Abstract
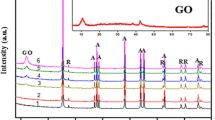
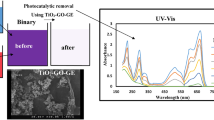
Photocatalytic behavior was investigated for TiO2–graphene nanocomposite in the degradation of acid orange 7 (AO7) as a model pollutant under ultraviolet light in aqueous solution. XRD, SEM, TEM, DRS, FT-IR and EDX techniques were used for the characterization of the prepared nanocomposite. The effect of synthesis variables such as weight ratio of TiO2 to graphene and operational key factors such as initial dye concentration, irradiation time, catalyst dosage and solution distance from UV lamp were studied in the photocatalytic degradation of AO7. This excellent catalytic ability is mainly attributed to the synergic effect of photocatalyst and adsorbent. The effect of operational variables was optimized for the photocatalytic degradation of AO7 as a pollutant model using the RSM technique. In this case, the amount of the determination coefficient (R2 = 0.97) shows that 97% of the variability in the response could be described by the model. The maximum degradation efficiency (96%) was achieved at the optimum operational conditions: catalyst dosage of 0.5 g L−1, the irradiation time of 50 min and distance the solution from UV lamp of 0.3 cm.











Similar content being viewed by others
References
T. Robinson, G. McMullan, R. Marchant, and P. Nigam (2001). Bioresour Technol 77, 247.
B. Y. Chen, M. M. Zhang, C. T. Chang, Y. Ding, K. L. Lin, C. S. Chiou, C. C. Hsueh, and H. Xu (2010). Bioresour Technol 101, 4737.
I. Arslan Alaton and J. L. Ferry (2002). Dyes Pigm 54, 25.
K. Golka, S. Kopps, and Z. W. Myslak (2004). Toxicol. Lett 151, 203.
F. Saadati, N. Keramati, and M. Mehdipour Ghazi (2016). Environmental Science and Technology 46, 757.
X. Li, F. Chen, C. Lian, S. Zheng, Q. Hu, S. Duo, W. Li, and C. Hu (2016). Journal of Cluster Science 27, 1877.
A. R. Nezamzadeh-Ejhieh and A. Shirzadi (2014). Chemosphere 107, 136.
L. Yue, Sh Wang, G. Shan, W. Wu, L. Qiang, and L. Zhu (2015). Applied Catalysis B: Environmental 176, 11.
Z. Xian, R. Liu, H. Li, S. Zhang, Z. Yang, W. Zheng, and C. Chen (2016). Journal of Cluster Science 27, 241.
Z. Yaping, J. Chengguang, P. Ran, M. A. Feng, and O. U. Guangnan (2014). Journal of Central South University 21, 310.
A. Azarian (2015). Journal of Cluster Science 26, 1607.
J. Zhang, G. F. Huang, D. Li, B. X. Zhou, S. Chang, A. Pan, and W. Q. Huang (2016). Appl. Phys. A 122, 994.
X. Wang, Y. Sang, X. Yu, B. Liu, and H. Liu (2016). Appl. Phys. A 122, 884.
A. Alinsafi, F. Evenou, E. M. Abdulkarim, M. N. Pons, O. Zahraa, A. Benhammou, A. Yaacoubi, and A. Nejmeddine (2007). Dyes Pigm 74, 439.
D. Beydoun, R. Amal, G. Low, and S. McEvoy (1999). J. Nanopart. Res 1, 4394.
T. Yoshida, N. Yaghi, R. Nakagou, A. Sugimura, and I. Umezu (2014). Appl. Phys. A, DOI: 10.1007/s00339-014-8378-3
J. Chen, Y. Qian, and X. Wei (2010). J. Mater. Sci 45, 6018.
A. Abbasi, D. Ghanbari, M. Salavati-Niasari, and M. Hamadanin (2016). Journal of Materials Science: Materials in Electronics. doi:10.1007/s10854-016-4361-4.
J. W. Shi, J. T. Zheng, and X. J. Ji (2010). Environmental Engineering Science 27, 923.
B. Paul, W. N. Martens, and R. L. Frost (2012). Applied Clay Science 57, 49.
H. Wang, B. Yang, and W. J. Zhang (2010). Advanced Materials Research 129, 733.
S. Liu, M. Lim, and R. Amal (2014). Chemical Engineering Science 105, 46.
F. Tavakoli and M. Salavati Niasari (2014). J. Ind & Eng chem 20, 3170.
M. Salavati Niasari and F. Tavakoli (2015). J. Ind & Eng chem 21, 1208.
V. Singh, D. Joung, L. Zhai, S. Das, S. Khondaker, and S. Seal (2011). Materials Science 56, 1178.
V. Singh, D. Joung, L. Zhai, S. Das, S. I. Khondaker, and S. Seal (2011). Progress in Materials Science 56, 1178.
X. Zhang, X. Liu, W. Zheng, and J. Zhu (2012). Carbohydrate Polymers 88, 26.
S. Escobedo, B. Serrano, A. Calzada, J. Moreira, and H. D. Lasa (2016). Fuel 181, 438.
I. V. Lightcap, T. H. Kosel, and P. V. Kamat (2010). Nano Lett 10, 577.
H. Zhang, X. J. Lv, Y. M. Li, Y. Wang, and J. H. Li (2010). ACS Nano 4, 380.
Y. H. Zhang, Z. R. Tang, X. Z. Fu, and Y. J. Xu (2010). ACS Nano 4, 7303.
X. Lin, J. Xing, W. Wang, Z. Shan, F. Xu, and F. Huang (2007). J. Phys. Chem. C 111, 18288.
L. Chen, D. Jiang, T. He, Z. Wu, and M. Chen (2013). Cryst. Eng. Commun 15, 7556.
M. A. Behnajady, N. Modirshahla, M. Shokri, H. Elham, and A. Zeininezhad (2008). J. Environ. Sci. Health. Part A Toxic/Hazard. Subst. Environ. Eng 43, 460.
S. Chakrabarti and B. K. Dutta (2004). J. Hazard. Mater 112, 269.
M. A. Behnajady, N. Modirshahla, N. Daneshvar, and M. Rabbani (2007). Chem. Eng. J 127, 167.
L. A. Ghule, A. A. Patil, K. B. Sapnar, S. D. Dhole, and K. M. Garadkar (2011). Toxicol. Environ. Chem 93, 623.
M.A. Behnajady, H. Eskandarloo, Res. Chem. Intermed. http://dx.doi.org/10.1007/s11164-013-1327-5.
B. Neppolian, H. C. Choi, S. Sakthivel, B. Arabindoo, and V. Murugesan (2002). J. Hazard. Mater 89, 303.
S. D. Perera, R. G. Mariano, K. Vu, N. Nour, O. Seitz, Y. Chabal, and K. J. Balkus (2012). ACS Catal 2, 949.
Acknowledgements
Authors are grateful to Council of University of Tehran and Center for International Scientific Studies Collaboration for providing financial support to undertake this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tavakoli, F., Badiei, A., Yazdian, F. et al. Optimization of Influential Factors on the Photocatalytic Performance of TiO2–Graphene Composite in Degradation of an Organic Dye by RSM Methodology. J Clust Sci 28, 2979–2995 (2017). https://doi.org/10.1007/s10876-017-1250-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-017-1250-9