart-based method for identification of delirium: validation compared with interviewer ratings using the confusion assessment method. J Am Geriatr Soc. 2005;53:312–18.
Chanques G, Garnier O, Carr J, Conseil M, de Jong A, Rowan CM, Ely EW, Jaber S. The CAM-ICU has now a French “official” version. The translation process of the 2014 updated Complete Training Manual of the Confusion Assessment Method for the Intensive Care Unit in French (CAM-ICU fr). Anaesth Crit Care Pain Med. 2017;36:297–300.
Hensel A, Angermeyer MC, Riedel-Heller SG. Measuring cognitive change in older adults: reliable change indices for the Mini-Mental State Examination. J Neurol Neurosurg Psychiatry. 2007;78:1298–303.
de Jager CA, Budge MM, Clarke R. Utility of TICS-M for the assessment of cognitive function in older adults. Int J Geriatr Psychiatry. 2003;18:318–24.
Deschamps A, Lambert J, Couture P, Rochon A, Lebon JS, Ayoub C, Cogan J, Denault A. Reversal of decreases in cerebral saturation in high-risk cardiac surgery. J Cardiothorac Vasc Anesth. 2013;27:1260–6.
Fudickar A, Peters S, Stapelfeldt SG, Leiendecker J, Meybohm P, Steinfath M, Bein B. Postoperative cognitive deficit after cardiopulmonary bypass with preserved cerebral oxygenation: a prospective observational pilot study. BMC Anesthesiol. 2011;14:11:7.
Lu H, Xu F, Rodrigue KM, Kennedy KM, Cheng Y, Flicker B, Hebrank AC, Uh J, Park DC. Alterations in cerebral metabolic rate and blood supply across the adult lifespan. Cereb Cortex. 2011;21:1426–34.
Heringlake M, Garbers C, Käbler JH, Anderson I, Heinze H, Schön J, Berger KU, Dibbelt L, Sievers HH, Hanke T. Preoperative cerebral oxygen saturation and clinical outcomes in cardiac surgery. Anesthesiology. 2011;114:58–69.
Ballard C, Jones E, Gauge N, Aarsland D, Nilsen OB, Saxby BK, Lowery D, Corbett A, Wesnes K, Katsaiti E, Arden J, Amoako D, Prophet N, Puurshothaman B, Green D. Optimised anaesthesia to reduce post operative cognitive decline (POCD) in older patients undergoing elective surgery, a randomised controlled trial. PLoS ONE. 2012;7:e37410.
Scheeren TWL, Kuizenga MH, Maurer H, Struys MMRF, Heringlake M. Electroencephalography and brain oxygenation monitoring in the perioperative period. Anesth Analg. 2018. https://doi.org/10.1213/ANE0000000000002812.
Fritz BA, Maybrier HR, Avidan MS. Intraoperative electroencephalogram suppression at lower volatile anaesthetic concentrations predicts postoperative delirium occurring in the intensive care unit. Br J Anaesth. 2018;121:241–48.
Nishikawa K, Nakayama M, Omote K, Namiki A. Recovery characteristics and post-operative delirium after long-duration laparoscope—assisted surgery in elderly patients: propofol-based vs. sevoflurane-based anesthesia. Acta Anaesthesiol Scand. 2004;48:162–8.
Ischii K, Makita T, Yamashita H, Matsunaga S, Akiyama D, Toba K, Hara K, Sumikawa K, Hara T. Total intravenous anesthesia with propofol is associated with a lower rate of postoperative delirium in comparison with sevoflurane anesthesia in elderly patients. J Clin Anesth. 2016;33:428–31.
Oh CS, Park S, Wan Hong S, Kang WS, Yoon TG, Kim SH. Postoperative delirium in patients undergoing off-pump coronary artery bypass grafting according to the anesthetic agent: a retrospective study. J Cardiothorac Vasc Anesth. 2017;31:1988–95.
Hanning CD. Postoperative cognitive dysfunction. Br J Anaesth. 2005;95:82–7.
Newman MF, Kirchner JL, Phillips-Bute B, Gaver V, Grocott H, Jones RH, Mark DB, Reves JG, Blumenthal JA, Neurological Outcome Research Group and the Cardiothoracic Anesthesiology Research Endeavors Investigators. Longitudinal assessment of neurocognitive function after coronary-artery bypass surgery. N Eng J Med. 2001;344:395–402.
Rudolph JL, Schreiber KA, Culley DJ, McGlinchey RE, Crosby G, Levitsky S, Marcantonio ER. Measurement of post-operative cognitive dysfunction after cardiac surgery: a systematic review. Acta Anaesthesiol Scand. 2010;54:663–77.
Evered L, Silbert B, Knopman DS, Scott DA, DeKosky ST, Rasmussen LS, Oh ES, Crosby G, Berger M, Eckenhoff RG. The Nomenclature Consensus Working Group Anesthesiology. Anesth Analgesia. 2018;129:872–9.
Acknowledgements
The authors would like to thank Prof. Zech for his advise regarding the statistical analyses.
Funding
This study has received a Grant from “The Belgian Society of Anesthesiology and Reanimation” and a research Grant from “The Sorin Group”. This work was supported as well by the Department of Anesthesiology of Cliniques Universitaires Saint Luc, Brussels, Belgium.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Mona Momeni has received speaker’s fees from Medtronic. The other authors declare no conflict of interest.
Ethical approval
Ethical approval for this study (2013/07NOV/508–B403201318880) was provided by Le Comité d’Ethique Hospitalo-Facultaire des Cliniques universitaires Saint Luc, Brussels, Belgium (Chairperson Prof. JM Maloteaux) on 25 November 2013. The trial was registered prior to patient enrollment at Clinicaltrials.gov (NCT02006212; Date of registration: 04/12/2013).
Informed consent
Written informed consent was obtained from all subjects.
Research involving human participants
This study was in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinhi declaration and its later amendments or comparable ethical standards.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Momeni, M., Meyer, S., Docquier, MA. et al. Predicting postoperative delirium and postoperative cognitive decline with combined intraoperative electroencephalogram monitoring and cerebral near-infrared spectroscopy in patients undergoing cardiac interventions. J Clin Monit Comput 33, 999–1009 (2019). https://doi.org/10.1007/s10877-019-00253-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10877-019-00253-8