Abstract
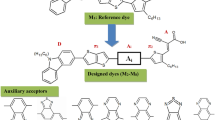
Herein, eight new donor-π-acceptor organic dyes namely M1–M8 have been theoretically investigated for their potential in optoelectronic properties. The M1–M8 were designed through structural modification of π-conjugated bridge of reference reported molecule IC2. The designed molecules contain Indolo[3,2,1-jk]carbazole as core donor unit and end capped cyanoacrylic acid as acceptor unit. DFT and TDDFT calculations using B3LYP, CAM-B3LYP, ωB97XD and M062X functional were performed to evaluate the photophysical and photovoltaic properties. Results indicate that HOMO–LUMO energy gaps in M1–M8 have been found smaller than IC2. Among all, M7 is a material with lowest energy gap 2.61 eV, red shifted absorption wavelength value 436 nm. Results of the calculated redox potential of the ground state, vertical excitation energy of the dye, oxidation potential of the dye in the excited state, free energy change for electron injection, dye regeneration and open circuit photovoltage and light harvesting efficiency indicates that π-bridges in M1–M8 would show better power conversion efficiency than IC2. Especially, dye M7 with π-bridge 5-(thiazol-5yl)thiazole is found to be the most promising candidate for highly effective DSSCs properties. This theoretical framework may provide new ways for experimentalists to design high-performance DSSCs materials for optoelectronic applications.
Graphic Abstract





Similar content being viewed by others
References
S. J. Sweeney and J. Mukherjee Optoelectronic Devices and Materials, Springer Handbook of Electronic and Photonic Materials (Springer, Berlin, 2017), p. 1.
S. Zhang, X. Yang, Y. Numata, and L. Han (2013). Energy Environ. Sci. 6, (5), 1443–1464.
M. K. Nazeeruddin, A. Kay, I. Rodicio, R. Humphry-Baker, E. Müller, P. Liska, N. Vlachopoulos, and M. Grätzel (1993). J. Am. Chem. Soc. 115, (14), 6382–6390.
M. K. Nazeeruddin, P. Pechy, and M. Grätzel (1997). Chem. Commun. 18, 1705–1706.
B. O’regan and M. Grätzel (1991). Nature 353, (6346), 737.
B. Nagarajan, S. Kushwaha, R. Elumalai, S. Mandal, K. Ramanujam, and D. Raghavachari (2017). J. Mater. Chem. A 5, (21), 10289–10300.
M. R. S. A. Janjua, M. U. Khan, B. Bashir, M. A. Iqbal, Y. Song, S. A. R. Naqvi, and Z. A. Khan (2012). Comp. Theor. Chem. 994, 34–40.
M. R. S. A. Janjua, M. Amin, M. Ali, B. Bashir, M. U. Khan, M. A. Iqbal, W. Guan, L. Yan, and Z. M. Su (2012). Eur. J. Inorg. Chem. 2012, (4), 705–711.
M. U. Khan, M. Ibrahim, M. Khalid, M. S. Qureshi, T. Gulzar, K. M. Zia, A. A. Al-Saadi, and M. R. S. A. Janjua (2019). Chem. Phys. Lett. 715, 222–230.
M. U. Khan, M. Ibrahim, M. Khalid, A. A. C. Braga, S. Ahmed, and A. Sultan (2019). J. Cluster Sci. 30, (2), 415–430.
M. U. Khan, J. Iqbal, M. Khalid, R. Hussain, A. A. C. Braga, M. Hussain, and S. Muhammad (2019). RSC Adv. 9, (45), 26402–26418.
F. Wu, L. T. L. Lee, J. Liu, S. Zhao, T. Chen, M. Wang, C. Zhong, and L. Zhu (2015). Synth. Met. 205, 70–77.
R. Soto-Rojo, J. Baldenebro-López, and D. Glossman-Mitnik (2015). Phys. Chem. Chem. Phys. 17, (21), 14122–14129.
S. S. Soni, K. B. Fadadu, J. V. Vaghasiya, B. G. Solanki, K. K. Sonigara, A. Singh, D. Das, and P. K. Iyer (2015). J. Mater. Chem. A 3, (43), 21664–21671.
Z.-S. Huang, H. Meier, and D. Cao (2016). J. Mater. Chem. C 4, (13), 2404–2426.
Y. Li, Y. Li, P. Song, F. Ma, J. Liang, and M. Sun (2017). RSC Adv. 7, (33), 20520–20536.
T. Higashino and H. Imahori (2015). Dalton Trans. 44, (2), 448–463.
J. K. Roy, S. Kar, and J. Leszczynski (2018). Sci. Rep. 8, (1), 10997.
P. Cowper, A. Pockett, G. Kociok-Köhn, P. J. Cameron, and S. E. Lewis (2018). Tetrahedron 74, (22), 2775–2786.
H. Li, M. Fang, R. Tang, Y. Hou, Q. Liao, A. Mei, H. Han, Q. Li, and Z. Li (2016). J. Mater. Chem. A 4, (42), 16403–16409.
H. Qiao, Y. Deng, R. Peng, G. Wang, J. Yuan, and S. Tan (2016). RSC Adv. 6, (74), 70046–70055.
Z. Shen, X. Zhang, F. Giordano, Y. Hu, J. Hua, S. M. Zakeeruddin, H. Tian, and M. Grätzel (2017). Mater. Chem. Front. 1, (1), 181–189.
B. Zhang, G. Shi, Z. Yang, F. Zhang, and S. Pan (2017). Angew. Chem. Int. Ed. 56, (14), 3916–3919.
G. Shi, Y. Wang, F. Zhang, B. Zhang, Z. Yang, X. Hou, S. Pan, and K. R. Poeppelmeier (2017). J. Am. Chem. Soc. 139, (31), 10645–10648.
Z. Yang, B.-H. Lei, W. Zhang, and S. Pan (2019). Chem. Mater. 31, (8), 2807–2813.
X. Chen, B. Zhang, F. Zhang, Y. Wang, M. Zhang, Z. Yang, K. R. Poeppelmeier, and S. Pan (2018). J. Am. Chem. Soc. 140, (47), 16311–16319.
T. Delgado-Montiel, J. Baldenebro-López, R. Soto-Rojo, and D. Glossman-Mitnik (2016). Theor. Chem. Acc. 135, (10), 235.
J. Liu, X. Yang, J. Zhao, and L. Sun (2013). RSC Adv. 3, (36), 15734–15743.
H. Tian, X. Yang, R. Chen, R. Zhang, A. Hagfeldt, and L. Sun (2008). J. Phys. Chem. C 112, (29), 11023–11033.
X. Xie, Z. Liu, W. Li, F.-Q. Bai, E.-C. Lee, and H.-X. Zhang (2019). Chem. Phys. Lett. 719, 39–44.
K.-H. Chang, C.-K. Tai, L. Chen, P.-L. Yeh, and B.-C. Wang (2019). Res. Chem. Intermed. 45, (1), 77–90.
M. Xie, L. Hao, R. Jia, J. Wang, and F.-Q. Bai (2019). New J. Chem. 43, (2), 651–661.
T. Delgado-Montiel, R. Soto-Rojo, J. Baldenebro-López, and D. Glossman-Mitnik (2019). Molecules 24, (21), 3897.
J. B. Henry, S. I. Wharton, E. R. Wood, H. McNab, and A. R. Mount (2011). J. Phys. Chem. A 115, (21), 5435–5442.
J.-H. Tsai, C.-C. Chueh, M.-H. Lai, C.-F. Wang, W.-C. Chen, B.-T. Ko, and C. Ting (2009). Macromolecules 42, (6), 1897–1905.
H.-P. Zhao, F.-Z. Wang, C.-X. Yuan, X.-T. Tao, J.-L. Sun, D.-C. Zou, and M.-H. Jiang (2009). Org. Electron. 10, (5), 925–931.
S. I. Wharton, J. B. Henry, H. McNab, and A. R. Mount (2009). Chem. Eur. J. 15, (22), 5482–5490.
C. Luo, W. Bi, S. Deng, J. Zhang, S. Chen, B. Li, Q. Liu, H. Peng, and J. Chu (2014). J. Phys. Chem. C 118, (26), 14211–14217.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery, J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. J. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski, and D. J. Fox Gaussian 09 (Gaussian Inc, Wallingford, 2009).
Z. Yang, C. Liu, C. Shao, X. Zeng, and D. Cao (2016). Nanotechnology 27, (26), 265701.
Z. Yang, C. Shao, and D. Cao (2015). RSC Adv. 5, (29), 22892–22898.
J. Autschbach (2009). Chemphyschem 10, (11), 1757–1760.
A. Dreuw and M. Head-Gordon (2005). Chem. Rev. 105, (11), 4009–4037.
R. Dennington, T. Keith, and J. Millam Gauss View Version 5 (Semichem Inc, Shawnee Mission, 2009).
M. D. Hanwell, D. E. Curtis, D. C. Lonie, T. Vandermeersch, E. Zurek, and G. R. Hutchison (2012). J. Cheminform. 4, (1), 17.
G.A. Andrienko, Chemcraft. Graphical Software for Visualization of Quantum Chemistry Computations, 2010. https://www.chemcraftprog.com.
N. M. O’boyle, A. L. Tenderholt, and K. M. Langner (2008). J. Comput. Chem. 29, (5), 839–845.
M. U. Khan, M. Khalid, M. Ibrahim, A. A. C. Braga, M. Safdar, A. A. Al-Saadi, and M. R. S. A. Janjua (2018). J. Phys. Chem. C 122, (7), 4009–4018.
M. R. S. A. Janjua (2017). J. Iran. Chem. Soc. 14, (9), 2041–2054.
M. R. S. A. Janjua (2012). Inorg. Chem. 51, (21), 11306–11314.
S. S. Amiri, S. Makarem, H. Ahmar, and S. Ashenagar (2016). J. Mol. Struct. 1119, 18–24.
R. G. Parr, L. Szentpaly, and S. Liu (1999). J. Am. Chem. Soc. 121, (9), 1922–1924.
P. K. Chattaraj, U. Sarkar, and D. R. Roy (2006). Chem. Rev. 106, (6), 2065–2091.
Z. Cai-Rong, L. Zi-Jiang, C. Yu-Hong, C. Hong-Shan, W. You-Zhi, and Y. Li-Hua (2009). J. Mol. Struct: Theochem 899, (1–3), 86–93.
J. Zhang, H.-B. Li, S.-L. Sun, Y. Geng, Y. Wu, and Z.-M. Su (2012). J. Mater. Chem. 22, (2), 568–576.
H. S. Nalwa Handbook of Advanced Electronic and Photonic Materials and Devices: Semiconductors, vol. 1 (Academic Press, Cambridge, 2001).
R. Katoh, A. Furube, T. Yoshihara, K. Hara, G. Fujihashi, S. Takano, S. Murata, H. Arakawa, and M. Tachiya (2004). J. Phys. Chem. B 108, (15), 4818–4822.
T. Marinado, K. Nonomura, J. Nissfolk, M. K. Karlsson, D. P. Hagberg, L. Sun, S. Mori, and A. Hagfeldt (2009). Langmuir 26, (4), 2592–2598.
R. G. Pearson (1988). Inorg. Chem. 27, (4), 734–740.
C.-R. Zhang, L. Liu, J.-W. Zhe, N.-Z. Jin, Y. Ma, L.-H. Yuan, M.-L. Zhang, Y.-Z. Wu, Z.-J. Liu, and H.-S. Chen (2013). Int. J. Mol. Sci. 14, (3), 5461–5481.
G. Boschloo and A. Hagfeldt (2009). Acc. Chem. Res. 42, (11), 1819–1826.
T. Manzoor, S. Asmi, S. Niaz, and A. HussainPandith (2017). Int. J. Quantum Chem 117, (5), e25332.
Acknowledgements
The authors are thankful to the financial support from Saudi Aramco Project # CHEM-2409.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
No conflicts declared.
Associated Content
Optimized Cartesian coordinates of our studied compounds are available in supporting information file
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Janjua, M.R.S.A., Khan, M.U., Khalid, M. et al. Theoretical and Conceptual Framework to Design Efficient Dye-Sensitized Solar Cells (DSSCs): Molecular Engineering by DFT Method. J Clust Sci 32, 243–253 (2021). https://doi.org/10.1007/s10876-020-01783-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-020-01783-x