Abstract
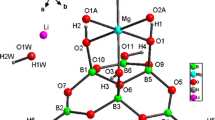
A new zinc borate [Zn(dap)2][B4O6(OH)2] (1, dap = 1,3-diaminopropane) has been hydrothermally synthesized and structurally characterized by IR, elemental analysis, thermogravimetric analysis, powder and single-crystal X-ray diffractions. It crystallized in the monoclinic P21/c space group, a = 7.0050(4) Å, b = 7.8160(2) Å, c = 18.4470(5) Å, β = 94.571(3) Å, V = 1006.78(7) Å3, Z = 4, R 1 = 0.0342, wR 2 = 0.0835. Compound 1 displays a novel two-dimensional inorganic–organic hybrid zinc borate layer constructed from the assembly of [B4O6(OH)2]2− clusters, ZnO2N2 tetrahedra and organic dap molecules. The occurrence of infinite Zn–amine–Zn chains in the structure is uncommon and noteworthy in borate materials.




Similar content being viewed by others
References
P. C. Burns (1995). Can. Mineral. 33, 1167.
P. C. Burns, J. D. Grice, and F. C. Hawthorne (1995). Can. Mineral. 33, 1131.
J. D. Grice, P. C. Burns, and F. C. Hawthorne (1999). Can. Mineral. 37, 731.
C. T. Chen, Y. B. Wang, B. C. Wu, K. C. Wu, W. L. Zeng, and L. H. Yu (1995). Nature. 373, 322.
M. S. Wang, G. C. Guo, W. T. Chen, G. Xu, W. W. Zhou, K. J. Wu, and J. S. Huang (2007). Angew. Chem. Int. Ed. 46, 3909.
M. Touboul, N. Penin, and G. Nowogrocki (2003). Solid State Sci. 5, 1327.
G. M. Wang, Y. Q. Sun, and G. Y. Yang (2004). J. Solid State Chem. 177, 4648.
M. A. Beckett, P. N. Horton, S. J. Coles, and D. W. Martin (2011). Inorg. Chem. 50, 12215.
E. L. Belokoneva, A. G. Ivanova, and O. V. Dimitrova (2006). Russ. J. Inorg. Chem. 51, 869.
D. M. Schubert, M. Z. Visi, S. Khan, and C. B. Knobler (2008). Inorg. Chem. 47, 4740.
M. Z. Visi, C. B. Knobler, J. J. Owen, M. I. Khan, and D. M. Schubert (2006). Cryst. Growth Des. 6, 538.
D. M. Schubert, M. Z. Visi, and C. B. Knobler (2000). Inorg. Chem. 39, 2250.
L. Wei, B.-F. Yang, H. He, and G.-Y. Yang (2014). J. Clust. Sci. 25, 617.
M. A. Beckett (2016). Coord. Chem. Rev. 323, 2.
Z. H. Liu, L. Q. Li, and W. J. Zhang (2006). Inorg. Chem. 45, 1430.
J. Ju, J. H. Lin, G. B. Li, T. Yang, H. M. Li, F. H. Liao, C. K. Loong, and L. P. You (2003). Angew. Chem. Int. Ed. 42, 5607.
L. Cheng, Q. Wei, H. Q. Wu, L. J. Zhou, and G. Y. Yang (2013). Chem. Eur. J. 19, 17662.
L. Cheng and G. Y. Yang (2014). Chem. Commun. 50, 44.
Y. Han, Y. Li, J. Yu, and R. Xu (2011). Angew. Chem. Int. Ed. 50, 3033.
X. H. Bu, P. Y. Feng, and G. D. Stucky (2000). Chem. Mater. 12, 1811.
X. Xu, C. L. Hu, F. Kong, J. H. Zhang, and J. G. Mao (2011). Inorg. Chem. 50, 8861.
H. Chen, Z. B. Yu, Z. Bacsik, H. S. Zhao, Q. X. Yao, and J. L. Sun (2014). Angew. Chem. Int. Ed. 53, 3608.
M. M. Wu, T. S. C. Law, H. H. Y. Sung, J. W. Cai, and I. D. Williams (2005). Chem. Commun. 14, 1827.
D. M. Schubert, F. Alam, M. Z. Visi, and C. B. Knobler (2003). Chem. Mater. 15, 866.
A. Choudhury, S. Neeraj, S. Natarajan, and C. N. R. Rao (2002). Dalton Trans. 7, 1535.
H. Yu, H. Wu, S. Pan, Z. Yang, X. Hou, X. Su, Q. Jing, K. R. Poeppelmeier, and J. M. Rondinelli (2014). J. Am. Chem. Soc. 136, 1264.
G. M. Wang, Y. Q. Sun, and G. Y. Yang (2005). J. Solid State Chem. 178, 729.
P. Zhao, L. Cheng, and G. Y. Yang (2012). Inorg. Chem. Commun. 20, 138.
P. Zhao, Z. E. Lin, Q. Wei, L. Cheng, and G. Y. Yang (2014). Chem. Commun. 50, 3592.
A. K. Paul, K. Sachidananda, and S. Natarajan (2010). Cryst. Growth Des. 10, 456.
Y. He, W. Chen, J. Yang, C. Xi, and J. S. Chen (2006). Chem. Res. Chin. Univ. 22, 271.
S. Natarajan, W. Klein, M. Panthöfer, L. Wüllen, and M. Jansen (2003). Z. Anorg. Allg. Chemie 629, 959.
G. M. Sheldrick (2008). Acta Crystallogr Sect. A: Found. Crystallogr. 64, 112.
G. M. Sheldrick SHELXS-97, Program for Solution of Crystal Structures (University of Göttingen, Germany, 1997).
G. M. Sheldrick SHELXS-97, Program for Solution of Crystal Refinement (University of Göttingen, Germany, 1997).
C. E. Weir (1966). J. Res. Nat. Bur. Stand. Sect. A 70A, 153.
J. Li, S. Xia, and S. Gao (1995). Spectrochim. Acta. 51A, 519.
Z. E. Lin, J. Zhang, Y. Q. Sun, and G. Y. Yang (2004). Inorg. Chem. 43, 797.
L. L. Huang, T. Y. Song, Y. Fan, L. Yang, L. P. Yang, H. Zhang, L. Wang, and J. N. Xu (2010). Microporous Mesoporous Mater. 132, 409.
C. L. Christ and J. R. Clark (1977). Phys. Chem. Miner. 2, 59.
G. Heller (1986). Top. Curr. Chem. 131, 39.
C.-Y. Pan, L.-J. Zhong, F.-H. Zhao, H.-M. Yang, and J. Zhou (2015). Chem. Commun. 51, 753.
Acknowledgements
This work was supported by the Natural Science Foundation of China (20901043, 21571111), Beijing National Laboratory for Molecular Sciences (BNLMS), China Postdoctoral Science Foundation (2016M592130), Postdoctoral Scientific Research Foundation of Qingdao (2016009) and the Taishan Scholar Program (ts201511027).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wei, L., Sun, AH., Xue, ZZ. et al. Hydrothermal Synthesis and Structural Characterization of a New Hybrid Zinc Borate, [Zn(dap)2][B4O6(OH)2]. J Clust Sci 28, 1453–1462 (2017). https://doi.org/10.1007/s10876-017-1158-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-017-1158-4